News
The framework, first introduced by FDA Commissioner Marty Makary and Center for Biologics Evaluation and Research head Vinay Prasad in November, was criticized for lacking detailed guidance. Agency leaders elucidated on the pathway for personalized medicines on Monday.
FEATURED STORIES
The first gene therapies approved to treat sickle cell disease in December 2023 are struggling on the market. But there are glimpses of forward momentum as Vertex and Genetix Bio provide updates.
After last year’s ‘stampede’ for FGF21 assets, the focus for the metabolic dysfunction-associated steatohepatitis space has shifted toward differentiated approaches, such as THR-β agonists and combination treatments, that seek to mirror the commercial success of Madrigal’s Rezdiffra.
Maintaining America’s momentum demands that policymakers resist policies that undermine research and development incentives.
FROM OUR EDITORS
Read our takes on the biggest stories happening in the industry.
Following the FDA’s refusal to review Moderna’s investigational mRNA flu vaccine last week, Commissioner Marty Makary faced questions from the U.S. president about the agency’s handling of vaccines. It’s a clear signal that the tension long brewing at the drug regulator has now gone all the way to the top.
THE LATEST
GenAI, the youngest of the AI family, is evolving quickly. The global life sciences industry is attempting to evolve with it. However, as a keynote session at DIA’s MASC suggests, taking a pause to understand AI risks and to prepare to comply with the EU AI Act is what is needed.
The biotech Tuesday reported that 56% of patients taking paltusotine in a late-stage trial achieved the primary endpoint of IGF-1 levels for their rare hormonal disorder. An NDA submission is slated for later this year with a launch anticipated in 2025.
Following in the footsteps of Bristol Myers Squibb and Eli Lilly, AstraZeneca on Tuesday jumped into the radiopharmaceuticals space by acquiring Fusion Pharmaceuticals in a deal worth $2.4 billion.
To help support the launch of Lyfgenia, bluebird bio on Monday entered into a five-year term loan deal with Hercules Capital that will extend the biotech’s cash runway through the first quarter of 2026.
An appellate court sided with Regeneron versus Novartis on Monday, agreeing that anti-VEGF pre-filled syringes constitute a distinct market than those sold in vials. The case involves Regeneron’s Eylea and Novartis’ Lucentis eye treatments.
Orchard Therapeutics on Monday secured the FDA’s first approval for an autologous gene therapy to treat the rare metabolic disease metachromatic leukodystrophy in children.
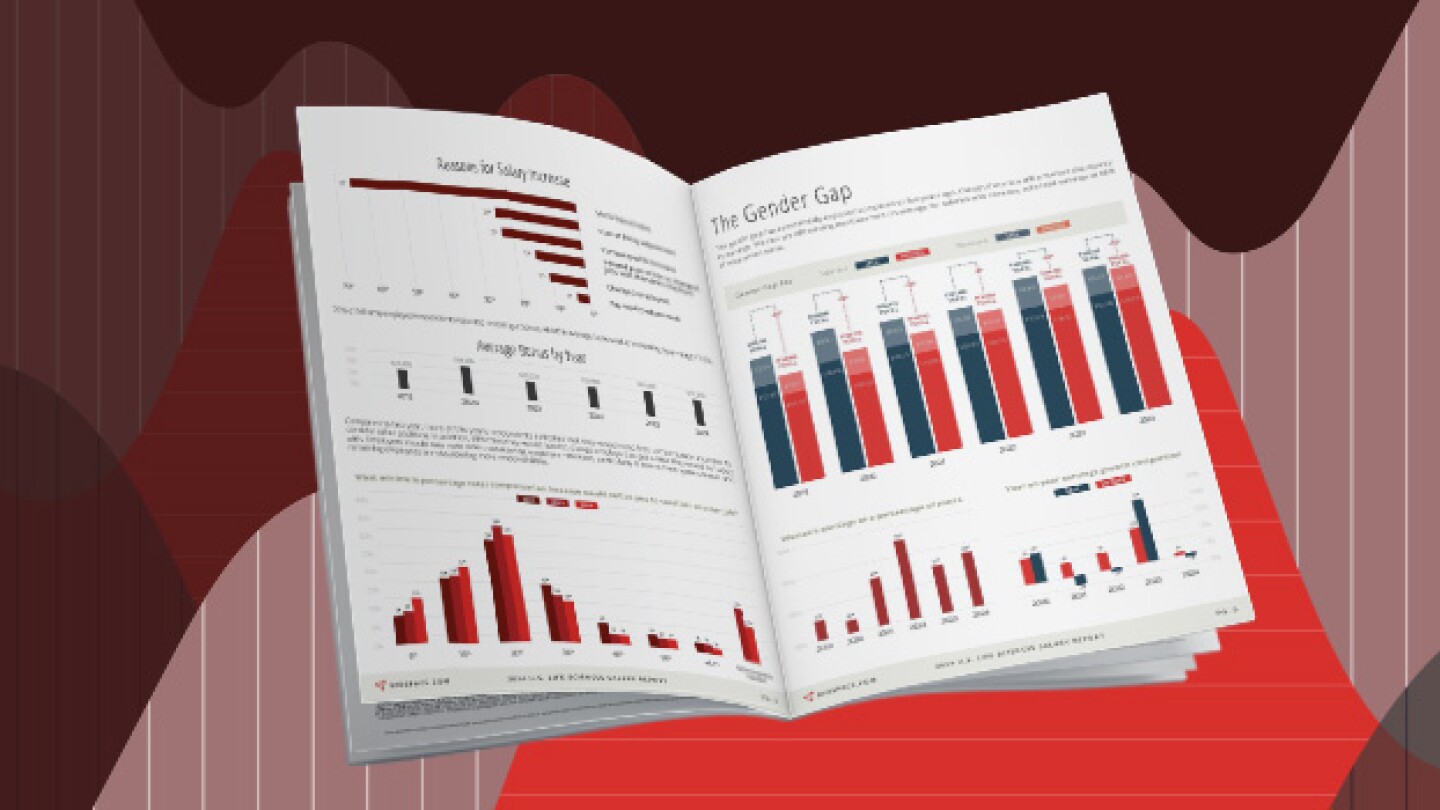
BioSpace’s 2024 Salary Report explores the average salaries and salary trends of life sciences professionals.
AstraZeneca reported Monday that adding Lynparza to Imfinzi improved outcomes in mismatch repair proficient endometrial cancer, more than doubling the median duration of response in patients.
With Boehringer Ingelheim’s announcement earlier this month that it was capping U.S. inhaler costs at $35 per month, AstraZeneca on Monday followed suit.
New late-stage trial results for GSK’s Jemperli show improved overall and progression-free survival in a broader range of endometrial cancer patients, which could lead to a potential label expansion.