News
In a letter to President Donald Trump, a group of biotech executives recommended former cancer regulator Richard Pazdur to lead the agency after the departure of Marty Makary.
FEATURED STORIES
European pharma companies splashed billions of dollars into the U.S. biopharma sector in a matter of days, but there are differing views on whether the activity represents the rise of a new buyer class or a quirk of timing.
Three pharma CEOs joined the $30 million compensation club in 2025 but Eli Lilly’s David Ricks exceeded his nearest peer by more than $4 million.
After years of suffering from a bear market and more than 14 months of geopolitical turmoil shaking the macroenvironment, biotech appears to be moving on.
FROM OUR EDITORS
Read our takes on the biggest stories happening in the industry.
The Department of Health and Human Services is spinning its wheels, unable to establish steady leadership at three major divisions—the CDC and the FDA’s two primary review units.
THE LATEST
A one-time treatment for Parkinson’s disease could be a ‘market changer,’ experts told BioSpace, adding that cell therapies could limit the adverse effects seen with current drugs.
Drugmakers, including the genetic testing company 23andMe, are using novel approaches to stimulate NK cells’ intrinsic ability to recognize and kill tumor cells.
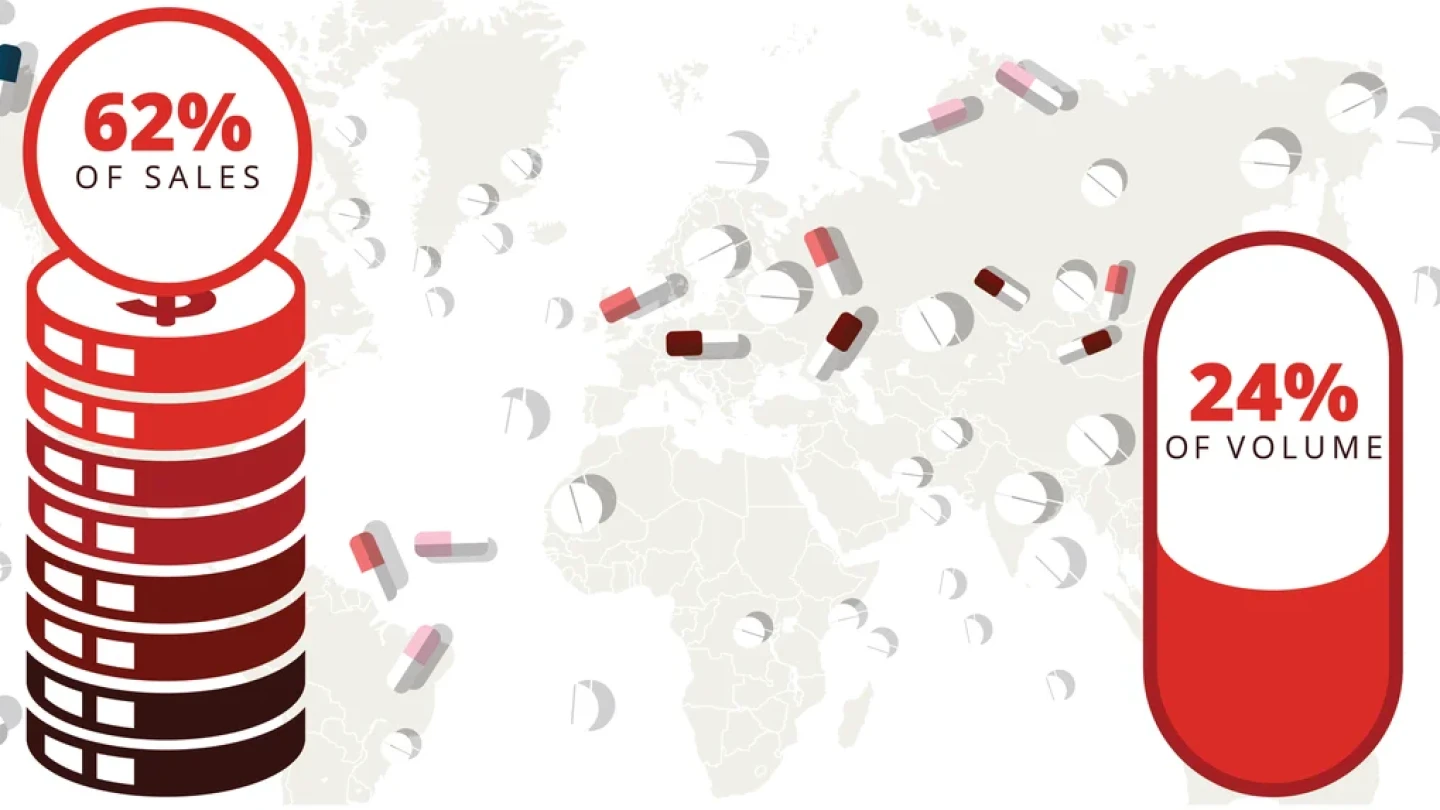
In this deep dive, BioSpace takes a closer look at the drug price crisis in the U.S. As President Joe Biden and former President Donald Trump gear up for a rematch in the 2024 election, we explore how federal reforms to lower costs could be leveraged on the campaign trail.
The clinical-stage biotech emerged from stealth on Thursday with several oral and injectable candidates, including a GLP-1 receptor agonist in Phase I trials, in an effort to challenge Novo Nordisk and Eli Lilly.
The FDA will close out April with five target action dates around indications that include pediatric seizures and a neurological cancer in children.
Data from the Phase III OCARINA II study shows the subcutaneous version of Ocrevus achieved near-complete suppression of relapses and brain lesions in relapsing or primary progressive multiple sclerosis.
Teva Pharmaceuticals and Alvotech got the FDA’s greenlight for their biosimilar to Johnson & Johnson’s blockbuster Stelara, which will launch in the U.S. in February 2025.
Nearly all doses of Eli Lilly’s highly popular weight-loss drug Zepbound and type 2 diabetes medication Mounjaro will have limited availability through the second quarter of 2024, according to the regulator.
Cerevel Therapeutics on Thursday reported positive data from its Phase III TEMPO-3 trial, showing that tavapadon can significantly improve symptom control in patients with Parkinson’s disease.
From gene-corrected cell therapies to a new CAR-T, the cell and gene therapy space looks to expand its reach into the market.