From conventional outsourcing to AI-driven strategic partnerships, the market is undergoing a profound digital revolution, accelerating drug development from years to months by pivoting from service vendors to strategic partners in personalized medicine. Powered by agentic AI, decentralized trials, and real-world evidence, these modern platforms transform traditional, siloed clinical studies into agile, patient-centric networks. By integrating multi-omics insights and automated GMP laboratory data, next-generation CROs are establishing a high-precision standard for accelerating advanced modalities in oncology, CNS, and rare diseases.
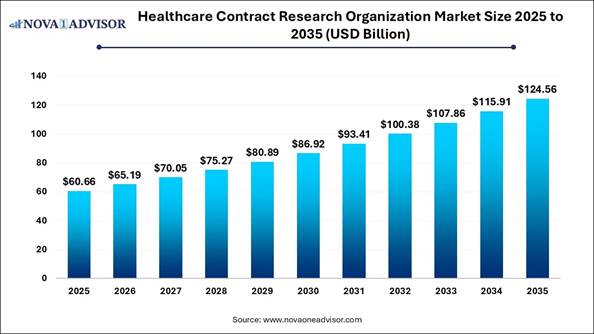
According to Nova One Advisor, the global healthcare contract research organization market is estimated to be valued at USD 60.66 billion in 2025 and is expected to reach USD 124.56 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 7.46% from 2026 to 2035.
Key Takeaways
⬥︎North America dominated with the largest market share in 2025.
⬥︎Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period.
⬥︎By type, the drug discovery segment captured the highest market share in 2025.
⬥︎By type, the clinical segment is poised to grow at a healthy CAGR between 2026 and 2035.
⬥︎By service, the project management and clinical supply management segment contributed the highest market share in 2025.
⬥︎By service, the regulatory and medical affairs segment is growing at a strong CAGR between 2026 and 2035.
⬥︎By therapeutic area, the oncology segment generated the biggest market share in 2025.
⬥︎By therapeutic area, the central nervous system disorders segment is expanding at the fastest CAGR between 2026 and 2035.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/9259
Market Overview: The New Age of Outsourced Research
A healthcare contract research organization acts as a strategic, indispensable partner to the pharmaceutical, biotechnology, and medical device industries, accelerating the journey from lab-bench discovery to patient bedside by managing complex outsourced research activities. Offering end-to-end solutions, CROs fuel innovation by handling everything from preclinical studies and comprehensive Phase I-IV clinical trial management to data analytics, biostatistics, and post-marketing surveillance, ensuring rigid regulatory compliance. Driven by the need for faster patient recruitment, heightened drug development complexity, and stringent regulatory requirements, focusing on core innovation.
Report Scope of Healthcare Contract Research Organization Market
|
Report Coverage |
Details |
|
Market Size in 2026 |
USD 65.19 Billion |
|
Market Size by 2035 |
USD 124.56 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 7.46% |
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
Segments Covered |
Type, Service, Therapeutic Area, Region |
|
Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
|
Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
|
Key Companies Profiled |
CTI Clinical Trial & Consulting, PSI, Medpace, Ergomed, WuXi AppTec, Worldwide Clinical Trials, Medidata Solutions, Inc,Pharmaron GMBH, SGS SA, KCR S.A., Advanced Clinical Research Services, LLC, Pharm-Olam, LLC (Allucent), ICON Plc, Charles River Laboratories, Syneos Health, IQVIA Inc., GVK Biosciences Private Limited (Aragen), LabCorp, Parexel International Corporation, Thermo Fisher Scientific |
For more information, visit the Nova One Advisor website or email the
team at sales@novaoneadvisor.com |
Call us: +1 804 420 9370 AI-Driven Clinical Trial Platforms:
Major Potential CROs are projected to adopt AI-native
platforms, transforming clinical
trials into predictive partnerships that reduce timelines and
operational costs through automation, data validation, and decentralized,
remote monitoring. This rapid shift offers a critical competitive advantage for
early adopters, enabling faster recruitment and higher-quality data for
sponsors, as AI moves from innovation to operational necessity. Regulatory Fragmentation and Data
Privacy Compliance: Major Limitation Navigating a chaotic patchwork of diverse
regional regulations and stringent data privacy laws like GDPR and HIPAA has
become the ultimate bottleneck for global clinical trials, crippling speed and
inflating costs. As CROs face intense cybersecurity threats and massive
compliance strains, this regulatory fragmentation introduces high-stakes risks,
including severe financial penalties, significant trial delays, and critical
reputational damage. Healthcare Contract Research
Organization (CRO) Market: Regional Analysis North America dominated the market in 2025.
