News
LB Pharma needed $350 million to advance a promising schizophrenia candidate at a time when the biotech markets were locked up tight. Fortunately, it wasn’t CEO Heather Turner’s first rodeo.
FEATURED STORIES
Corsera Health’s Chief Operating Officer Rena Denoncourt and CFO Meredith Kaya speak with BioSpace about the biotech’s mission and vision for the next generation of cardiovascular care.
Billions of dollars’ worth of cancer drugs are discarded each year. Manufacturers must refund Medicare for some of this waste. A data-driven approach offers a practical path to greater efficiency.
Sales of Merck’s longtime oncology blockbuster Keytruda will erode more starkly in about 2033 rather than 2029, predicts Bloomberg Intelligence, translating to some $22 billion more in revenue.
FROM OUR EDITORS
Read our takes on the biggest stories happening in the industry.
Following the FDA’s refusal to review Moderna’s investigational mRNA flu vaccine last week, Commissioner Marty Makary faced questions from the U.S. president about the agency’s handling of vaccines. It’s a clear signal that the tension long brewing at the drug regulator has now gone all the way to the top.
THE LATEST
Orchard Therapeutics on Monday secured the FDA’s first approval for an autologous gene therapy to treat the rare metabolic disease metachromatic leukodystrophy in children.
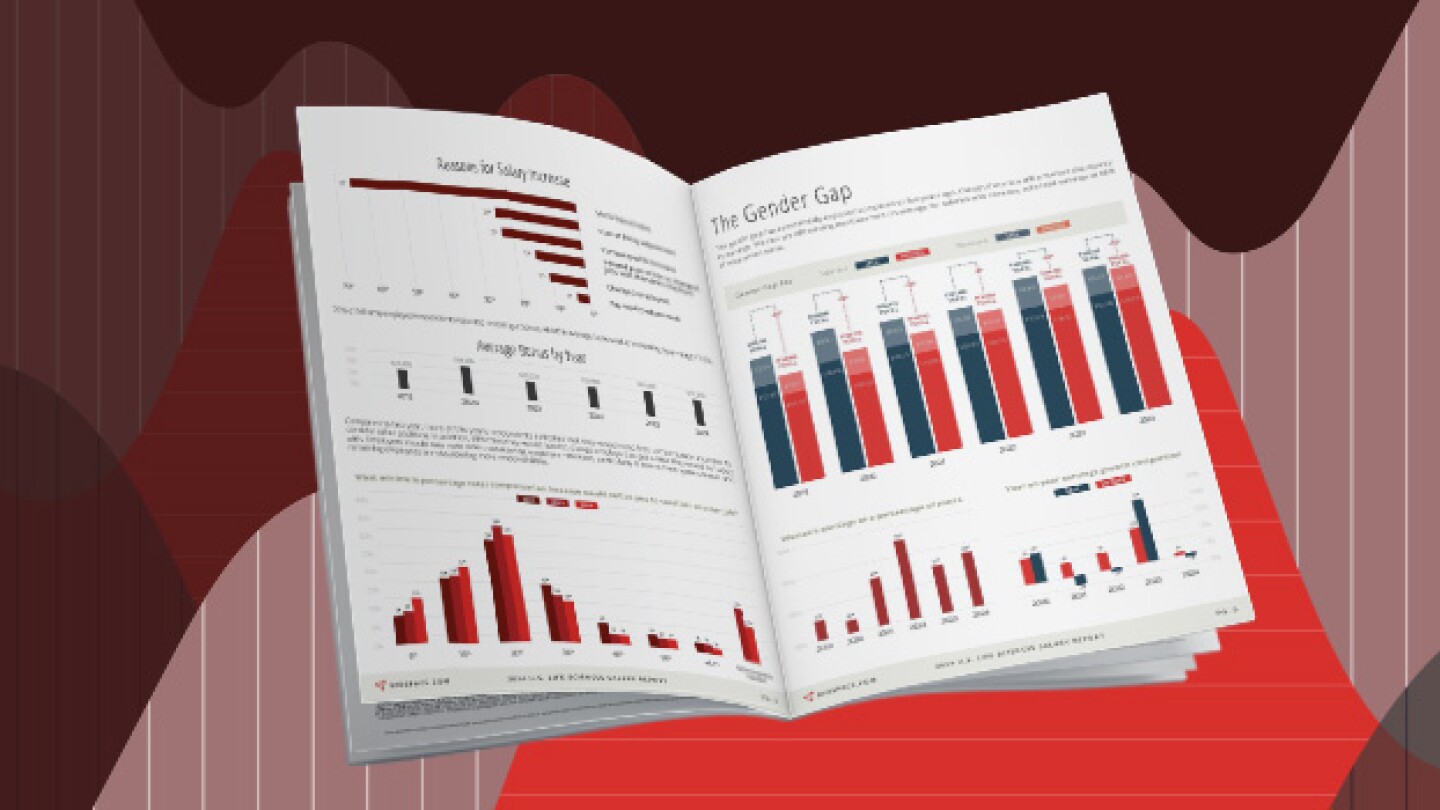
BioSpace’s 2024 Salary Report explores the average salaries and salary trends of life sciences professionals.
