MAESTRO Phase 3clinical studies proceed, with more flexible allowances in visits and study drug dispensing for enrolled and screened patients at impacted sites, intended to maintain patient safety and trial integrity during the COVID pandemic.
- MAESTRO Phase 3clinical studies proceed, with more flexible allowances in visits and study drug dispensing for enrolled and screened patients at impacted sites, intended to maintain patient safety and trial integrity during the COVID pandemic.
- Abstracts accepted for oral presentations at postponed or cancelled scientific meetings (EASL 2020; AASLD, Emerging Topics, 2020 are described here. Key findings include:
- New analyses demonstrate that liver fat reduction at 3 months after starting treatment has clear predictive power for NASH resolution and fibrosis reduction on subsequent liver biopsy.
- Once daily oral 80 mg and 100 mg Phase 3 doses of resmetirom deliver at least 50% to more than 60% reductions in liver fat, respectively, and, based on new analyses of Phase 2 data, are associated with a statistically significant 64% NASH resolution (p<0.0001), of which >60% had fibrosis reduction.
- New data demonstrate that resmetirom robustly and statistically significantly (p<0.001) reduces markers of net collagen deposition in the liver supporting the anti-fibrotic action of resmetirom.
CONSHOHOCKEN, Pa., April 14, 2020 (GLOBE NEWSWIRE) -- Madrigal Pharmaceuticals, Inc. (NASDAQ:MDGL) today announced that in response to guidance from regulatory agencies, measures for COVID-19 at impacted sites have been put in place for its Phase 3 MAESTRO-NASH and MAESTRO-NAFLD-1 studies, allowing both studies to continue without changes to the protocol. Both MAESTRO studies are well-established and have been recruiting according to plan. At a recently conducted Data Monitoring Committee (DMC) meeting it was recommended that Phase 3 studies proceed without modification.
“In response to direction from regulatory agencies, a guidance to allow more flexible processes at sites impacted by COVID-19 was rapidly put in place to allow patients to progress through the screening process or continue their enrollment in the study,” stated Paul Friedman, M.D., Madrigal’s Chief Executive Officer. “Thus far, except for a slow down in new screening at impacted sites, the effect has been minimal on patients already in screening or enrolled in the studies.”
Dr. Friedman continued, “The recent DMC meeting gave the go ahead with no recommended changes to the protocols. We have sufficient drug supplies in hand to complete both studies without interruption and sufficient cash available to complete both the non-invasive 52 week MAESTRO-NAFLD-1 study and the 900 patient 52-week serial biopsy MAESTRO-NASH study that support the planned subpart H submission to FDA for accelerated approval.”
Becky Taub, M.D., Chief Medical Officer and President of Research & Development of Madrigal, stated, “As shown in our EASL abstract publication now rescheduled for oral presentation at the end of August, fat reduction as measured by week 12 MRI-PDFF predicts NASH resolution on biopsy at week 36. This finding, incorporating data from both placebo and resmetirom treated patients, was confirmed by two independent blinded central pathology readers. Higher fat reduction ( >50%) was correlated with a greater than 60% likelihood of NASH resolution with fibrosis reduction. Liver fat reductions at 80 mg and 100 mg, the Phase 3 doses, are impressive with relative: absolute fat reductions of 50%: 9.7% and 64%: 15.7%, respectively, as demonstrated in Phase 2. Available data, including our own, also indicate that liver fat reduction and NASH resolution are directly associated to a reduction in fibrosis.
Dr. Stephen Harrison, M.D., Principal Investigator of the study, and Medical Director for Pinnacle Clinical Research, San Antonio, Texas, and Visiting Professor of Hepatology, Oxford University, and Principal Investigator of the MAESTRO studies commented, “There is significant rationale for NASH patients to continue in clinical trials during the COVID-19 pandemic. With their high prevalence of diabetes and metabolic syndrome, NASH patients are at higher risk for developing life-threatening complications from COVID-19 infection. Controlling the liver disease and metabolic risk of such patients may help them survive COVID-19. Moreover, NASH patients in clinical trials can be well-managed for safety. Because resmetirom is in Phase 3, supported by substantial safety data and has shown benefit in NASH and parameters of metabolic syndrome, I believe MAESTRO trials are well-positioned to maintain enrolled patients in the studies, continue recruiting at unaffected sites and restart quickly at affected sites once the pandemic conditions are alleviated.”