This is mainly due to high research and development spending, a high
concentration of biopharmaceutical
firms, advanced healthcare infrastructure, and stringent FDA regulations that
necessitate specialized outsourcing partners. Substantial financial investments
from pharmaceutical, biotechnology companies, and government entities drive
rapid drug development, necessitating external expertise. Complex,
high-standard regulatory requirements from the FDA and Health Canada demand
expert oversight, driving reliance on specialized CROs to ensure compliance. The U.S. drives the industry through a
powerful trifecta of unrivaled infrastructure, innovative focus on oncology
and gene therapies, and regulatory leadership via FDA-supported adaptive
trials. Fueling this dominance is an aggressive shift towards AI-driven,
tech-enabled solutions that revolutionize patient recruitment and shorten study
timelines, solidifying the U.S. as the primary hub for complex, high-value
clinical development. 🔹In December 2025, the FDA will deploy voluntary agentic AI tools for
all employees, enabling automated, multi-step workflows to accelerate
regulatory reviews, inspections, and scientific tasks. Building on the adoption
of their Elsa LLM, this initiative includes a staff competition to drive
innovation in complex, AI-driven workflows. Canada is elevating its role in the global
market by offering lower costs, faster Health Canada approvals for early-phase
studies, and specialized expertise in oncology, neuroscience, and rare
diseases. Positioned as a premier, cost-effective, and innovative partner. Supported
by robust academic partnerships and high-tech biotech hubs, Canada acts as a
crucial niche research partner, bridging the gap between innovative therapies
and global commercialization. Asia Pacific is anticipated to have the
fastest growth during the forecast period. This growth is driven by high-volume
patient access, favorable regulatory shifts, and significant cost advantages.
Countries such as China and India have streamlined regulatory pathways,
reducing approval timelines significantly. China's National Medical Products
Administration reforms have halved review times, accelerating clinical trial
initiation. The region is rapidly developing world-class clinical research
centers, with high adoption of Electronic
Health Records in tertiary hospitals in South Korea and China, ensuring
high-quality, digitalized data. India has solidified its position as a
premier, cost-effective global hub for high-quality preclinical services and
complex, multi-center clinical trials. Leveraging a massive treatment-naïve
patient pool, diverse therapeutic expertise, and a skilled English-speaking
workforce, the market is rapidly expanding, with a strategic focus on comprehensive,
tech-driven research, accelerating drug development, and monitoring globally. 🔹In February 2026, the ₹10,000 crore Biopharma SHAKTI initiative aims
to transform India into a global powerhouse for biologics and biosimilars.
Proposed in the Union Budget for 2026–27, this strategy focuses on boosting
domestic manufacturing, upgrading the NIPERs, and establishing robust networks
for clinical trials. China has firmly established itself as the
leader in the market, shifting from a cost-driven landscape to a
high-capability hub driven by significant domestic innovation and substantial
government support. The country specializes in oncology, rare diseases, and
advanced cell and gene therapies. China's CRO sector is rapidly scaling up,
benefiting from improved infrastructure for early-phase trials and widespread
integration of artificial
intelligence (AI) in clinical data management. Healthcare Contract Research
Organization Market By Regional, 2025-2035 (USD Billion) Year 2025 2026 2027 2028 2029 2030 2031 2032 2033 2034 2035 North America 24.72 26.31 28.01 29.81 31.73 33.77 35.93 38.24 40.68 43.28 46.04 Europe 15.72 16.77 17.89 19.08 20.35 21.7 23.15 24.68 26.32 28.06 29.92 Asia Pacific 12.35 13.63 15.04 16.57 18.26 20.1 22.11 24.31 26.72 29.35 32.22 Latin America 1.68 1.81 1.94 2.09 2.24 2.41 2.59 2.78 2.99 3.21 3.45 Middle East and
Africa (MEA) 1.68 1.81 1.94 2.09 2.24 2.41 2.59 2.78 2.99 3.21 3.45
Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/9259
Healthcare Contract Research
Organization Market: Segmental Analysis By Type Analysis The drug
discovery segment dominated the market in 2025. This can be attributed
to the growth of early-stage research outsourcing, fueled by increasing
research and development investments, advanced AI integration, and the urgent
need for new therapeutics targeting chronic diseases. There is a growing demand
for novel treatments for rare and chronic diseases, which require specialized
expertise that CROs provide through in vitro and in vivo studies. By