AstraZeneca reported Monday that adding Lynparza to Imfinzi improved outcomes in mismatch repair proficient endometrial cancer, more than doubling the median duration of response in patients.
With Boehringer Ingelheim’s announcement earlier this month that it was capping U.S. inhaler costs at $35 per month, AstraZeneca on Monday followed suit.
New late-stage trial results for GSK’s Jemperli show improved overall and progression-free survival in a broader range of endometrial cancer patients, which could lead to a potential label expansion.
Contineum Therapeutics joined the 2024 initial public offering class on Friday with an SEC filing. The biotech will use the IPO proceeds to complete a Phase II trial for its most mature candidate targeting multiple sclerosis.
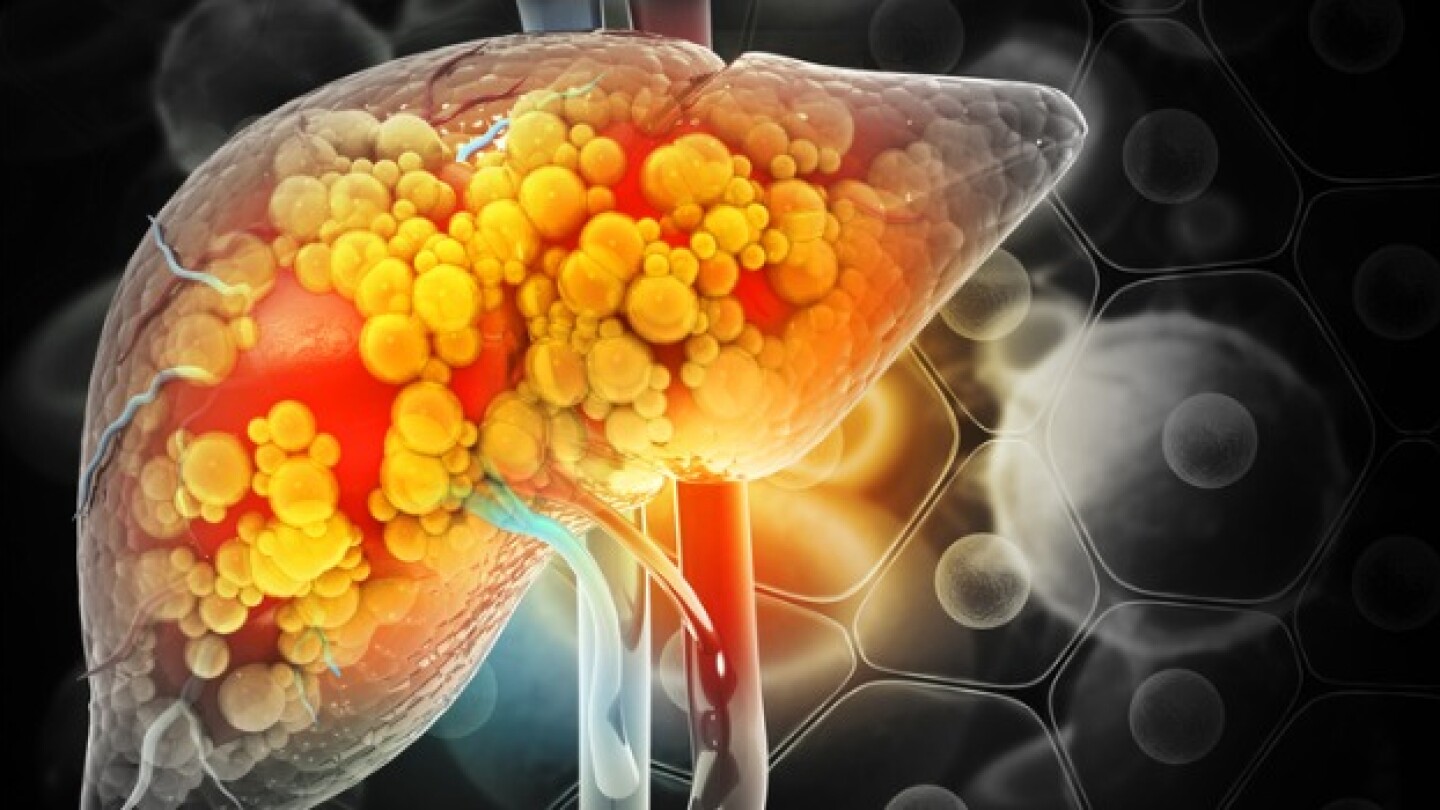
With its FDA approval last week and first-to-market advantage, Madrigal Pharmaceuticals’ Rezdiffra will set the standard for other metabolic dysfunction-associated steatohepatitis candidates in development.
By votes of 11-0 and 8-3, respectively, an FDA advisory committee Friday deemed the risks of early death for both Johnson & Johnson’s Carvykti and Bristol Myers Squibb’s Abecma acceptable.
Asgard Therapeutics, a Swedish gene therapy biotech, has closed a $32 million Series A round with help from prominent pharma players as it prepares for a 2026 IND.
Bayer will co-create a novel target identification platform that leverages Aignostics’ artificial intelligence technology and proprietary multimodal patient cohorts.