Incidence rates for liver cancer began rising in the mid-1970s, and they are expected to go up through at least 2030. This report aims to give an overview of liver cancer, as well as explore current therapies, drugs in the pipeline and the future treatment landscape for patients and their caregivers living with liver cancer.
Liver cancer, also known as hepatic cancer and primary hepatic cancer, is a relatively rare form of cancer that has a high mortality rate. As with other cancers, liver cancer starts when cells grow out of control and crowd out normal cells. The liver is made up mainly of cells called hepatocytes, but also includes cells that line its blood vessels and cells that line small tubes in the liver called bile ducts. The different types of cells in the liver can form several types of malignant and benign tumors. These tumors have different causes, are treated differently, and have a different prognosis.
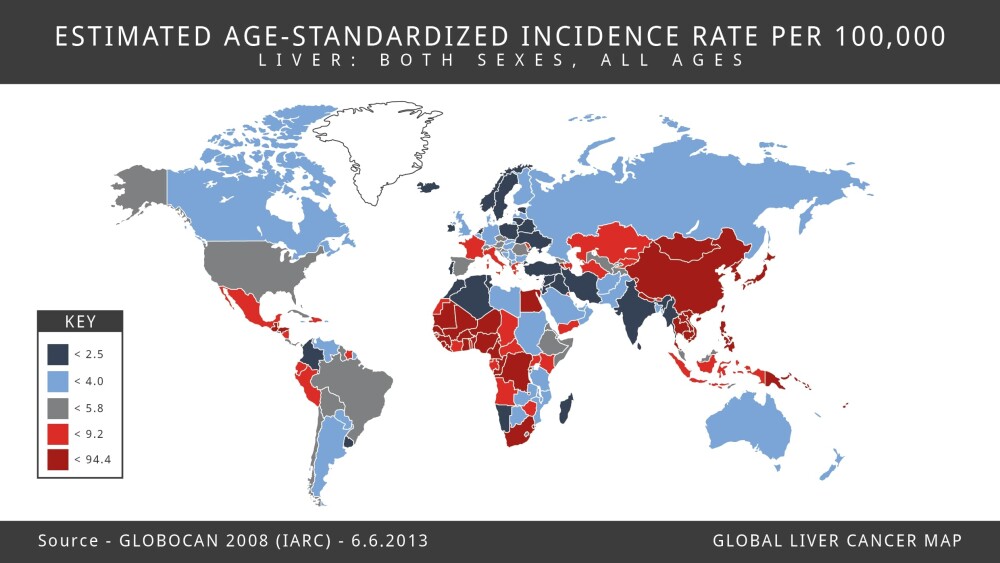
Incidence rates for liver cancer began rising in the mid-1970s, and they are expected to go up through at least 2030. Rates are also increasing globally. A study published in the December 2017 JAMA Oncology found the incidence of liver cancer increased by 75 percent worldwide between 1990 and 2015. Liver cancer has the highest incidence rates in sub-Saharan Africa and Southeast Asia, and the incidence rates in Mongolia far exceed those of any other country.
The American Cancer Society estimates new diagnoses for primary liver cancer and intrahepatic bile duct cancer in the United States for 2020 will be approximately 42,810 new cases (30,170 in men and 12,640 in women). More than 800,000 people are diagnosed with this cancer each year throughout the world. Rates of both incidence and mortality are 2 to 3 times higher among men in most world regions; thus, liver cancer ranks fifth in terms of global cases and second in terms of deaths for males.
Liver cancer is now the fastest increasing cause of cancer death in the U.S. The American Cancer Society estimates that in 2020 about 30,160 people (20,020 men and 10,140 women) will die from primary liver cancer and intrahepatic bile duct cancer. On a global scale, liver cancer was the sixth most commonly diagnosed cancer and the fourth leading cause of cancer death in 2018, with about 841,000 new cases and 782,000 deaths annually.
This report aims to give an overview of liver cancer, as well as explore current therapies, drugs in the pipeline and the future treatment landscape for patients and their caregivers living with liver cancer.
Overview
Liver cancers can be classified into two types. They are either primary, when the cancer starts in the liver itself, or metastatic (secondary), when the cancer has spread to the liver from some other part of the body.
Primary liver cancer can be grouped into the following types, each based on the type of cells that becomes cancerous.
- Hepatocellular carcinoma: this is the most common form of liver cancer in adults. Hepatocellular cancer can have different growth patterns. Some begin as a single tumor that grows larger. Only late in the disease does it spread to other parts of the liver. A second type seems to start as many small cancer nodules throughout the liver, not just a single tumor. This is the most common pattern seen in the United States.
- Intrahepatic cholangiocarcinoma (bile duct cancer): about 10% to 20% of cancers that start in the liver are intrahepatic cholangiocarcinomas. These cancers start in the cells that line the small bile ducts within the liver.
- Angiosarcoma and hemangiosarcoma: These are rare cancers that begin in cells lining the blood vessels of the liver. These are fast growing tumors and are usually too widespread to be surgically removed by the time they are discovered. Chemotherapy and radiation therapy may help slow the disease, but these cancers are usually very hard to treat.
- Hepatoblastoma: This is a very rare kind of cancer that develops in children, usually in those younger than 4 years old. About 2 out of 3 children with these tumors are treated successfully with surgery and chemotherapy, although the tumors are harder to treat if they have spread outside the liver.
Secondary liver cancer (metastatic liver cancer): Most of the time when cancer is found in the liver it did not start there, but has metastasized from somewhere else in the body, such as the pancreas, colon, stomach, breast or lung. These tumors are named and treated based on their primary site. In the United States and Europe, metastatic liver tumors are more common than primary liver cancer. The opposite is true for many areas of Asia and Africa.
Causes and Risk Factors
Liver cancer happens when liver cells develop mutations in their DNA. One result of these mutations is that cells may begin to grow out of control and eventually form a tumor — a mass of cancerous cells. Various factors can increase the risk of developing liver cancer. These include the following:
- Chronic hepatitis B or C infection. Worldwide this is the most significant risk factor for liver cancer. These viruses can spread from person to person through sharing contaminated needles, through unprotected sex and through childbirth.
