Modern radiation therapy is tightly targeted to dramatically reduce toxicity and side effects and often can be administered on an outpatient basis.
Telix MTR Therapy/Courtesy Telix Pharmaceuticals
After years of disinterest from pharmaceutical companies, radiation therapy is regaining a foothold in oncology. This isn’t the therapy your parents remember, though. Modern radiation therapy is tightly targeted to dramatically reduce toxicity and side effects and often can be administered on an outpatient basis.
“There’s a lot of innovation in this field,” Bernard Lambert, Ph.D., president and chief operating officer for Telix Pharmaceuticals (U.S.) told BioSpace. Telix’s own program is a case in point. The company has molecularly-targeted radiation (MTR) products under development or under review by the regulatory authorities for imaging and therapy. Telix has programs from preclinical to Phase III underway, as well as one imaging agent in final review at the U.S. Food and Drug Administration.
“MTR molecules are a concept of precision and personalized medicine,” Lambert said. “Rather than flooding the body with radiation and killing healthy cells in addition to tumors, we can selectively target the tumor (and tumor supportive cells) and deliver the radioisotope directly to it, saving healthy cells and, therefore, reduce the side effects.”
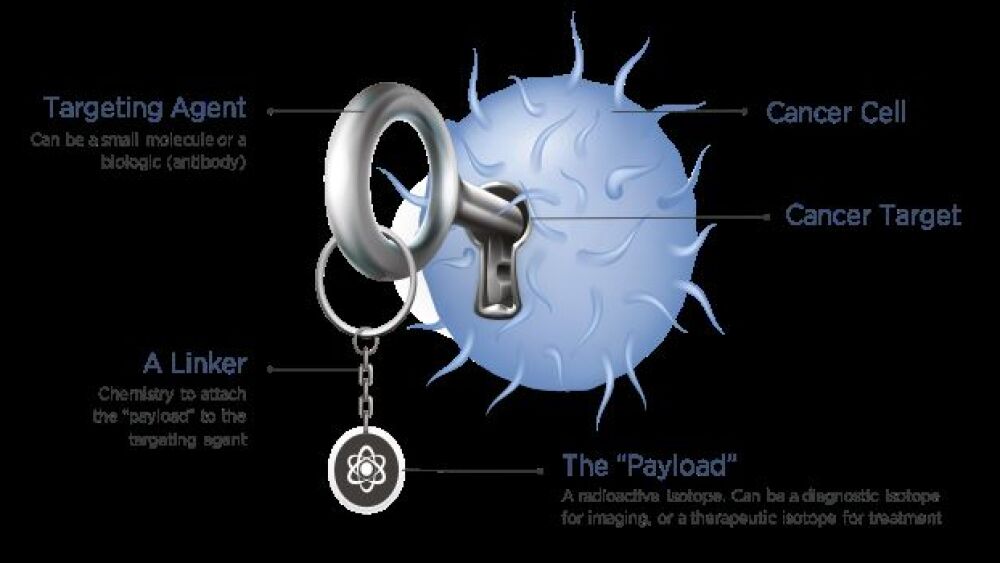
At a basic level, Telix attaches a radioactive isotope to a small molecule or antibody that targets the surface antigen on cancer cells. Once in the body, these molecules home-in on the cancer cell and deliver their radioactive payload, killing only the cancer cell. The decay rate varies from an hour or two to several days.
What really sets Telix apart from its competitors in this space, Lambert said, is that “we are covering a different stage of clinical development. Many (other companies) are in preclinical development. Telix, in contrast, is a pioneer in the industry. Our portfolio ranges from commercial to preclinical products in several indications, but mainly in oncology.” Notably, the same MTR platform can be used therapeutically to treat multiple indications by changing the delivery molecule. The platform also can deliver lower-energy radioisotopes for imaging, which can be used to determine treatment effectiveness or to stage cancer treatments.
Currently, the company is prioritizing prostate, kidney, brain and bone-marrow cancers, and just dosed its first patient in a Phase II triple-negative breast cancer trial studying TLX250-CDx (89Zr-DFO-girentuximab) in France. “That particular trial targets an antigen, carbonic anhydrase IX (CA9), that is overexpressed in patients with renal cell carcinoma,” he said, as well as lung and oesophageal cancers.
The breast cancer study, led by Caroline Rousseau at the Institut de Cancérologie de l’Ouest (ICO) in St. Herblain, France, evaluates how CA9 imaging and positron emission tomography can be used to diagnose triple-negative breast cancer (a particularly aggressive form of cancer with high unmet need) and to stage treatments. It is the second in a series of studies evaluating CA9 as a target for cancers beyond renal cell carcinoma.
Other programs targeting CA9 are in a Phase II therapeutic study for renal cell carcinoma and a Phase III imaging study for that indication. A separate study is underway in bladder cancer.
The side effects of MTR products vary depending on the molecule Telix uses, but Lambert said, “our safety profile is quite better than with chemotherapy, although like any of the drugs, it has some side effects, such as headaches, vomiting and pain at the injection point. They’re manageable.”
“Historically, radiation therapy was used at the end of the patient journey to extend the patient’s life when everything else had failed. Yet, research is showing it works well as a front-line treatment,” Lambert said. “It can’t be used for every tumor type, but it does address a lot of unmet needs and contribute to patients’ quality of life.” The challenge is convincing physicians that it can be used effectively as an early-stage treatment.
There’s another challenge, too: Delivering products with very short half-lives to patients throughout the U.S. and Europe. “We need a high level of coordination with our manufacturers in the U.S. and Europe,” Lambert pointed out. To manage the logistical hurdles, Telix is working with Cardinal Health and other radiopharmacy networks to ensure the products are near radiologic imaging centers. It is also establishing manufacturing sites in the U.S. and Europe.
In terms of upcoming milestones, Lambert said he is anticipating what he hopes will be FDA approval for TLX591-CDx, Telix’s prostate imaging product, and then submitting TLX250-CDx, its kidney cancer imaging product, for review. “We have a therapeutic program that’s active for prostate and kidney cancers, too,” he said. Telix also is planning to meet with the FDA to bring its glioblastoma product to Phase II/III trials that he anticipates will occur in 2022.