Biopharma has been stepping up research and drug development against COVID-19 -though results haven’t always been encouraging. Here are the latest stories from this space:
More than two years into the COVID-19 pandemic, it seems that the end is still not on our immediate horizons. The U.S. Centers for Disease Control and Prevention revealed that as of last week, the Omicron subvariants BA.4 and BA.5 accounted for around 20% of all coronavirus infections in the country, respectively.
Biopharma, in response, has been stepping up research and drug development against COVID-19 - though results haven’t always been encouraging. Here are the latest stories from this space.
Pfizer’s Paxlovid Disappoints in Standard-Risk Patients
Pharmaceutical giant Pfizer announced Tuesday that it is halting enrollment for a trial meant to assess the efficacy of its antiviral agent Paxlovid (nirmatrelvir tablets and ritonavir tablets) in standard-risk patients. The termination came after initial results showed that the drug induced a mere 51% reduction in the relative risk of developing coronavirus symptoms among people who were free of health conditions that could lead to severe COVID-19. This effect was not statistically significant, the company said.
Meanwhile, a recent analysis has found that after a five-day oral regimen of Paxlovid, only a handful of high-risk patients developed rebound symptoms. The drug’s overall benefit in this population was excellent, and all cases of rebound were mild and resolved quickly without additional treatment. More research is required to determine why some patients rebound after treatment.
Paxlovid was approved under emergency use authorization (EUA) in December 2021 for the treatment of patients 12 years and older with mild-to-moderate COVID-19 who are at high risk of progressing to severe disease, including both hospitalization and death.
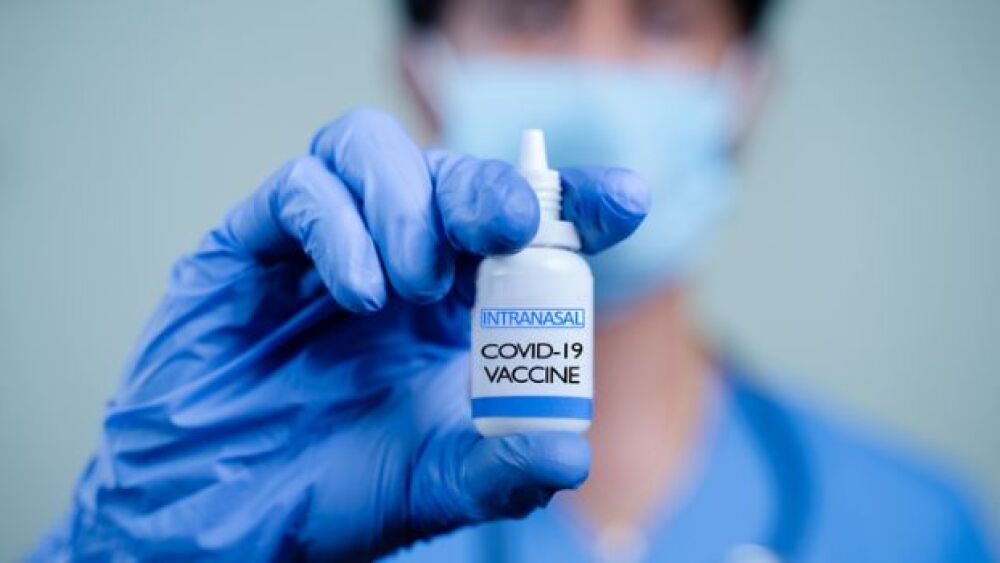
Oragenics Delivers Ray of Hope with Nasal Spray Animal Model Data
Infectious disease biotech company Oragenics announced encouraging results regarding NT-CoV2-1, its intranasal COVID-19 vaccine candidate.
The study, co-authored by Oragenics and published in Scientific Reports, a Nature journal, showed that the intranasal administration of NT-CoV2-1 in mice induced a strong antigen-specific IgG response in the blood, coupled with high titers of IgA in the lungs. In a parallel hamster experiment, the nasal spray vaccine performed impressively, leading to marked reductions in viral load.
In both the mice and hamster models, antibodies in the serum demonstrated robust neutralizing activity against the ancestral strain of COVID-19, as well as its Beta and Delta variants.
“We’re proud to have our innovative intranasal vaccine candidate studies published in a reputable, peer-reviewed scientific journal, and believe this heightened recognition underscores the need for vaccines that address the limitations of currently available options,” Frederick W. Telling, Ph.D., executive chairman of Oragenics, said in a statement. “These published data, and our ongoing GLP toxicology study will be part of our preclinical data package seeking permission to enter our first-in-human clinical trials.”
PureTech’s Deupirfenidone Safe but Falls Short of Efficacy Target
Yesterday, Boston-based clinical-stage biotherapeutics company PureTech Health reported that Phase II data confirmed the safety and tolerability of its therapeutic candidate LYT-100-COV (deupirfenidone). However, the drug showed no significant treatment effect in patients suffering from post-acute long COVID-19 respiratory symptoms.
The global, double-blind, randomized, placebo-controlled study enrolled 177 patients who were experiencing persistent COVID-19-related respiratory complications after clearing the initial acute infection. LYT-100-COV demonstrated an excellent safety profile, with nausea as its only potentially related adverse event.
The primary efficacy outcome, a 3-month change in the 6-minute walk test distance, improved in both treatment and placebo arms, but no between-group difference was observed. Based on these results, the company is planning to pivot deupirfenidone to idiopathic pulmonary fibrosis instead.
“Although we had hoped to offer a treatment to long COVID patients, these data further strengthen our confidence that LYT-100 has the potential to offer improved tolerability and therefore potentially improved treatment adherence and patient outcomes in IPF,” Daphne Zohar, founder and chief executive officer of PureTech, said in a statement. Registration-enabling studies for IPF are set to begin later this month.