- Cirrhosis
- Certain inherited liver diseases. These may include hemochromatosis and Wilson’s disease.
- Certain inherited metabolic diseases
- Diabetes and/or obesity
- Nonalcoholic fatty liver disease
- Exposure to aflatoxins. Aflatoxins are poisons produced by molds that grow on crops that are stored poorly. Crops can become contaminated with aflatoxins and end up in foods made of these products. In the United States, safety regulations limit aflatoxin contamination. Aflatoxin contamination is more common in certain parts of Africa and Asia.
- Exposure to arsenic
- Excessive alcohol consumption
Life Expectancy
The life expectancy for patients with liver cancer depends on the stage and location of the cancer. In general, survival rates are higher for people who can have surgery to remove their cancer, regardless of the stage. The American Cancer Society relies on information from the SEER database, maintained by the National Cancer Institute (NCI), to provide survival statistics for different types of cancer. The SEER database tracks 5-year relative survival rates for liver cancer in the United States, based on how far the cancer has spread. The relative 5-year survival rates for people diagnosed with liver cancer are as follows, based on group (cancer stage):
- Localized means the cancer is still confined to the liver, and includes stages I, II, and some stage III cancers. This includes a wide range of cancers, some of which are easier to treat than others. The 5-year relative survival rate for people with localized liver cancer is about 31%.
- Regional means the cancer has grown into nearby organs or has spread to nearby lymph nodes and includes stages IIIC and IVA cancers. For regional stage liver cancer, the 5-year survival rate is about 11%.
- Distant means that the cancer has spread to distant organs or tissues and is the same as stage IVB. The 5-year relative survival rate for distant stage liver cancer is about 3%.
Annual Cost
Liver cancer treatment typically involves surgery, which can involve partial liver removal or total removal with a transplant. Other treatments include cancer-targeting drugs and radiation. Typical costs may include the following:
- For patients with health insurance, out-of-pocket costs for liver cancer treatment typically consist of doctor visit, lab and prescription drug copays as well as coinsurance of 10%-50% for surgery and other procedures. Liver cancer treatment typically is covered by health insurance, although individual drugs or treatments might not be covered by some plans.
- For patients not covered by health insurance, liver cancer treatment can cost up to $50,000 or more for targeted radiation therapy. It can cost about $50,000-$100,000 or more for partial removal of the liver, depending on the provider and whether there are complications. It can cost $150,000 or more for a year of treatment with targeted therapy drugs, and more than $500,000 if a transplant is required.
- Stereotactic radiosurgery, a targeted radiation treatment sometimes used for liver cancer, typically costs $12,000-$55,000.
- A liver transplant, sometimes used as a treatment when cancer has not spread outside the liver, can cost up to $575,000 or more, including pre-transplant screening, donor matching, the surgery and the first six months of medication. If a transplant is required, anti-rejection drugs will need to be administered for the remainder of the patient’s life. These drugs can include the anti-rejection drug Prograf as well as Prednisone and can cost more than $3,000 per month. These drugs are typically covered by health insurance.
Diagnostic Strategies
Most liver cancers are found because they are causing symptoms, which may include unintended weight loss, loss of appetite, nausea or vomiting, an enlarged liver, an enlarged spleen, pain in the abdomen or near the right shoulder blade, swelling or fluid build-up in the abdomen, itching and jaundice. If symptoms and/or the results of a physical exam suggest liver cancer may be present, tests will likely be done. These might include imaging tests, lab tests, and/or biopsies of liver tissue.
- Ultrasound is often the first test used to look at the liver. This test can show tumors growing in the liver, which then can be tested for cancer if needed.
- Imaging tests use x-rays, magnetic fields, or sound waves to create pictures of the inside of the body.
- A Computed Tomography (CT) Scan is an x-ray test that makes detailed images. A CT scan of the abdomen can help find many types of liver tumors. It can give specific information about the size, shape, and location of any tumors in the liver or elsewhere in the abdomen, as well as nearby blood vessels.
- Magnetic resonance imaging (MRI) scans provide detailed images of soft tissues in the body by using radio waves and strong magnets instead of x-rays. Sometimes they can tell a benign tumor from a malignant one. They can also be used to look at blood vessels in and around the liver to see any blockages and can help show if liver cancer has spread to other parts of the body.
- An angiogram is an x-ray test that looks at blood vessels. Contrast medium, or a dye, is injected into an artery to outline blood vessels while x-ray images are taken.
- Angiography can be used to show the arteries that supply blood to liver cancer, which can help doctors decide if cancer can be removed and to help plan the operation. It can also be used to help guide some types of non-surgical treatment.
Other types of tests may be done if the imaging results are uncertain. These may include the following:
- Biopsy: A biopsy is the removal of a sample of tissue to see if it is cancer. Sometimes, the only way to be sure that liver cancer is present is to take a biopsy and look at it in the pathology lab.
- Lab Tests can help to diagnose liver cancer and help to determine what might have caused the liver cancer. Lab tests can also determine how well the liver and other organs are functioning, which can affect the course of treatment.
- Alpha-fetoprotein blood (AFP) test. AFP is a protein that can be found in high levels in adults with liver disease, liver cancer, who are pregnant, or other cancers. If AFP levels are very high in someone with a liver tumor, it can be a sign that liver cancer is present. This test is also useful in people already diagnosed with liver cancer. The AFP level can help determine treatment options. During treatment, the test can be used to give an idea of how well it is working, as the AFP level should go down if treatment is effective. The test can also be used after treatment, to look for possible signs that the cancer has recurred.
- Other tests may include tests for viral hepatitis, liver and kidney function, blood clotting and complete blood count.
