News
Just a few days after FDA Commissioner Makary resigned, ally Tracy Beth Høeg is also leaving the agency. Her departure comes amid reports of tension over a commissioner’s voucher for Sanofi’s diabetes drug.
FEATURED STORIES
European pharma companies splashed billions of dollars into the U.S. biopharma sector in a matter of days, but there are differing views on whether the activity represents the rise of a new buyer class or a quirk of timing.
Three pharma CEOs joined the $30 million compensation club in 2025 but Eli Lilly’s David Ricks exceeded his nearest peer by more than $4 million.
After years of suffering from a bear market and more than 14 months of geopolitical turmoil shaking the macroenvironment, biotech appears to be moving on.
FROM OUR EDITORS
Read our takes on the biggest stories happening in the industry.
The Department of Health and Human Services is spinning its wheels, unable to establish steady leadership at three major divisions—the CDC and the FDA’s two primary review units.
THE LATEST
Research on Alzheimer’s, specifically in neuroscience, continues with the Alzheimer’s Drug Discovery Foundation, Critical Path Institute, Abbvie, AlzeCure Pharma and more.
The FDA approved bluebird bio’s lentiviral vector (LVV) eli-cel Friday as the first therapy to slow the progression of neurologic dysfunction in juvenile boys with early, active CALD.
Both Bristol Myers Squibb and AbbVie filed a WARN notice with the California state government stating they intend to lay off up to 360 people.
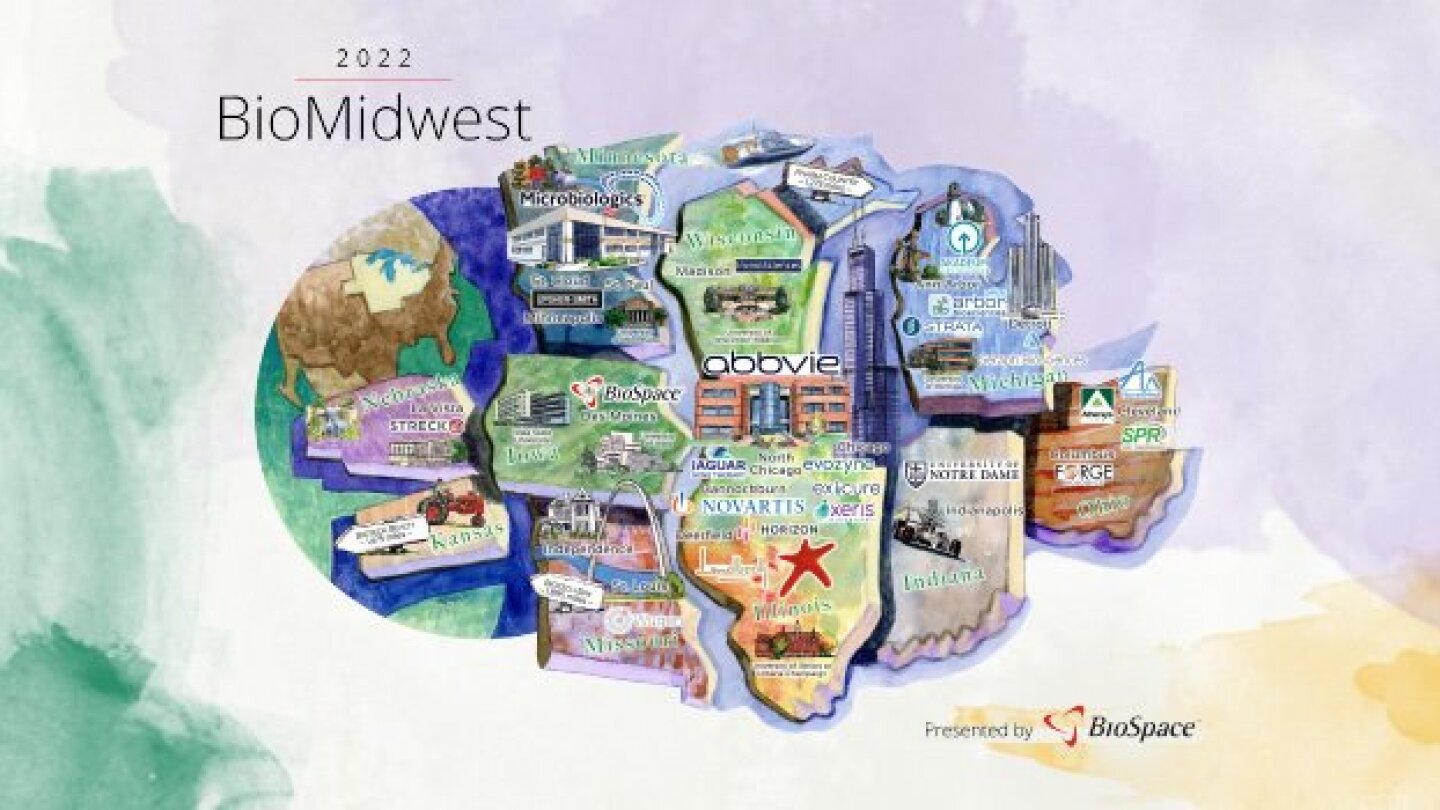
As a leading biotech hub, the second city still ranks far below many other U.S. locations. That is starting to change, however, as new companies move into the heart of the BioMidwest region.
With 33 approvals from the FDA, Merck’s checkpoint inhibitor Keytruda has become a powerhouse oncology drug and is likely to continue to rack up more regulatory wins.
The FDA has a broad range of activities in the drug and medical device space. Here’s a look at the agency’s work this week.
The FDA has two PDUFA dates on its calendar this week, with two extra days slotted for an advisory committee meeting to evaluate drug applications from three companies.
Intellia Therapeutics reported early, positive signs from its in vivo gene therapies for both transthyretin (ATTR) amyloidosis and hereditary angioedema Friday.
AstraZeneca’s rare disease subsidiary Alexion posted positive late-stage results from a potential complementary treatment for paroxysmal nocturnal hemoglobinuria.
The U.S. FDA approved Mallinckrodt’s Terlivaz in adults with hepatorenal syndrome and is the only FDA-approved drug for that indication.