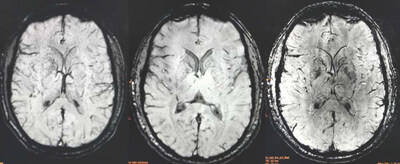
- Potentially Distinct Brain Profile: Across multiple brain regions, AL001 and lithium carbonate appeared to trend in opposite directions in brain chemistry measures, suggesting that AL001 may interact with the brain in a distinct manner and generate a lower neurochemical footprint than lithium carbonate
- Expected Trends for Myo-Inositol Reduction: Both AL001 and lithium carbonate showed a trend toward reducing myo-inositol, potentially supporting the hypothesis that AL001 retains lithium's core mechanism of action
- Potentially Preserved Glutamate Balance: Lithium carbonate showed large effects across all brain regions whereas AL001 showed minimal glutamate effect in most brain regions, which may suggest better long-term tolerability
ATLANTA, April 7, 2026 /PRNewswire/ -- Alzamend Neuro, Inc. (Nasdaq: ALZN) ("Alzamend"), a clinical-stage biopharmaceutical company focused on developing novel products for the treatment of Alzheimer's disease ("Alzheimer's"), bipolar disorder type 1 ("BD"), major depressive disorder ("MDD") and post-traumatic stress disorder ("PTSD"), today announced encouraging pharmacodynamic findings from a brain magnetic resonance spectroscopy ("MRS") analysis conducted in healthy human subjects (N=6) in a trial conducted at Massachusetts General Hospital. The study assessed changes in five key brain metabolites across 18 brain regions when participants received two-weeks of blood bioequivalent and lithium-dose equivalent AL001 or lithium carbonate relative to baseline. Early data suggest AL001 may work like lithium carbonate by selectively impacting brain chemicals where needed, however, AL001 appears to be leaving other, healthy brain chemicals more undisturbed than lithium carbonate, a potentially meaningful tolerability advantage. These interpretations are based solely on qualitative review of all analyses and need to be further statistically confirmed in additional patient populations, the first of which is currently underway.
Pharmacodynamic MRS Preliminary Findings
- Potential Distinct Neurochemical Footprint: When participants took AL001, multiple brain chemicals trended downward, while the same chemicals trended upward in those same participants when they took lithium carbonate. This suggests that AL001 may interact with the brain in a distinct manner and have a less disruptive effect on healthy brain tissue than lithium carbonate.
- Trends Towards Myo-Inositol Reduction: Both AL001 and lithium carbonate reduced levels of a key brain chemical called myo-inositol - which is exactly what lithium-based treatments are supposed to do. Notably, AL001 affected this target in nearly twice as many brain regions (17 out of 18) as lithium carbonate (8 out of 18), suggesting AL001 may deliver lithium's intended benefits more broadly throughout the brain.
- Potentially Preserved Glutamate Homeostasis: Glutamate, a key chemical messenger in the brain, was largely undisturbed in 10 of the 18 brain regions of patients after two-weeks of AL001, while two-weeks of lithium carbonate seemed to have caused disruptions to glutamate levels across every brain region measured. Keeping glutamate stable is important for long-term brain health, which suggests AL001 may have fewer side effects than lithium carbonate over time, however, this needs confirmation over a longer duration of exposure.
"Lithium carbonate has been a cornerstone of psychiatric treatment for over 55 years, but its harsh side effect profile has always limited how widely and how long it can be used," said Stephan Jackman, Chief Executive Officer of Alzamend. "These findings suggest AL001 may finally change that equation, delivering what lithium does best, without much of what makes it difficult to tolerate. That is a potential game changer for 43+ Million Americans living with BD, Alzheimer's, MDD and PTSD. We are seeking to confirm these findings in larger, adequately powered studies."
Hypotheses Generated for Future Confirmatory Studies
Based on these initial findings, Alzamend has identified the following pharmacodynamic hypotheses to be tested in future confirmatory studies involving subjects with Alzheimer's, BD, MDD and PTSD:
- AL001 causes less disruption to healthy brain tissue than lithium carbonate, potentially resulting in fewer side effects and better long-term tolerability;
- AL001 produces the same beneficial brain response that makes lithium an effective treatment, by reducing a key brain chemical, myo-inositol, suggesting it works through the same proven mechanism of action as lithium carbonate, just with a potentially better safety profile;
- AL001 appears to leave glutamate levels in healthy brain tissue largely undisturbed, a potentially important advantage over lithium carbonate, which appears to disrupt glutamate broadly. Stable glutamate levels in healthy tissue may mean fewer cognitive side effects and better long-term tolerability for patients; and
- Unlike lithium carbonate, AL001 may help preserve the health of brain cell membranes in healthy tissue, the protective outer layer of brain cells that play a critical role in how they function and communicate. If confirmed, this could mean AL001 is better tolerated by patients over the long-term than lithium carbonate.