outsourcing preclinical activities to CROs, companies can manage the high costs
and risks associated with early-stage drug development. The clinical segment is expected to
experience the fastest growth during the forecast period. This growth is
primarily driven by the increasing demand for outsourced Phase I-IV trials,
especially as complexities in oncology and chronic disease studies rise and the
urgency for accelerated drug development increases. CROs are leveraging AI and
advanced data analytics to enhance patient recruitment, data management, and
trial oversight, effectively reducing trial timelines. The rising prevalence of
chronic illnesses necessitates more clinical studies, further expanding the
demand for specialized clinical research services. Healthcare Contract Research
Organization Market by Type, 2025-2035 (USD Billion) Year 2025 2026 2027 2028 2029 2030 2031 2032 2033 2034 2035 Drug Discovery 7.86 8.39 8.94 9.54 10.18 10.85 11.57 12.34 13.16 14.03 14.96 Pre-Clinical 14.6 15.62 16.72 17.9 19.15 20.5 21.94 23.48 25.12 26.89 28.77 Clinical 33.69 36.32 39.16 42.2 45.49 49.04 52.86 56.97 61.42 66.19 71.35
By Service Analysis The project
management and clinical supply management led the market in 2025. This
is due to the need for efficient, compliant, and timely execution of complex,
large-scale clinical trials. Effective Clinical Trial Supply Management is
crucial, as it ensures the reliable procurement, production, and distribution
of investigational products, which directly impacts trial timelines and success
rates. As drug development costs continue to rise, pharmaceutical firms
increasingly outsource these resource-intensive services to CROs for cost
efficiency, technology integration, and faster trial execution to evolving
global and local regulations. The regulatory and medical affairs segment
is projected to grow the fastest during the forecast period. This growth is
driven by increasing global regulatory complexity and stringent compliance
requirements for new drug approvals, necessitating specialized expertise to
navigate approvals across different regions. Governments are imposing stricter
regulations on clinical trials, quality standards, and product safety, making
specialized regulatory support essential for obtaining approvals. The rise of
complex, personalized
medicines and gene therapies also demands intense regulatory oversight
throughout the development lifecycle. By Therapeutic Area Analysis The oncology segment dominated the market
in 2025, largely due to the high global incidence of cancer, which requires
extensive research and development efforts. This segment saw a surge in
complex, targeted therapy trials, with oncology trials constituting over
one-third of ongoing clinical trials worldwide. The substantial investment in
immunotherapies, targeted treatments, and combination therapies underscores the
need for CRO expertise in navigating the complexities of cancer trials. The central nervous system disorders
segment is expected to grow the fastest during the forecast period. This growth
is primarily connected to accelerating research and development in
neurodegenerative diseases and mental health. CNS trials are particularly
complex, requiring specialized trial designs, advanced imaging techniques,
biomarker analysis, and long-term monitoring, areas where CROs provide
essential expertise for monitoring and remote cognitive assessments. Key Emerging Innovations in the Healthcare
Contract Research Organization (CRO) Innovation Description Impact on CRO Market AI-Native Clinical Trials Deployment of AI/ML for protocol optimization, predictive patient
eligibility, and automated data cleaning. Reduces study startup times and speeds up patient recruitment;
reduces data cleaning cycles. Hybrid and Decentralized Trials (DCTs) Use of wearables, remote monitoring, and telemedicine to allow
patient participation outside traditional sites. Improves patient retention/diversity; lowers operational costs;
supports patient-centric study designs. Generative AI Copilots GenAI tools for automated generation of clinical study reports
(CSRs), protocols, and regulatory submissions. Accelerates document generation and reduces manual errors, shortening
submission timelines. Real-World Evidence (RWE) Integration Utilizing data from EHRs, claims, and wearables to support
regulatory filings and post-market surveillance. Enhances trial feasibility studies and provides insights into drug
efficacy outside controlled settings. Unified Digital Platforms Shifting from fragmented eClinical solutions to unified,
data-driven platforms for real-time monitoring. Increases operational transparency, improves data integrity, and
enables real-time risk-based monitoring.