He added, “I am particularly excited about the high predictive power of liver fat reduction on NASH resolution with fibrosis reduction demonstrated in resmetirom’s Phase 2 study. Moreover, PRO-C3 is a robust non-invasive biomarker demonstrated to be strongly correlated with NASH fibrosis. New analyses show that resmetirom statistically significantly reduces markers (PRO-C3/C3M) of net collagen deposition in the liver further supporting the potential anti-fibrotic action of resmetirom.”
EASL (European Association for the Study of the Liver) Abstract Publication - Magnetic resonance imaging-proton density fat fraction (MRI-PDFF) to predict treatment response on NASH liver biopsy: a secondary analysis of the resmetirom randomized placebo controlled Phase 2 clinical trial. Loomba, R, Bedossa, P, Guy, C, Taub, R, Bashir, M, Harrison, SA
Summary and findings:
Resmetirom treatment resulted in significant reduction in hepatic fat as assessed by MRI-PDFF after 12 and 36 weeks that was associated with higher rates of NASH resolution compared to placebo on week 36 liver biopsy assessment. The aim of this secondary analysis was to examine the potential of early reduction in MRI-PDFF to predict histologic response in patients with NASH including both placebo and resmetirom treated patients. The study cohort was assessed for those who had a ≥ 30, ≥ 40 and ≥ 50 % decline in MRI-PDFF between baseline and week 12 as predictors of NASH resolution.
To determine the potential predictive power of liver fat reduction on NASH resolution, data were analyzed from 107 NASH patients (n=34 placebo, n=73 resmetirom) with paired baseline/week 36 liver biopsies (read by two blinded central pathologists) and paired baseline/week 12 MRI-PDFFs. Including the MRI-PDFF/biopsy data from both MRI-PDFF responders and non-responders (including placebo patients) the relationship to a NASH resolution response was assessed.
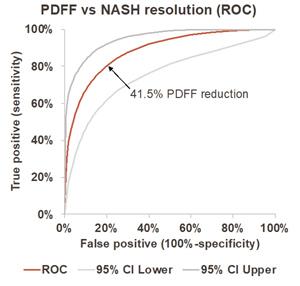
In patients with NASH resolution, the mean week 12 fat reduction (MRI-PDFF) was 56%. The predictive accuracy was determined by calculating the area under the receiver operating characteristic curve (AUROC) and its 95% confidence intervals (CI)s. The AUROC was 0.89, the optimal MRI-PDFF reduction with best balance of true positive and false negative rates was 41.5% (p<0.001) with a sensitivity of 82% (95% confidence interval (CI) 61%, 93%) and specificity of 83% (95%CI 0.74%, 90%) (Figure), and was similarly observed by both central readers. Compared to MRI-PDFF non-responders with <30% fat reduction (n=56), MRI-PDFF responders (≥30% fat reduction) (n=51) had significantly higher odds of NASH resolution (40% versus 3.7%) with odds ratio (OR) of 18.0, 95% CI (3.9-82.3), p <0.0001. Additionally, the percentages with NASH resolution were higher with greater reductions in liver fat content, ≥ 40% and ≥50% MRI-PDFF reduction showing an OR of 16.5, and 25.3, respectively, compared to PDFF non-responders, p<0.0001. In patients with ≥50% fat reduction at week 12 (placebo=2; resmetirom= 22) 64% had NASH resolution with a component response driven primarily by ballooning and inflammation reduction. The positive predictive values of PDFF reduction for NASH resolution at cutoffs of ≥30%, ≥40%, ≥50% were 40%, 50% and 67%, respectively, and the negative predictive values were >90% for all three cutoffs. Importantly, the analysis also confirmed the direct association of NASH resolution to fibrosis reduction, with fibrosis reduction in 61% of resmetirom treated patients with NASH resolution. Reductions in other biomarkers (e.g. ALT) were not strongly associated with NASH resolution or fibrosis reduction.