AL001: A Differentiated Lithium Therapy for a Large Unmet Need
Although lithium has remained the gold standard treatment for bipolar disorder for more than 55 years, its clinical utility is constrained by a narrow therapeutic window and the need for regular therapeutic drug monitoring to manage renal, thyroid, and other systemic toxicity risks. AL001 is Alzamend's patented ionic cocrystal formulation of lithium combined for delivery with L-proline and salicylate, which is designed to deliver a full therapeutic amount of lithium to the brain with less systemic exposure than lithium carbonate, potentially enabling a safer, better-tolerated therapy across Alzheimer's, BD, MDD and PTSD.
About this Study
Brain metabolite concentrations were assessed using ultra-high field high-resolution magnetic resonance spectroscopic imaging (MRSI) in six healthy volunteers following blood-bioequivalent and lithium-dose equivalent 14-day multiple doses of AL001 and lithium carbonate treatments in a randomized, crossover design across 18 brain regions. The five metabolites analyzed were: total creatine (Cr+PCr), glutamate (Glu), glycerophosphocholine plus phosphocholine (GPC+PCh), myo-inositol (Ins), and N-acetylaspartate plus N-acetylaspartylglutamate (NAA+NAAG). Statistical analyses used the Wilcoxon signed-rank test and Hedges' g effect-size measure, with a pre-specified ≥20% absolute threshold to screen for pharmacodynamically relevant signals. The MRS neuroimaging methodology was developed by the lab of Dr. Ovidiu C. Andronesi, the study's principal investigator, Associate Professor of Radiology at Harvard University, and the Director of Multinuclear MR Imaging, Martinos Center for Biomedical Imaging, Department of Radiology, Massachusetts General Hospital, Harvard Medical School. All findings are exploratory, hypothesis-generating, and require confirmation in adequately powered studies.
About Alzamend Neuro
Alzamend is a clinical-stage biopharmaceutical company developing novel therapies for Alzheimer's, BD, MDD and PTSD. Our mission is to rapidly develop and market safe and effective treatments. Our current pipeline consists of two novel therapeutic drug candidates, AL001, a patented ionic cocrystal delivering lithium with salicylate and L-proline designed to improve brain delivery and safety compared to conventional lithium, and ALZN002, a patented cell-based therapeutic vaccine designed to restore the immune system's ability to clear Alzheimer's beta-amyloid. The latter is a next-generation active-immunity approach offering potential advantages in dosing frequency and cost compared to approved passive-immunity antibody therapies. Both candidates are exclusively licensed from the University of South Florida Research Foundation under royalty-bearing worldwide licenses.
Forward-Looking Statements
This press release contains "forward-looking statements" within the meaning of Section 27A of the Securities Act of 1933, as amended, and Section 21E of the Securities Exchange Act of 1934, as amended. These forward-looking statements generally include statements that are predictive in nature and depend upon or refer to future events or conditions, and include words such as "believes," "plans," "anticipates," "projects," "estimates," "expects," "intends," "strategy," "future," "opportunity," "may," "will," "should," "could," "potential," or similar expressions. Statements that are not historical facts are forward-looking statements. Forward-looking statements are based on current beliefs and assumptions that are subject to risks and uncertainties. Forward-looking statements speak only as of the date they are made, and Alzamend undertakes no obligation to update any of them publicly in light of new information or future events. Actual results could differ materially from those contained in any forward-looking statement as a result of various factors. More information, including potential risk factors, that could affect Alzamend's business and financial results are included in Alzamend's filings with the U.S. Securities and Exchange Commission. All filings are available at www.sec.gov and on Alzamend's website at www.Alzamend.com.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/alzamend-neuro-reports-encouraging-pharmacodynamic-data-from-phase-ii-clinical-trial-of-al001-lithium-in-brain-study-in-a-trial-conducted-at-massachusetts-general-hospital-302735711.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/alzamend-neuro-reports-encouraging-pharmacodynamic-data-from-phase-ii-clinical-trial-of-al001-lithium-in-brain-study-in-a-trial-conducted-at-massachusetts-general-hospital-302735711.html
SOURCE Alzamend Neuro, Inc.