TYME-18 leverages unique metabolism of cancer to create an intra-tumoral treatment for inoperable cancers
- TYME-18 leverages unique metabolism of cancer to create an intra-tumoral treatment for inoperable cancers
- Preclinical animal studies demonstrated TYME-18’s potential to dramatically shrink and/or resolve tumors
- TYME-18 treatment resulted in a 91.6% (11/12) complete resolution of established tumors within two weeks
- Novel mechanism of action contributes to a strong anti-cancer effect while minimizing systemic and local toxicity
- Preclinical studies are ongoing with a goal of identifying an IND-enabling program
NEW YORK--(BUSINESS WIRE)-- Tyme, Inc. Technologies, Inc. (NASDAQ: TYME), an emerging biotechnology company developing cancer metabolism-based therapies (CMBTs™), announced new preclinical findings that describe the unique anti-cancer effects of its cancer metabolism-based pipeline candidate, TYME-18, that are being presented at the American Association for Cancer Research 2020 Virtual Meeting from June 22 to June 24, 2020.
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20200623005100/en/
(Photo: Business Wire)
TYME’s CMBTs are proprietary investigational compounds that leverage cancer’s altered metabolism and associated vulnerabilities to specifically disrupt fundamental cellular processes. TYME-18 is focused on leveraging decreasing pH levels and toxic tumor environment, while compromising lipid barriers and membrane proteins.
"We believe the opportunity for a safe and effective intra-tumoral agent like TYME-18 could be substantial for both cancer patients with tumors that are difficult or impossible to surgically resect, as well as situations where radiotherapy is considered high-risk," said Dr. Giuseppe Del Priore, MD, Chief Medical Officer at TYME. “TYME-18’s preclinical development is ongoing with the goal of identifying an IND pathway during 2020.”
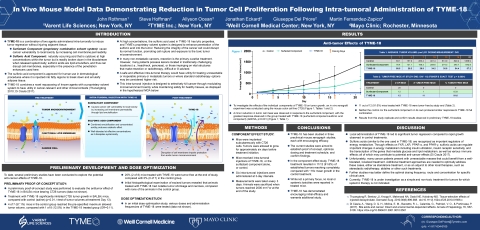
TYME-18 has been evaluated in three preclinical mouse xenograph studies, each with encouraging efficacy. To investigate the effects of the individual components of TYME-18 on tumor growth, an in vivo xenograft experiment was conducted using the mouse colon cell line CT26 (Figure 1). A minor reduction in tumor cell mass was observed in response to TYME-18’s surfactant component alone, with a statistically significant response observed in the group treated with TYME-18 (surfactant component + sulfonic acid component)1,2 (p=0.001) (Figure 1). The preclinical data showed that 91.6% (11/12) of mice treated with TYME-18 were tumor-free by study end compared with tumor growth of approximately 16 times mean growth in the control group. None of the mice in the control or surfactant alone group achieved tumor-free status, although 25% (3/12) of the surfactant alone group had tumor shrinkage from baseline. Results from this study replicate and confirm observations in preliminary TYME-18 studies.
While not a primary focus of the preclinical studies, TYME-18 did not show local or systemic toxicities in treated mice.
Results of this preclinical study were presented at the American Association for Cancer Research Virtual Meeting from June 22 to June 24, 2020. The poster is available on our website (www.tymeinc.com/data-publications).
Details for the TYME-18 poster presentation are as follows:
Title: In Vivo Mouse Model Data Demonstrating Reduction in Tumor Cell Proliferation Following Intra-tumoral Administration of TYME-18
Authors: John Rothman1, Steve Hoffman2, Jonathan Eckard2, Giuseppe Del Priore2, Allyson Ocean3, Martin Fernandez-Zapico4
Institutions: (1) Varent Life Sciences, New York, NY, (2) TYME Inc., New York, NY, (3) Weill Cornell Medical Center, New York, NY, (4) Mayo Clinic, Rochester, MN
Virtual Session Date: June 22-24, 2020
Virtual Session Location: AACR e-poster website
Abstract Number: 20-A-6858
About TYME-18
TYME-18 is composed of a proprietary surfactant delivery agent with a specific sulfonic acid component2. It is designed for intra-tumoral administration of difficult to treat tumors and leverages the acidic tumor microenvironment and signaling pathways to kill cancer cells. TYME-18 is distinct in composition, but like SM-88, aims to leverage susceptibilities of a cancer that are related to its altered metabolism. Initial preclinical data for TYME-18 in animal tumor models demonstrate rapid and complete tumor regression, with no reported local or systemic toxicities. TYME-18 continues to be studied as a potential therapy for difficult to treat tumors that may not be eligible for surgical or other interventions.