In conclusion, the early MRI-PDFF response in resmetirom and placebo treated patients achieved good sensitivity and specificity for the identification of patients with NASH resolution and corresponding fibrosis reduction. Compared with the Phase 2 dose of 60 mg (39% PDFF reduction), the higher doses of resmetirom (80 mg and 100 mg) being used in the ongoing Phase 3 MAESTRO-NASH study demonstrated an average PDFF reduction of 50% and 64%, respectively, that associate with high rates of NASH resolution and liver fibrosis reduction.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/d2656c0a-e309-4d39-aa52-7fc3ff9c5e94
American Association for the Study of Liver Diseases (AASLD) Emerging Topic Conference 2020---Nuclear Receptors in Nonalcoholic Fatty Liver Diseases (conference cancelled), Resmetirom, a beta selective thyroid hormone receptor agonist, reduces net collagen III deposition in nonalcoholic Steatohepatitis Harrison SA, Taub R, Barbone JM, Franc, J, Karsdal, MA
Summary and findings:
Type III collagen is a key component of liver fibrosis in NASH patients. N-terminal type III collagen pro-peptide (PRO-C3) are serum markers reflecting formation of type III collagen (pathologic collagen that is increased in the liver in advanced NASH fibrosis). Matrix metalloproteinase-degraded collagen III (C3M) degrades existing collagen III which is present in fibrotic livers. A reduction in PRO-C3 and/or elevation in C3M levels reflected by the PRO-C3/C3M ratio may be indicative of an overall decrease in liver fibrosis. We evaluated the effect of resmetirom treatment on PRO-C3, C3M and PRO-C3/C3M ratio in the Phase 2 study.
MGL-3196-05 (NCT02912260) was a 36-week multicenter, randomized, double-blind, placebo-controlled serial MRI-PDFF, paired liver biopsy Phase 2 study in adults with biopsy-confirmed NASH (NAS ≥4, F1-F3) and hepatic fat fraction ≥10%, assessed by MRI- PDFF (main study). Thirty-one patients (including 14 former placebo patients) received open-label MGL-3196 in the 36-week extension study.
Baseline PRO-C3 and PRO-C3/C3M ratio levels were significantly correlated with baseline fibrosis stage (Correlation coefficient (cc)= 0.24, p = 0.001) and ballooning (cc=0.29, p=0.003). PRO-C3 levels declined in patients treated with resmetirom relative to placebo (p < 0.0001) in the main study [Lancet 394, 2012-2024, 2019]. C3M levels increased slightly with resmetirom treatment, with no change in placebo. The PRO-C3/C3M ratio declined in resmetirom compared with placebo treatment in the main study (resmetirom, -0.24; placebo, 0.42, p<0.001) and significantly decreased in the extension study, in which all patients were treated with resmetirom (-0.50, p < 0.0001) (figure).
In conclusion, PRO-C3 levels and PRO-C3/C3M ratios were correlated to fibrosis stage and NAS components at baseline. Treatment of NASH patients with resmetirom produced significant reductions in PRO-C3 levels and PRO-C3/C3M ratio, which may reflect a net reduction in collagen deposition and fibrosis.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/c7bce372-1dda-4e08-8414-a0b6001f1cae
About Resmetirom (MGL-3196)
Thyroid hormone, through activation of its β-receptor in hepatocytes, plays a central role in liver function impacting a range of health parameters from levels of serum cholesterol and triglycerides to the pathological buildup of fat in the liver. Thyroid hormone receptor (THR)-β action in the liver is key to proper function of the liver, including regulation of mitochondrial activity such as breakdown of liver fat and control of the level of normal, healthy mitochondria. Patients with NASH have reduced levels of thyroid hormone activity in the liver with resultant impaired hepatic function, in part due to the inflamed state of the liver that causes degradation of thyroid hormone.