Current Therapies
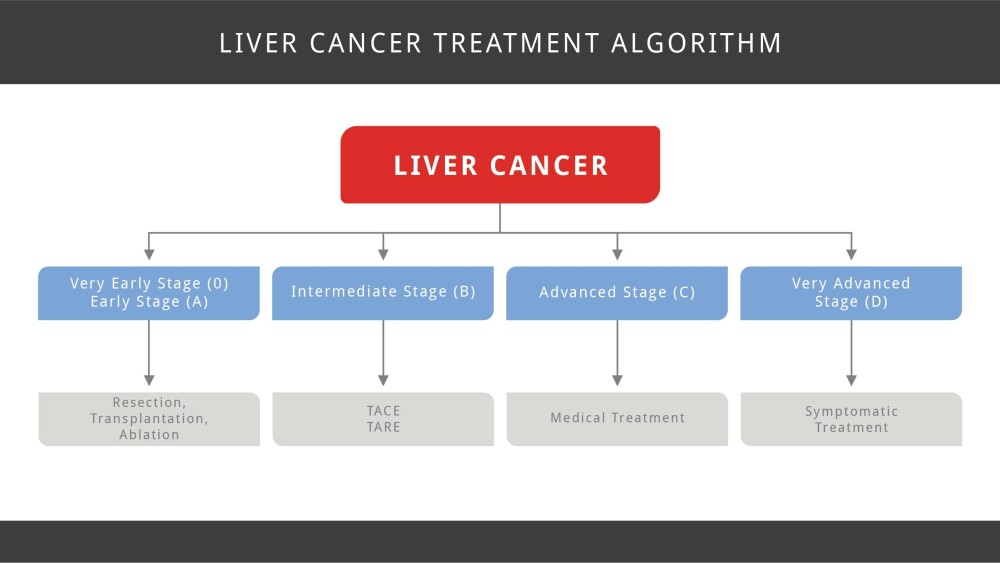
Treatment for liver cancer can include surgery, ablation, embolization therapy, radiation, targeted therapy, chemotherapy and immunotherapy. Treatment is determined by the stage of liver cancer.
- Surgery: The best option to cure liver cancer is with either surgical resection (removal of the tumor with surgery) or a liver transplant.
- Ablation: Ablation is a treatment that destroys liver tumors without removing them. These techniques can be used in patients with a few small tumors and when surgery is not a good option (often because of poor health or reduced liver function).
- Embolization is a procedure that injects substances directly into an artery in the liver to block or reduce the blood flow to a tumor in the liver. Embolization therapy is used for patients who cannot have surgery to remove the tumor or ablation therapy and whose tumor has not spread outside the liver.
- Radiation therapy uses high-energy rays to kill cancer cells.
- Chemotherapy
- Targeted Therapies: this is a type of cancer treatment that uses drugs or other substances to more precisely identify and attack cancer cells while bypassing healthy cells. Targeted therapies can also help other therapies, such as chemotherapy, identify and target cancer cells. Targeted therapies to treat liver cancer include Bayer’s Nexavar (sorafenib) and Eisai’s Lenvima (lenvatinib). In some cases, sorafenib becomes ineffective other treatments are used in its place. These include Bayer‘s Stivarga (regorafenib), Cabometyx (cabozantinib), manufactured by Exelixis and Eli Lilly’s Cyramza (ramucirumab).
- Immunotherapies, also called immune checkpoint inhibitors, use a person’s own immune system to find and destroy cancer cells. Examples of immunotherapies include Merck’s Keytruda (pembrolizumab) and Bristol-Myers Squibb’s Opdivo (nivolumab). Both drugs target PD-1, a protein on immune system cells called T cells that normally helps keep these cells from attacking other cells in the body. By blocking PD-1, these drugs boost the immune response against cancer cells. This can shrink some tumors or slow their growth. These drugs can be used in people with liver cancer who have previously been treated with the targeted drug sorafenib (Nexavar).
Drug Pipeline
Note: This report is not all-inclusive and may unintentionally exclude some drugs in development or clinical trials.
A variety of therapies are currently being explored through clinical trials. In addition to the treatment options, targeted therapies and immunotherapies listed above, common targets and mechanisms include the following:
- Alpha-fetoprotein (AFP) inhibitors: Alpha-fetoprotein (AFP) is a diagnostic marker for hepatocellular carcinoma (HCC). A direct relationship between poor prognosis and the concentration of serum AFP has been observed. Elevated AFP concentrations are seen in approximately 70% of patients with hepatocellular carcinoma.
- Multi-kinase inhibitors work by inhibiting multiple intracellular and cell surface kinases, some of which are implicated in tumor growth and metastatic progression of cancer, thus decreasing tumor growth and replication.
- Fibroblast growth factor receptor 4 (FGFR4) inhibitors: FGFR4 is a tyrosine kinase receptor for FGFs and is involved in diverse cellular processes. High activation of FGFR4 is strongly associated with the amplification of its specific ligand FGF19 in many types of solid tumors and hematologic malignancies, where it acts as an oncogene driving the cancer development and progression.
- PD-1 antibodies: Programmed cell death protein 1 (PD-1) is an inhibitory receptor that is expressed on some tumor cells and causes down regulation of the immune system by reducing T-cell activity. Anti-PD-1 monoclonal antibodies block the PD-1 receptor, so the T cells are no longer inhibited and therefore activate the immune response against the tumor.
- TGF beta: The transforming growth factor-β (TGF-β) is a family of structurally related proteins that control numerous cellular functions. in late stages of cancer TGF-β exerts tumor promoting effects, increasing tumor invasiveness, and metastasis. Elevated TGF-β activity has been associated with poor clinical outcome.
- A3 adenosine receptor (A3AR) agonists: A3AR mediates anti-inflammatory, anticancer and anti-ischemic protective effects. The receptor is overexpressed in inflammatory and cancer cells.