About SM-88
SM-88 is an oral investigational modified proprietary tyrosine derivative that is believed to interrupt the metabolic processes of cancer cells by breaking down the cells’ key defenses and leading to cell death through oxidative stress and exposure to the body’s natural immune system. Clinical trial data have shown that SM-88 has demonstrated encouraging tumor responses across 15 different cancers, including pancreatic, lung, breast, prostate and sarcoma cancers with minimal serious grade 3 or higher adverse events.
About Tyme Technologies
Tyme Technologies, Inc., is an emerging biotechnology company developing cancer therapeutics that are intended to be broadly effective across tumor types and have low toxicity profiles. Unlike targeted therapies that attempt to regulate specific mutations within cancer, the Company’s therapeutic approach is designed to take advantage of a cancer cell’s innate metabolic weaknesses to compromise its defenses, leading to cancer cell death through oxidative stress and exposure to the body’s natural immune system. For more information, visit tymeinc.com. Follow us on social media: Twitter, LinkedIn, Instagram, Facebook and YouTube.
Forward-Looking Statements/Disclosure Notice
In addition to historical information, this press release contains forward-looking statements under the Private Securities Litigation Reform Act that involve substantial risks and uncertainties. Such forward-looking statements within this press release include, without limitation, statements regarding our drug candidates, including SM-88 and TYME-18, and their clinical potential and non-toxic safety profiles, our drug development plans and strategies, ongoing and planned clinical trials, preliminary data results and the therapeutic design and mechanisms of our drug candidates; and readers can identify forward-looking statements by sentences or passages involving the use of terms such “believes,” “expects,” “hopes,” “may,” “will,” “plan,” “intends,” “estimates,” “could,” “should,” “would,” “continue,” “seeks,” or “anticipates,” and similar words including their use in the negative or by discussions of future matters such as effect of the novel coronavirus (COVID-19) pandemic and the associated economic downturn and impacts on the Company's ongoing clinical trials, the cost of development and potential commercialization of our lead drug candidate and of other new products, expected releases of interim or final data from our clinical trials, possible collaborations, the timing, scope and objectives of our ongoing and planned clinical trials and other statements that are not historical. The forward-looking statements contained in this press release are based on management’s current expectations, which are subject to uncertainty, risks and changes in circumstances that are difficult to predict and many of which are outside of TYME’s control. These statements involve known and unknown risks, uncertainties and other factors which may cause the Company’s actual results, performance or achievements to be materially different from any historical results and future results, performances or achievements expressed or implied by the forward-looking statements. These risks and uncertainties include, but are not limited to, the severity, duration, and economic impact of the COVID-19 pandemic; that the information is of a preliminary nature and may be subject to change; uncertainties inherent in the cost and outcomes of research and development, including the cost and availability of acceptable-quality clinical supply and the ability to achieve adequate clinical study design and start and completion dates; the possibility of unfavorable study results, including unfavorable new clinical data and additional analyses of existing data; risks associated with early, initial data, including the risk that the final data from any clinical trial may differ from prior or preliminary study data; final results of additional clinical trials that may be different from the preliminary data analysis and may not support further clinical development; that past reported data are not necessarily predictive of future patient or clinical data outcomes; whether and when any applications or other submissions for SM-88 may be filed with regulatory authorities; whether and when regulatory authorities may approve any applications or submissions; decisions by regulatory authorities regarding labeling and other matters that could affect commercial availability of SM-88; the ability of TYME and its collaborators to develop and realize collaborative synergies; competitive developments; and the factors described in the section captioned “Risk Factors” of TYME’s Annual Report on Form 10-K filed with the U.S. Securities and Exchange Commission on May 22, 2020, as well as subsequent reports we file from time to time with the U.S. Securities and Exchange Commission available at www.sec.gov.
The information contained in this press release is as of its release date and TYME assumes no obligation to update forward-looking statements contained in this release as a result of future events or developments.
1) Di Ciaula, A., Wang, D. Q. H., Molina, E. M., Baccetto, R. L., Calamita, G., Palmieri, V. O., & Portincasa, P. (2017). Bile acids and cancer: Direct and environmental-dependent effects. Annals of Hepatology, 16, S87- S105. https://doi.org/10.5604/01.3001.0010.5501
2) Thuangtong R, Bentow JJ, Knopp K, Mahmood NA, David NE, Kolodney MS. Tissue-selective effects of injected deoxycholate. Dermatol Surg. 2010;36(6):899‐908. doi:10.1111/j.1524-4725.2010.01566.x
View source version on businesswire.com: https://www.businesswire.com/news/home/20200623005100/en/
Contacts
For Investor Relations & Media Inquiries:
Investor Relations
1-212-461-2315
investorrelations@tymeinc.com
media@tymeinc.com
Source: Tyme Technologies, Inc.