To exploit the thyroid hormone receptor (THR)-β pathway for therapeutic purposes in cardio-metabolic and liver diseases, it is important to avoid activity at the THR-α receptor, the predominant systemic receptor for thyroid hormone that is responsible for activity outside the liver including in heart and bone. The lack of selectivity of older thyromimetic compounds, chemically-related toxicities and undesirable distribution in the body led to safety concerns. Madrigal recognized that greater selectivity for thyroid hormone receptor (THR)-β and liver targeting might overcome these challenges and deliver the full therapeutic potential of THR-β agonism. Resmetirom has been shown to be highly selective based on 1) THR-β receptor functional selectivity based on both in vitro and in vivo assays 2) specific uptake into the liver, its site of action, virtually avoiding any uptake into tissues outside the liver. In short and long term human and animal studies, resmetirom has been confirmed to be safe and devoid of activity at the THR-α receptor and without impact on bone or cardiac parameters. Resmetirom does not impact the thyroid axis hormones, including the central thyroid axis. Madrigal believes that resmetirom is the first orally administered, small-molecule, liver-directed, truly β-selective THR agonist.
About the Phase 3 Registration Program for the Treatment of NASH (Non-alcoholic steatohepatitis)
The Phase 3 MAESTRO-NASH trial is expected to enroll 900 patients with biopsy-proven NASH (fibrosis stage 2 or 3), randomized 1:1:1 to receive resmetirom 80 mg once a day, 100 mg once a day, or placebo. After 52 weeks of treatment a second biopsy is performed. The primary surrogate endpoint on biopsy will be NASH resolution, with at least a 2-point reduction in NAS (NASH Activity Score), and with no worsening of fibrosis. Two key secondary endpoints are liver fibrosis improvement of at least one stage, with no worsening of NASH, and lowering of LDL-cholesterol [ClinicalTrials.gov/NCT03900429].
A second 52-week Phase 3 multi-center, double-blind, randomized, placebo-controlled study of resmetirom, MAESTRO-NAFLD-1, was initiated in December 2019 in 700 patients with non-alcoholic fatty liver disease (NAFLD), presumed NASH, randomized 1:1:1 to receive resmetirom 80 mg once a day, 100 mg once a day, or placebo. MAESTRO-NAFLD-1 also includes a 100 mg resmetirom open label arm in up to 100 patients. Unlike MAESTRO-NASH, MAESTRO-NAFLD-1 is a non-biopsy study and represents a “real-life” NASH study. NASH or presumed NASH is documented using historical liver biopsy or non-invasive techniques including fibroscan and MRI-PDFF. Using non-invasive measures, MAESTRO-NAFLD-1 is designed to provide incremental safety information to support the NASH indication as well as provide additional data regarding clinically relevant key secondary efficacy endpoints to better characterize the potential clinical benefits of resmetirom on cardiovascular and liver related endpoints. These key secondary endpoints include LDL-cholesterol, apolipoprotein B and triglyceride (TG) lowering; reduction of liver fat as determined by magnetic resonance imaging, proton density fat fraction (MRI-PDFF); and reduction of PRO-C3, a NASH fibrosis biomarker. [ClinicalTrials.gov/NCT04197479] Additional secondary and exploratory endpoints will be assessed including reduction in liver enzymes, fibroscan scores and other fibrosis and inflammatory biomarkers.
These and other data, including safety parameters, form the basis for potential subpart H submission to FDA for accelerated approval for the treatment of NASH. The original 900 patients in the MAESTRO-NASH study will continue on therapy after the initial 52-week treatment period; up to another 1,100 patients are to be added using the same randomization plan and the study is expected to continue for up to 54 months to accrue and measure clinical events, most relevantly progression to cirrhosis.
About Resmetirom’s Potential to Confer Cardiovascular Risk Reduction in NASH patients
Additionally, resmetirom lowers multiple atherogenic lipids, including LDL cholesterol, apolipoprotein B, triglycerides, lipoprotein (a) as demonstrated in Phase 2, a key differentiating factor compared with other NASH therapeutics. The magnitude of reduction of these lipids support a potential indication for treatment of hyperlipidemia in NASH patients and predicts a potential for benefit on cardiovascular (CV) events in NASH patients who die most frequently of CV, not liver disease.
Because of their diabetes, dyslipidemia, hypertension, obesity in concert with an inflamed, fatty liver, NASH patients, particularly those with advanced fibrosis, are at a substantially increased CV risk compared to the general population. Resmetirom’s ability to decrease liver fat, which is an independent risk factor for CV events, and resmetirom’s effect to reduce atherogenic lipids are being further evaluated in several key secondary endpoints in both MAESTRO Phase 3 clinical studies.