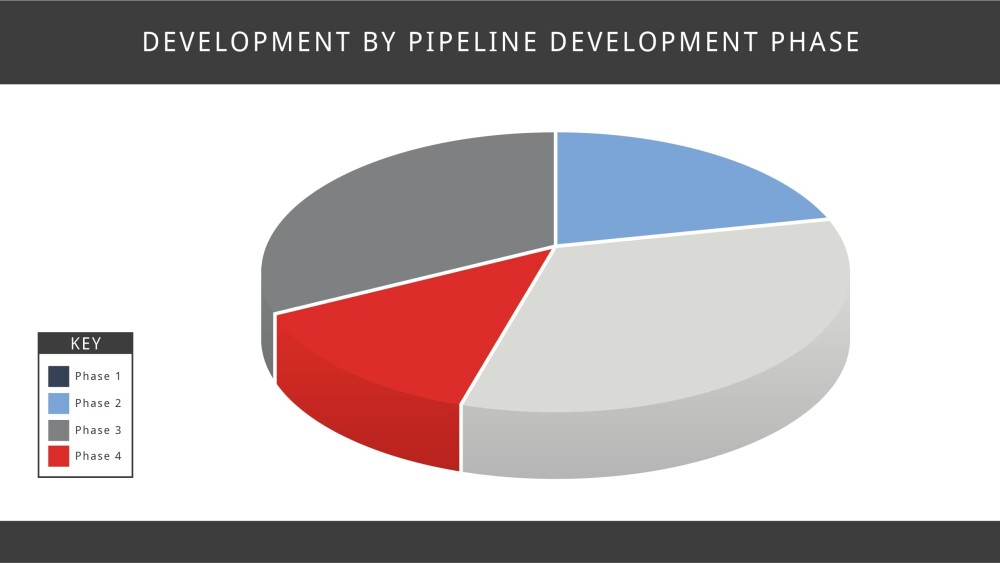
As of January 2020 there were 892 clinical trials (Recruiting, Not yet recruiting, Active, not recruiting) listed on clinicaltrials.gov. Of these: 255 are industry-led and funded studies and the remainder are funded by the NIH/U.S. govt. agency or are funded by universities, hospitals, etc. Studies using various interventions account for 721 studies and the remainder are observational. When broken down by location: 315 are in the United States and the remainder are across global sites. When broken down by phase, there are 192 trials in phase 1, Phase 2 has 285 trials and 115 trials are in phase 3. The remainder are in phase 4 or are other types of studies, including behavioral modification or observational studies, those using dietary supplements and various devices.
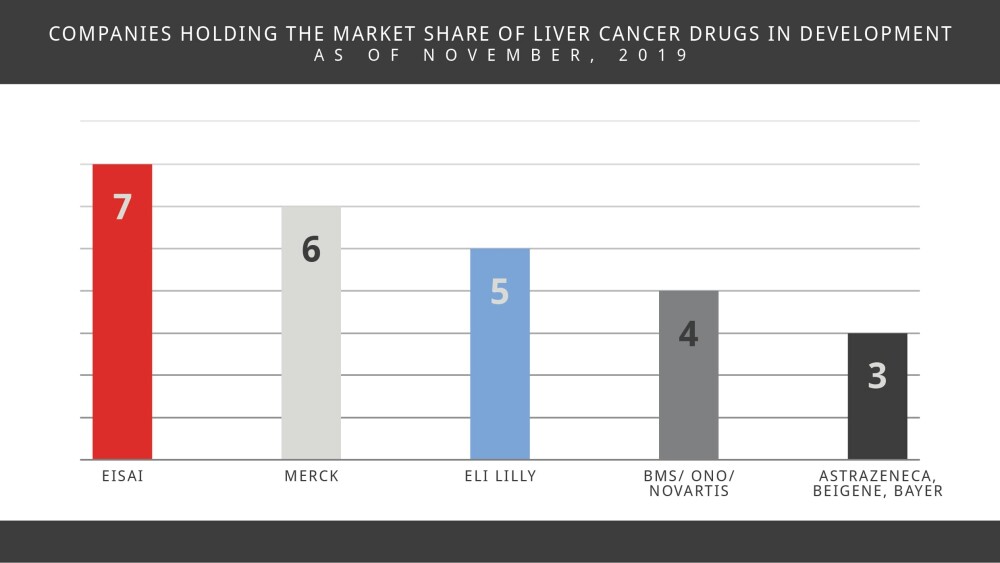
The liver cancer market contains a mixture of big and smaller sized pharma and biotech companies. The market share is dominated by Eisai, with 7 studies underway, followed by Merck (6 studies), Eli Lilly (5 studies), Bristol-Myers Squibb/Ono Pharmaceuticals and Novartis (4 studies each). AstraZeneca, BeiGene and Bayer bring up the rear, with 3 studies each. Note: These are industry-led and funded studies.
The following analysis of selected drugs in the pipeline will briefly discuss how each drug works and where it is in clinical trials. This information was up to date as of November 2019. Any text in italics represents failed or terminated trials.
Note: This section is not meant to be completely comprehensive and may unintentionally exclude some drugs in development or clinical trials, especially those trials outside of the United States.
Eisai is evaluating lenvatinib, a kinase inhibitor, as monotherapy and in various combinations across 7 studies, both phase 1 and phase 3. Lenvatinib has been approved by the FDA as Lenvima for use in patients with thyroid cancer, renal cell carcinoma and inoperable hepatocellular carcinoma.
Merck is evaluating pembrolizumab and tepotinib across 6 studies, including phase 1/2, phase 2 and phase 3. Pembrolizumab is a programmed death receptor-1 (PD-1)-blocking antibody and has been approved as Keytruda for a variety of cancer indications. These studies are evaluating pembrolizumab as monotherapy and in combination with lenvatinib. Merck is also investigating tepotinib, an inhibitor of the c-Met receptor tyrosine kinase. Results from the ongoing phase 1/2 trial were reported in June of 2018 showing the primary endpoint of time to progression was met.
