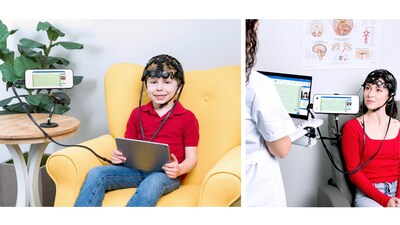
New product developed to expand access to EEG in outpatient clinics
RALEIGH, N.C., April 2, 2026 /PRNewswire/ -- Zeto, Inc., a medical technology company dedicated to making brain health insights through EEG a foundational vital sign in healthcare, announced that it has received U.S. Food and Drug Administration (FDA) 510(k) clearance for Zeto New Wave™, its newest EEG system for outpatient clinics and homes.
Patients across the U.S. often wait weeks for an EEG appointment, delaying critical answers and care. Clinics are also under pressure, facing shortages of specialized EEG technologists and relying on traditional setups that can be slow and cumbersome. Together, these challenges can significantly delay both patient testing and related follow-up care.
Zeto New Wave™ is designed to change that. It offers a complete, easy-to-use outpatient routine EEG solution, featuring a full head coverage with 21 soft-tip, no mess electrodes placed according to the widely used 10–20 system. Paired with a compact recording device with built-in display and controls, the system also captures synchronized video and audio, along with optional signals such as ECG, EOG, EMG, orientation sensors, external triggers for event related potentials (ERPs), and integrated photic stimulation detection.
Designed for short-term recordings of up to 2.5 hours, Zeto New Wave™ enables clinics to perform EEG efficiently and provide timely care to patients.
"New Wave™ is our newest outpatient EEG system, built on lessons learned from our first-generation device, the Zeto WR19," said Florian Strelzyk, PhD, CEO of Zeto. "We listened to neurologists' needs and designed a system that helps clinics expand access to EEG, address staffing challenges, and improve patient experience. In 2024, we released the ONE™ specifically for ICUs, EDs and for patient transport, and now with New Wave™ we custom-tailored an ideal solution for physician practices. Both systems run on our Zeto Cloud platform and enable remote access from anywhere. Now, providers can choose which system is best for their needs and budget."
The FDA clearance of the Zeto New Wave™ marks Zeto's third FDA-cleared device, alongside Zeto WR19 and Zeto ONE. Zeto offers EEG solutions for critical and emergency care, academic research, clinical trials, and outpatient clinics, and also provides AI-powered seizure detection, seizure burden analysis, and other quantitative analytics.
Zeto is committed to unlocking the potential of brain health assessment through accessible and actionable neurodiagnostic insights, empowering clinicians to improve routine EEG monitoring, and long-term patient outcomes.
About Zeto, Inc.
Zeto, Inc., a trailblazer in next-generation neurotechnology, is on a mission to make brain health insights as ubiquitous as any other vital sign. Powered by its FDA-cleared EEG platform, Zeto enables scalable, real-time measurement of brain activity and provides actionable data to support clinical decision-making - bringing brain monitoring into everyday care environments.
The company plans to leverage its platform technology to improve access and quality of medical EEG testing and to facilitate adjacent biomedical research and clinical trials. Zeto's EEG systems, which received FDA clearances in 2018, 2024, and 2026 are commercially available in the United States.
To learn more, visit https://zeto-inc.com/.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/zeto-announces-fda-510k-clearance-of-zeto-new-wave-advancing-efficiency-in-outpatient-eeg-302732590.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/zeto-announces-fda-510k-clearance-of-zeto-new-wave-advancing-efficiency-in-outpatient-eeg-302732590.html
SOURCE Zeto, Inc.