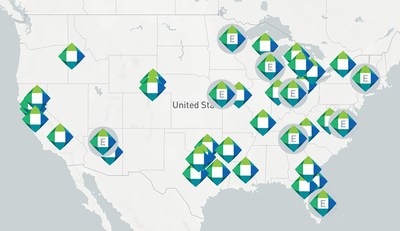
GT Medical Technologies Inc., creators of GammaTile Therapy, a Surgically Targeted Radiation Treatment for operable brain tumors, today announced its 2021 roster of ELITE Distinguished Brain Tumor Specialists that have completed 10 or more GammaTile Therapy procedures in 2021.
|
TEMPE, Ariz., Feb. 22, 2022 /PRNewswire/ -- GT Medical Technologies Inc., creators of GammaTile Therapy, a Surgically Targeted Radiation Treatment for operable brain tumors, today announced its 2021 roster of ELITE Distinguished Brain Tumor Specialists that have completed 10 or more GammaTile Therapy procedures in 2021. This year's introduction into the ELITE program includes Piedmont Atlanta Hospital, Vidant Medical Center, Emory Healthcare, and The University of Kansas Health System. These hospitals join the existing ELITE Distinguished Brain Tumor specialists awarded in 2020, including Memorial Sloan Kettering Cancer Center, M Health Fairview, HonorHealth Scottsdale Osborn Medical Center, NorthShore University HealthSystem, and Mayfield Brain & Spine. GammaTile has allowed selected [brain tumor] patients to achieve astonishing survival durations.
GammaTile ELITE institutions share GT Medical Technologies' purpose of improving the lives of patients with brain tumors. These medical centers are each committed to excellence by putting quality of life for their patients first and embracing evidence-based innovation. "GammaTile represents a major upgrade compared to standard radiotherapy options," said Roukoz Chamoun, MD, Neurosurgeon and Associate Professor, Department of Neurosurgery, The University of Kansas Health System in Kansas City, KS. "With GammaTile, radiation is delivered precisely where it is needed resulting in increased efficacy and minimal potential for side effects. In addition, because it is placed during surgery immediately after tumor resection, it eliminates unnecessary and potentially dangerous delay in radiation therapy." "GammaTile allows us to treat previously untreatable patients and has allowed selected patients to achieve astonishing survival durations. At Piedmont we love having this tool in our toolbox," said Adam Nowlan, MD, MPH, Medical Director, Radiation Oncology, Piedmont Atlanta Hospital in Georgia. GammaTile Therapy features a bioresorbable, conformable, 3D-collagen tile that is implanted in the last five minutes of brain tumor removal surgery. The sustained, controlled, and therapeutic dose of radiation immediately begins targeting tumor cells, sparing healthy tissue. Patients receive their radiation treatment while going about their daily life, without the necessity of daily trips to a medical center for external radiation. Over time, the tile naturally resorbs into the adjacent tissue with no need for additional surgery to remove it. GammaTile Therapy is shown to improve local tumor control, which can extend a patient's life. GammaTile was FDA-cleared in 2018 for recurrent brain tumors, including recurrent high-grade gliomas, glioblastomas, meningiomas, and brain metastases. In 2020, the FDA expanded that indication to include newly diagnosed malignant brain tumors. "We are pleased to see this technology become available to patients with brain tumors across the United States, with over 60 leading institutions now offering GammaTile," said Matthew Likens, President & CEO of GT Medical Technologies. "We take great pleasure in welcoming these prestigious institutions into the GammaTile ELITE program and commend them on their commitment to innovation and quality patient care." GT Medical Technologies was founded by five Arizona brain tumor specialists in 2017 to overcome the limitations of the current standard of care for patients with brain tumors. The treatment has since helped hundreds of patients receive life-altering cancer treatment in top brain tumor centers across the U.S. Healthcare providers can learn more by visiting https://www.gtmedtech.com/healthcare-providers. About GT Medical Technologies, Inc. Driven to overcome the limitations of current treatments for brain tumors and raise the standard of care, a team of brain tumor specialists joined forces and formed GT Medical Technologies with the purpose of improving the lives of patients with brain tumors. Its GammaTile Therapy received FDA 510(k) regulatory clearance for the treatment of all types of recurrent brain tumors and newly diagnosed malignant tumors. 3D, resorbable collagen tiles, embedded with radiation sources, are implanted during the last five minutes of brain tumor resection procedures, providing an immediate, dose-intense treatment to eliminate residual tumor cells. This "one and done" procedure allows patients to receive their course of radiation while going about their daily lives, requiring no additional trips to the hospital or clinic for radiation therapy. GammaTile is available in top brain tumor centers across the United States. For more information or to find a GammaTile center near you, visit https://www.gtmedtech.com/ and follow @GammaTile on Twitter and LinkedIn. Contact: Sheena Moore, smoore@gtmedtech.com
SOURCE GT Medical Technologies |