Eli Lilly has two drugs in development for hepatocellular carcinoma across 5 studies, galunisertib (LY2157299) and ramucirumab. Galunisertib is a potent small-molecule antagonist that specifically inhibits Transforming Growth Factor-β receptor I (TβRI). Results from a phase 2 trial were published in April of 2019 in the journal Liver International. In this trial galunisertib treatment had a manageable safety profile in patients with HCC. Lower baseline AFP and a response in AFP or TGF‐β1 levels (vs no response) correlated with longer survival. Ramucirumab is a human vascular endothelial growth factor receptor 2 (VEGFR2) antagonist. It has been approved as Cyramza for the treatment of gastric cancer, non-small cell lung cancer, colorectal cancer and AFP-high hepatocellular carcinoma in patients who have been treated with sorafenib.
Bristol-Myers Squibb in collaboration with Ono Pharmaceuticals is evaluating nivolumab as monotherapy and in combination across 4 studies. Nivolumab is a programmed death receptor-1 (PD-1) blocking antibody. It has been approved as Opdivo for a variety of cancers, including accelerated approval for patients with hepatocellular carcinoma (HCC) who have been previously treated with sorafenib.
Novartis currently has 4 studies underway evaluating PDR001 as a single agent or in combinations in phase 1, 1/2 and 2 trials. PDR001 is a fully humanized monoclonal antibody designed to inhibit the negative immunoregulatory receptor programmed death-1 (PD-1). It is being evaluated in combination with the following: Novartis monoclonal antibody NIS 793, which targets transforming growth factor beta (TGF-β), Capmatinib (INC280) is an oral, highly potent and selective MET inhibitor licensed to Novartis by Incyte Corporation in 2009, and sorafenib (Nexavar).
AstraZeneca is currently conducting three phase 3 studies to evaluate durvalumab, a human immunoglobulin G1 kappa (IgG1κ) monoclonal antibody that blocks the interaction of programmed cell death ligand 1 (PD-L1) with the PD-1. Durvalumab has been approved as Imfinzi for non-small cell lung cancer. The current studies include EMERALD-2 (durvalumab in combination with bevacizumab or durvalumab alone), EMERALD-1 (transarterial chemoembolization (TACE) in combination with durvalumab and bevacizumab) and HIMALAYA (durvalumab plus tremelimumab combination therapy and durvalumab monotherapy versus sorafenib). The safety and clinical activity of durvalumab monotherapy were reported in June of 2017.
Bayer has 3 studies underway. Two of these studies are using regorafenib (Stivarga) and include REFINE, to evaluate regorafenib (Stivarga) under real-world practice conditions in patients diagnosed with unresectable hepatocellular carcinoma, and a phase 1b in collaboration with Merck to evaluate the combination of regorafenib and pembrolizumab (Keytruda) in patients with advanced HCC with no prior systemic therapy. The third study is evaluating copanlisib- a kinase inhibitor, in combination with Opdivo (nivolumab) in patients with solid tumors.
BeiGene is evaluating tislelizumab (BGB-A317) - a humanized monoclonal antibody against the immune checkpoint receptor PD-1, and sitravatinib- a spectrum-selective kinase inhibitor which potently inhibits receptor tyrosine kinases, including RET, TAM family receptors (TYRO3, Axl, MER), and split family receptors (VEGFR2, KIT). Three clinical trials are currently underway evaluating tislelizumab monotherapy (phase 2), tislelizumab in combination with sorafenib (phase 3) and sitravatinib as monotherapy and in combination with tislelizumab (phase 1/2).
Phase 1
Adaptimmune is conducting a phase 1 study of ADP-A2AFP, a SPEAR T-cell product targeting alpha-fetoprotein (AFP). The trial is evaluating genetically changed T cells that target AFP in patients who have a blood test positive for appropriate HLA-A*02 and have adequate AFP protein in blood or tumor, and whose noncancerous liver tissue has very little AFP protein.
Amgen and Merck are collaborating on MASTERKEY-318, which is studying talimogene laherparepvec when injected into liver tumors alone and in combination with systemically administered pembrolizumab (Keytruda) in patients with hepatocellular carcinoma. Talimogene laherparepvec is an oncolytic immunotherapy derived from herpes simplex virus type 1 (HSV-1). It has been approved as Imlygic for the treatment of melanoma.
Blueprint Medicines has developed fisogatinib (BLU-554), an orally administered, potent, irreversible inhibitor of FGFR4. Fisogatinib was specifically designed to selectively inhibit FGFR4 and spare other kinases. Blueprint Medicines is working to develop fisogatinib for the treatment of patients with FGFR4-driven HCC. A phase 1 trial is ongoing and is expected to be complete in October of 2020.
Chipscreen Biosciences is evaluating Chiauranib, which simultaneously targets against VEGFR/Aurora B/CSF-1R, several key kinases involved in tumor angiogenesis, tumor cell mitosis, and chronic inflammatory microenvironment. Chiauranib showed broad preclinical anti-tumor efficacy and the potential to improve conventional TKI kinase inhibitor therapy in various cancer indications. The current phase 1b trial is enrolling patients with advanced hepatocellular carcinoma.
Immatics has developed IMA202, an ACTengine® product based on genetically engineering a patient’s own T cells to express an exogenous T-cell receptor (TCR) upon lentiviral transduction. This approach essentially “reprograms” a patient’s T cells to attack tumor cells. A phase 1 trial is enrolling patients with solid tumors that express melanoma-associated antigen 1 (MAGEA1). The trial is being conducted in collaboration with the M.D. Anderson Cancer Center.
Incyte is currently enrolling patients in an open label phase I study to evaluate INCB062079 in subjects with advanced hepatocellular carcinoma and other malignancies. INCB062079 is an orally bioavailable, selective inhibitor of human FGFR4, with potential antineoplastic activity.
Provectus Biopharmaceuticals is investigating an injectable formulation of rose bengal disodium, called PV-10. Provectus has shown that PV-10 selectively accumulates in the lysosomes of only cancer cells upon contact, disrupts them, and causes the cancer cells to die. A phase 1 trial is evaluating a single intralesional injection of PV-10 in subjects with either (a) hepatocellular carcinoma (HCC) that is not amenable to resection, transplant or other potentially curative therapy or (b) cancer metastatic to the liver.