Healthcare Contract Research
Organization (CRO) Market Companies ·
CTI Clinical Trial and Consulting ·
PSI ·
Medpace ·
Ergomed ·
WuXi AppTec ·
Worldwide Clinical Trials ·
Medidata Solutions, Inc. Major Shifts in the Healthcare Contract
Research Organization (CRO) Market ·
In January 2026, Profusa, Inc. launched the
Healthcare Research version of its Lumee tissue oxygen monitoring system for
the CRO market, aiming to generate service-based revenue. This product targets
a high-growth segment of the pharmaceutical services industry, responding to
increasing demand for real-time biological data to enhance drug development
efficiency for a variety of research applications, supporting studies in
pharmacology, toxicology, and oncology. ·
In July 2025, Auriga Research partnered with the
San Francisco Research Institute to enhance global healthcare through AI-driven
clinical research and expedited regulatory compliance, focusing on wellness
products across India, North America, and Africa. ·
In February 2026, IQVIA Holdings Inc. announced
its acquisition of discovery services assets from Charles River Laboratories,
which include in vitro drug discovery services and a small molecule AI
platform. This integration aims to strengthen IQVIA’s drug discovery capabilities
and support a wide range of therapeutic areas, enhancing clients' research and
development lifecycle support. Related Report 🔹 Collagen Dressings Market - https://www.novaoneadvisor.com/report/collagen-dressings-market
🔹 U.S. Antimicrobial Catheters Market- https://www.novaoneadvisor.com/report/us-antimicrobial-catheters-market
🔹 Tangential Flow Filtration Market- https://www.novaoneadvisor.com/report/tangential-flow-filtration-market 🔹 Contract Research Organization (CRO) Market- https://www.novaoneadvisor.com/report/contract-research-organization-cro-market
🔹 U.S. Microprocessor Market- https://www.novaoneadvisor.com/report/us-microprocessor-market
🔹 Effective Microorganisms Market - https://www.novaoneadvisor.com/report/effective-microorganisms-market
🔹 Automated Microscopy Market- https://www.novaoneadvisor.com/report/automated-microscopy-market
🔹 Legionella Testing Market- https://www.novaoneadvisor.com/report/legionella-testing-market
🔹 Clinical Microbiology Market - https://www.novaoneadvisor.com/report/clinical-microbiology-market 🔹 Pharmaceutical CRO Market- https://www.novaoneadvisor.com/report/pharmaceutical-cro-market
🔹 Micro Lending Market- https://www.novaoneadvisor.com/report/micro-lending-market
🔹 In Vivo CRO Market- https://www.novaoneadvisor.com/report/in-vivo-cro-market
🔹 Biopharmaceutical CMO And CRO Market- https://www.novaoneadvisor.com/report/biopharmaceutical-cmo-and-cro-market
🔹 Automated Microbiology Market- https://www.novaoneadvisor.com/report/automated-microbiology-market
🔹 Antibiotics Market- https://www.novaoneadvisor.com/report/antibiotics-market
🔹 U.S. Preclinical CRO Market- https://www.novaoneadvisor.com/report/us-preclinical-cro-market
Segments Covered in the Report This report forecasts revenue growth at
country levels and provides an analysis of the latest industry trends in each
of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc.
has segmented the healthcare contract research organization market. By Type By Service By Therapeutic Area By Regional Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/9259 About-Us Nova One Advisor is a global leader
in market intelligence and strategic consulting, committed to delivering deep,
data-driven insights that power innovation and transformation across
industries. With a sharp focus on the evolving landscape of life sciences, we
specialize in navigating the complexities of cell and gene therapy, drug
development, and the oncology market, enabling our clients to lead in some of
the most revolutionary and high-impact areas of healthcare. Our expertise spans the entire biotech
and pharmaceutical value chain, empowering startups, global enterprises,
investors, and research institutions that are pioneering the next generation of
therapies in regenerative medicine, oncology, and precision medicine. Web: https://www.novaoneadvisor.com/ Our Trusted Data Partners: Nova One Advisor - Market
Research Reports & Consulting Firm Nova One Advisor offers
comprehensive market research reports with in-depth industry analysis and
market data. Call us: +1 804 420 9370 Email: sales@novaoneadvisor.com Web: https://www.novaoneadvisor.com/ You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344