About Madrigal Pharmaceuticals
Madrigal Pharmaceuticals, Inc. (Nasdaq: MDGL) is a clinical-stage biopharmaceutical company pursuing novel therapeutics that target a specific thyroid hormone receptor pathway in the liver, which is a key regulatory mechanism common to a spectrum of cardio-metabolic and fatty liver diseases with high unmet medical need. Madrigal’s lead candidate, resmetirom, is a first-in- class, orally administered, small-molecule, liver-directed, thyroid hormone receptor (THR)-β selective agonist that is in currently in two Phase 3 clinical studies, MAESTRO-NASH and MAESTRO-NAGLD-1, designed to demonstrate multiple benefits across a broad spectrum of NASH (non-alcoholic steatohepatitis) and NAFLD (non-alcoholic fatty liver disease) patients. For more information, visit www.madrigalpharma.com.
Forward-Looking Statements
This communication contains “forward-looking statements” made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, that are based on our beliefs and assumptions and on information currently available to us, but are subject to factors beyond our control. Forward-looking statements include but are not limited to statements or references concerning: our clinical trials, research and development activities, and the timing and results associated with the future development of our lead product candidate, MGL-3196 (resmetirom); our primary and secondary study endpoints for resmetirom and the potential for achieving such endpoints and projections; optimal dosing levels for resmetirom; projections regarding potential future NASH resolution, safety, fibrosis treatment, cardiovascular effects, lipid treatment or biomarker effects with resmetirom; the predictive power of liver fat reduction on NASH resolution with fibrosis reduction; the achievement of enrollment objectives concerning patient number, safety database and/or timing for our studies; the risks attendant with conducting trials that are substantially larger than our past trials; potential NASH or NAFLD patient risk profile benefits with resmetirom; our possible or assumed future results of operations and expenses, business strategies and plans, capital needs and financing plans, trends, market sizing, competitive position, industry environment and potential growth opportunities, among other things. Forward-looking statements: reflect management’s current knowledge, assumptions, judgment and expectations regarding future performance or events; include all statements that are not historical facts; and can be identified by terms such as “anticipates,” “be,” “believes,” “continue,” “could,” “demonstrates,” ”design,” “estimates,” “expects,” “forecasts,” “future,” “goal,” “intends,” “may,” “might,” “plans,” “potential,” “predicts,” ”predictive,” “projects,” “seeks,” “should,” “will,” “would” or similar expressions and the negatives of those terms. Although management presently believes that the expectations reflected in such forward-looking statements are reasonable, it can give no assurance that such expectations will prove to be correct and you should be aware that actual results could differ materially from those contained in the forward-looking statements.
Forward-looking statements are subject to a number of risks and uncertainties including, but not limited to: our clinical development of resmetirom; enrollment uncertainties, generally and in relation to COVID-19 mandatory lock-down measures and individual precautionary measures that may be implemented for an uncertain period of time; outcomes or trends from competitive studies; the risks of achieving potential benefits in a study that includes substantially more patients than our prior study; the timing and outcomes of clinical studies of resmetirom; and the uncertainties inherent in clinical testing. Undue reliance should not be placed on forward- looking statements, which speak only as of the date they are made. Madrigal undertakes no obligation to update any forward-looking statements to reflect new information, events or circumstances after the date they are made, or to reflect the occurrence of unanticipated events. Please refer to Madrigal's filings with the U.S. Securities and Exchange Commission for more detailed information regarding these risks and uncertainties and other factors that may cause actual results to differ materially from those expressed or implied.We specifically discuss these risks and uncertainties in greater detail in the section entitled "Risk Factors" in Part I, Item 1A of our Annual Report on Form 10-K for the year ended December 31, 2019, as well as in our other filings with the SEC.
Investor Contact:
Marc Schneebaum, Madrigal Pharmaceuticals, Inc. IR@madrigalpharma.com
Media Contact:
Mike Beyer, Sam Brown Inc. mikebeyer@sambrown.com 312 961 2502