Therapure Biopharma is in the early stages of testing TBI 302, a hemoglobin-conjugated floxuridine compound. Floxuridine is a potent chemotherapy drug used for the treatment of liver tumors. Therapure’s protein drug conjugation platform technology takes advantage of hemoglobin, a natural protein the body uses to transport oxygen to tissues. Linking drugs to hemoglobin may provide improved delivery and transport of drugs to specific sites of action, as well as minimize exposure to non-target tissues.
Phase 1/2
Abivax is studying ABX196, a synthetic glycolipid agonist of invariant natural killer T cells (iNKT) in a liposomal formulation. A phase 1 clinical trial in healthy subjects has been completed and demonstrated safety and tolerability as well as a potent activation of iNKT cells. The current study is evaluating ABX196 administered in combination with nivolumab.
AVEO Oncology, in collaboration with AstraZeneca, is evaluating the combination of tivozanib with durvalumab. Tivozanib is an oral, once-daily VEGF tyrosine kinase inhibitor (TKI). Tivozanib was licensed from Kyowa Kirin in 2006 and has been approved for advanced renal cell carcinoma (RCC) in the European Union plus Norway, New Zealand and Iceland. Interim results from a separate phase 1/2 study were presented in January of 2018 and showed efficacy and safety of the combination.
Boston Biomedical has two drugs in the pipeline: napabucasin (BBI-608) and amcasertib (BBI-503). Two clinical trials are currently underway for hepatocellular carcinoma, although no longer recruiting patients. Napabucasin is an orally administered agent designed to inhibit cancer stemness pathways by targeting STAT3. Amcasertib is an orally administered agent designed to inhibit cancer stemness pathways, including Nanog, by targeting stemness kinases.
Eureka Therapeutics is moving forward with their autologous T-cell product (ET140202) in adult subjects with advanced Alpha-fetoprotein (AFP) positive/Human Leukocyte Antigen (HLA) A-2 positive Hepatocellular Carcinoma (HCC). ET140202 autologous T cells are genetically modified to carry a TCR-mimic (TCRm) construct capable of mediating cell killing by targeting tumor specific intracellular antigens. Data presented in September of 2018 from an ongoing proof-of-concept first-in-human study of ET140202 in China demonstrated a favorable safety profile with no observed cytokine release syndrome or drug-related neurotoxicity.
Genoscience Pharma has developed GNS561, a quinoline derivative. These are a novel class of oral small molecules able to induce multiple cellular effects such as inhibition of autophagy, induction of apoptosis, and cell cycle modulation. The ongoing phase 1/2 trial is expected to be complete in January 2020.
Medivir is conducting a trial to evaluate MIV-818, a liver-targeted nucleotide prodrug of troxacitabine. It has been developed to be an orally administered therapeutic with a high level of anti-tumor activity that is targeted for delivery to the liver. The trial is enrolling patients with hepatocellular carcinoma, as well as patients with intrahepatic cholangiocarcinoma and patients with liver metastases from solid tumors.
OBI Pharma is investigating OBI-3424, a first-in-class novel small-molecule prodrug that selectively targets cancers overexpressing the enzyme aldo-keto reductase 1c3 (AKR1C3) and selectively releases a potent DNA alkylating agent in the presence of the AKR1C3 enzyme. AKR1C3 overexpression has been documented in a number of treatment-resistant and difficult to treat cancers including hepatocellular carcinomas. A phase 1/2 trial was initiated in June of 2018 in patients with solid tumors, including HCC and castrate-resistant prostate cancer.
Polaris Pharma is assessing ADI-PEG 20 (pegylated arginine deiminase), which exerts its effect by breaking down the amino acid arginine which blocks the growth of cancer cells that need arginine to grow. The phase 1 and 1/2 trials are evaluating ADI-PEG 20 in a variety of tumors types, including HCC.
Teclison has developed a hypoxic tissue-selective cytotoxin called tirapazamine (TPZ). TPZ, when combined with embolization (TATE), creates a persistent hypoxic environment selectively in the liver tumor to activate the compound. Conversely normal liver or other organs do not lack oxygen, and thus are not able to activate the compound and side effects in normal organs are avoided. Three clinical trials are active, although no longer recruiting patients.
Phase 2
Can-Fite BioPharma has advanced namodenoson (CF-102), a small orally bioavailable drug that binds with high affinity and selectivity to the A3 adenosine receptor (A3AR). In September of 2019 Can-Fite concluded a successful End-of-Phase II Meeting with the U.S. FDA regarding a recently completed Phase II study in the treatment of HCC. The FDA agreed with Can-Fite’s proposed pivotal Phase III trial design to support a New Drug Application submission and approval. Can-Fite expects to initiate the study in the second half of 2020.
Innovent Biologics is conducting a phase 2/3 study (ORIENT-32) to evaluate the combination of sintilimab and IBI305. Sintilimab is a fully humanized IgG4 monoclonal antibody. It can specifically bind to PD-1 expressed on the surface of T cells to block this immune escape pathway, reactivate T cells, and release cytokines to kill tumor cells. IBI302 is a biosimilar product candidate of bevacizumab and a recombinant humanized anti-VEGF monoclonal antibody for injection.
Regeneron in collaboration with Sanofi is evaluating neoadjuvant cemiplimab therapy in a phase 2 trial in patients with resected tumors, including HCC. Cemiplimab is a programmed death receptor-1 (PD-1) blocking antibody. It has been approved as Libtayo for the treatment of patients with metastatic cutaneous squamous cell carcinoma.
TaiRx is evaluating the combination of CVM-1118 with Sorafenib. CVM-118 targets the formation of vasculogenic mimicry (VM). VM has been associated with tumor metastasis and poor clinical outcomes. VM is reported to be particularly active in tumor under hypoxia state when patients are treated with the potent vascular endothelial growth factor (VEGF) inhibitor like sorafenib.
Yiviva has developed YIV-960 (PHY906, KD018), an oral form of a spray dried aqueous extract of four herbs, which have been used in the Orient for more than 1800 years for a variety of gastrointestinal symptoms including diarrhea, nausea, and vomiting. Extensive clinical and pre-clinical research has indicated that YIV-906 may have synergistic anticancer activities as well as GI toxicity reduction to cancer treatments, including sorafenib. The planned phase 2 trial will investigate the efficacy and mechanisms of YIV-906 as an adjuvant to sorafenib in the treatment of patients with Hepatitis B(+)-associated advanced hepatocellular carcinoma.
Phase 3
Exelixis is evaluating cabozantinib in combination with atezolizumab (Roche) in a phase 3 trial, COSMIC-312, and a phase 1/2 trial, COSMIC-021. Cabozantinib is a a targeted agent that inhibits the activity of receptor tyrosine kinases including MET, AXL, VEGF Receptors, and RET. Data from the phase III CELESTIAL trial were presented as an oral presentation at the 2018 ASCO GI Cancers Symposium and in a poster at the 2018 ASCO Annual Meeting. Compared with placebo, cabozantinib significantly improved overall survival and progression-free survival in patients with advanced hepatocellular carcinoma who were previously treated with sorafenib (Nexavar), with no new safety signals.
Immunitor has developed once-daily oral immunotherapeutic vaccine tablet, hepcortespenlisimut-L (Hepko-V5). Results from a phase 2 trial were published in the open access Journal of Hepatocellular Carcinoma in April of 2017. The open-label immunotherapeutic vaccine was evaluated in 75 patients with late stage HCC and 90% of patients were alive after a median 12 months of follow up. In addition, after a median 2 months of treatment 50 out of 75 patients had their AFP decline, which indicates that two thirds of the study population responded to immunotherapy, since the decrease in AFP was correlated with tumor shrinkage.
Jiangsu Hengrui Medicine Co. is investigating camrelizumab (SHR-1210), a humanized monoclonal antibody recognizing the programmed death-1 (PD-1) receptor. A phase 2 study is recruiting patients with primary liver or biliary cancer to examine SHR-1210 in combination with apatinib (a tyrosine kinase inhibitor that selectively inhibits VEGFR2), or chemotherapy (FOLFOX4 or GEMOX regimen). A phase 3 study is also recruiting patients with advanced HCC who have never received prior systemic treatment.
Patients in this study will receive SHR-1210 plus FOLFOX4. Results from a phase 2 trial showed camrelizumab plus FOLFOX4 or GEMOX chemotherapy was tolerable and showed promising signs of efficacy, based on the objective response rate and disease control rate.
Roche is evaluating atezolizumab - a fully humanized, engineered monoclonal antibody of IgG1 isotype against the protein PD-L1, in combination with bevacizumab- a vascular endothelial growth factor inhibitor, in 2 phase 3 studies, IMbrave050 and IMbrave150. Results from IMbrave150 were reported in October 2019 and showed the combination led to meaningful benefits by extending overall survival and prolonging progression-free survival.
Atezolizumab has been approved as Tecentriq for lung cancer and bevacizumab has been approved as Avastin for a variety of cancers.
Preclinical Candidates
The following is a sampling of companies that are developing and investigating drugs and other therapeutics in the preclinical laboratory setting. Data gathered from these preclinical trials will be used to determine whether the drug/therapy will move forward with clinical testing in humans.
Auransa, an artificial intelligence (AI)-driven biotechnology company developing precision medicines, is developing lead drug candidate AU-409, which was generated leveraging the company’s proprietary AI-driven SMarTR™ Engine. The SMarTR Engine tackles disease heterogeneity and leverages gene expression profiles to predict responder patient populations, as well as compounds that might be effective against those patient populations. Preclinical data were presented in September of 2019 at the 13th Annual Conference of the International Liver Cancer Association (ILCA) in Chicago. The study results demonstrated promising antitumor activity for AU-409 in a preclinical orthotopic liver cancer model as evidenced by statistically significant reductions in tumor burden. Auransa is currently conducting IND-enabling studies of AU-409 and expects to initiate first-in-human Phase 1 trials in the first half of 2020.
Siranomics is evaluating preclinical candidate STP705, which consists of a combination of 2 silencer RNAs (siRNAs) silencing the TGF-β1 and COX-2 genes, delivered using a polypeptide nanoparticle delivery system based on Histidine-Lysine co-Polymer (HKP). Preclinical data demonstrated that STP705 inhibits the growth of HuCCt-1 (a human cholangiocarcinoma cell line) as a xenograft tumor in nude mice. Sirnaomics is working to develop siRNA therapeutics that augment the action of the immune checkpoint inhibitors to improve the clinical outcomes for patients with liver cancer (HCC).
SFA Therapeutics is developing SFA001, a microbiome-derived drug that is developed from natural substances using the Company’s proprietary discovery process to match metabolites and adjuvants to treat chronic inflammatory diseases. The FDA granted Orphan Drug Status in October of 2019 based on preclinical studies in transgenic HBX animal models in which SFA001 significantly reduced the number of HCC tumors as well as the size of the tumors. Additionally, the mechanism of action of SFA001 was found to be non-chemotoxic in 2 different animal models of HCC.
Outlook
With hepatocellular carcinoma on the rise both in the United States and globally, coupled with a high mortality rate, new prevention and treatment strategies are needed to reduce the incidence and burden of HCC. These strategies may include the development of vaccines, advanced screening techniques, different types of neoadjuvant therapies, newer ablation techniques, targeted therapies and the identification of specific biomarkers (proteins).
Vaccines
Personalized peptide vaccines (PPVs) are an immunotherapeutic approach based on the administration of multiple cancer peptides selected from a list of candidates to complement the pre-existing immunity of a patient. Personalized peptide vaccines against peptides that are recognized by patient T cells elicit immediate, tumor-specific immune responses. Furthermore, advances in genomics and bioinformatics have facilitated the generation of personalized peptide vaccines derived from neo-antigens, which have proven safe in clinical trials.
Results from a PPV feasibility study were published in September of 2017 in the journal Cancer Science. The peptides for the PPV were selected from 31 peptide candidates based on the pre‐existing immunity of 26 HCC patients refractory to locoregional therapies (cohort 1) and 30 patients refractory to both locoregional and systemic therapies (cohort 2). At the end of the first cycle of six vaccinations, successful CTL or IgG (immune) boosting was observed in 57% or 46% of patients in cohort 1 and in 54% or 52% of patients in cohort 2, respectively. Successful IgG boosting at the end of the second cycle was observed in the majority of patients tested.
The connection between hepatitis B infection and subsequent development of HCC underlies the need for global hepatitis B vaccination protocols to be put in place. According to an article published in the journal Frontiers in Biosciences, across geographic regions where the hepatitis B vaccine is common practice, a strong correlation has been observed between the numbers of people getting vaccinated and a decline in hepatocellular carcinoma rates. The article cited several studies demonstrating reductions in HCC and mortality rates following the institution of HBV vaccination.
There is no vaccine available against hepatitis C, which is also linked to subsequent hepatocellular carcinoma diagnosis. However, research is currently underway one treatment has shown promise in eliciting immune responses in mice.
Advanced screening techniques
Several new blood tests are being studied to see if they can detect liver cancer earlier than using AFP and ultrasound. Ones being studied include DCP, Glypican-3, osteopontin and Golgi protein-73. CT scans and MRI scans are also being studied as different imaging tests to screen for liver cancer as an alternative to ultrasound.
- Des-gamma-carboxy prothrombin (DCP) is an abnormal form of the coagulation protein, prothrombin. An elevated DCP level is associated with increased risk of recurring HCC. In patients with an elevated DCP result, the risk of developing HCC is 36.5% while the risk of developing HCC with a negative DCP result is 7.6%.
- Glypican-3 (GPC3) is under evaluation as a potential biomarker for both targeted therapy and diagnosis of HCC as it is over-expressed in HCC but not expressed or expressed at low levels in normal adult tissue. Preclinical studies have shown promise in utilizing GPC3 as a diagnostic molecule and a therapeutic target in HCC immunotherapy.
- Osteopontin (OPN) is a versatile protein that mediates various biological events involving the immune system and the vascular system. Studies of surgically resected HCC have demonstrated that osteopontin is highly expressed in tumor tissue and correlates with tumor grade, stage and recurrence. In addition, studies have suggested that high serum osteopontin concentrations correlate with poor prognosis, reduced liver function, a worse Child-Pugh score and worse disease-free and overall survival. These findings suggest that osteopontin may provide a novel therapeutic avenue in the treatment of malignancies selectively expressing osteopontin, such as HCC.
- Golgi protein-73 (GP73) has emerged as a serum biomarker for HCC. Many studies have found that the GP73 protein is associated with liver diseases, especially with liver cancer disease, and therefore, it is expected to become a new serological tumor marker for early diagnosis of liver cancer. An article published in October of 2018 in the journal Cell Proliferation reviews GP73 and summarizes these global studies addressing the relationship between GP73 and liver diseases in various settings.
Neoadjuvant therapies
Neoadjuvant therapy is the administration of therapeutic agents before a main treatment. Neoadjuvant therapy aims to reduce the size or extent of the cancer before using radical treatment intervention, thus both making procedures easier and more likely to succeed and reducing the consequences of a more extensive treatment technique, which would be required if the tumor were not reduced in size or extent. Different types of neoadjuvant therapies include targeted therapy, chemotherapy, ablation, embolization, and radiation therapy.
Results from a clinical study were published in July of 2019 in the Journal of Clinical Oncology. The study was designed to compare the survival outcomes of neoadjuvant three-dimensional conformal radiotherapy (RT) followed by hepatectomy with hepatectomy alone in patients with hepatocellular carcinoma (HCC) and portal vein tumor thrombus (PVTT). Results showed that for patients with resectable HCC and PVTT, neoadjuvant RT provided significantly better postoperative survival outcomes than surgery alone.
One type of emerging ablation technique is called irreversible electroporation. This is an ablation procedure that does not use heat or cold to destroy cancer cells. It uses high voltage to open the “pores” of the cell (like the pores of the skin) which causes the cancer cells to die. It may be very useful for cancers that are in difficult areas to treat, such as near blood vessels.
Targeted Therapies
As seen with recently approved drugs and drugs currently in the pipeline, targeted drug therapies are the focus of much ant-cancer drug development. Targeted cancer therapies are drugs or other substances that block the growth and spread of cancer by interfering with specific molecules that are involved in the growth, progression, and spread of cancer. Targeted cancer therapies are sometimes called “molecularly targeted drugs,” “molecularly targeted therapies” or “precision medicines”.
Targeted therapies differ from standard chemotherapy in several ways:
- Targeted therapies act on specific molecular targets that are associated with cancer, whereas most standard chemotherapies act on all rapidly dividing normal and cancerous cells.
- Targeted therapies are deliberately chosen or designed to interact with their target, whereas many standard chemotherapies were identified because they kill cells.
- Targeted therapies are often cytostatic (that is, they block tumor cell proliferation), whereas standard chemotherapy agents are cytotoxic (that is, they kill tumor cells).
The treatment landscape for liver cancer/hepatocellular carcinoma has shifted dramatically in the last decade. In the time since the first drug treatment, Nexavar, was approved in 2007, six more have followed. The next phase of drug treatment is likely to explore various drug combinations in order to give patients the ultimate chance for recovery, as well as further exploring checkpoint and cellular immunotherapies. The future of liver cancer treatment is also likely to focus on molecular pathways, some of which have been well described in the development and progression of HCC. These molecular pathways could theoretically be exploited to slow down, halt or reverse the progression of HCC. As drug development moves forward, personalized treatments for hepatocellular carcinoma, based on biomarkers, molecular pathways, immune response, etc. are poised to make up the therapeutic landscape of this disease.