Subtle Medical, Inc., a leading healthcare technology company using artificial intelligence (AI) to improve the speed and quality of medical imaging, announced the issuance of U.S. Patent No. 10,997,716 for their licensed innovative software that uses deep learning to drastically reduce contrast dosage needed during contrast-enhanced medical imaging exams.
|
MENLO PARK, Calif., May 4, 2021 /PRNewswire/ -- Subtle Medical, Inc., a leading healthcare technology company using artificial intelligence (AI) to improve the speed and quality of medical imaging, announced the issuance of U.S. Patent No. 10,997,716 for their licensed innovative software that uses deep learning to drastically reduce contrast dosage needed during contrast-enhanced medical imaging exams. The patent was invented by Drs. Greg Zaharchuk and Enhao Gong prior to co-founding Subtle Medical and is based on technology exclusively licensed from Stanford University. This marks another milestone in Subtle's mission to accelerate universal access to faster, safer, and smarter medical imaging. SubtleGAD's ability to reduce gadolinium dosage by 90% addresses real safety and liability concerns
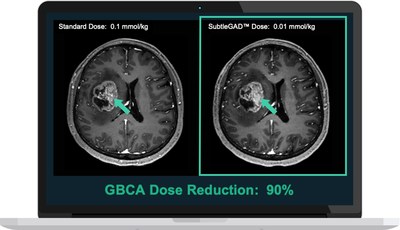
Contrast agents are routinely used in over 100 million medical imaging exams each year, including MR, CT, and Ultrasound, to improve the visualization of anatomies and possible pathologies. "This patent recognizes our AI software as a fundamentally new solution that can significantly improve the visualization before and after contrast administration with lower doses of contrast than are currently used," said Enhao Gong, PhD, Co-founder and CEO of Subtle Medical. "As a result, significantly reduced contrast agent dosage can be used to obtain diagnostic information in medical imaging, enabling safer exams for patients worldwide." Subtle Medical's vendor-neutral software derived from the patented technology, SubtleGAD™ (currently in the investigational phase), uses proprietary deep learning (DL) algorithms to achieve equivalent diagnostic image quality using only 10% of the gadolinium contrast dose when compared to MRI images conducted using the full dose. When lower gadolinium dosage is administered for any reason, SubtleGAD™ software can improve image quality, diagnostic accuracy, and lesion detectability. The ability to reduce gadolinium dosage by 90% when combined with SubtleGAD™ software addresses real and perceived safety and liability concerns for patients and imaging institutions around the long term effects of Gadolinium-based contrast agents (GBCAs) used in contrast-enhanced MRI procedures. Lowering contrast dosage has the potential to benefit tens of millions of patients, including pediatric and neonatal patients, as well as patients with chronic neurological diseases such as multiple sclerosis (MS) who have frequent contrast-enhanced MRI exams. Other potential applications include breast MRI studies, used for screening and diagnostic studies in patients with dense breasts, patients with advanced chronic kidney disease (CKD) or marginal kidney function issues who cannot currently undergo full-dose contrast-enhanced MRI, and other patients with concerns about gadolinium retention in the body. Research on the newly patented technology was published in the Journal of Magnetic Resonance Imaging (JMRI) in 2018 demonstrating feasibility, and it was highlighted by RSNA as one of the key advances of the year, winning an RSNA research prize. Subtle received a $1.6 million SBIR NIH Grant in 2019 to pursue research of SubtleGAD™, and in 2020, Phase II funding was awarded based on the results achieved during Phase I, confirming the scientific and technical merit of the research. Several papers highlighting the clinical validation and technical aspects of the work have recently been published using the SubtleGAD™ technology. A recent study by radiologists at Tiantan Hospital was published in the journal European Radiology demonstrating improved image quality and excellent lesion detectability. Another manuscript published in Magnetic Resonance in Medicine (MRM) highlighted recent progress on the technical improvements that have been made by the company since the initial development of the patent. These clinical studies have further validated the efficacy of SubtleGAD™ and its corresponding AI technology by highlighting the generalizability across a large cohort of patients from multiple institutions acquired with various scanner models and GBCA products, and also shows significant potential and value to clinical applications. "The diagnostic quality seen with low dose images enhanced by SubtleGAD's state-of-the-art technology has shown to be clinically equivalent to the full-dose images in our research," said Greg Zaharchuk, MD, PhD, Co-founder of Subtle Medical. "By adding novel products like SubtleGAD to their AI portfolio, centers will be able to address concerns around gadolinium contrast retention from MRI exams, providing peace of mind and ultimately better patient care." SubtleGAD™ is the third software product under development in Subtle Medical's growing portfolio of AI technologies aimed at improving the quality, safety and efficiency of medical imaging. SubtleMR™ applies two methods of image enhancement—denoising and increasing image sharpness—to improve the image quality of standard and accelerated MRI protocols, while SubtlePET™ denoises PET images that were conducted in 1/4th of the original scan duration. Both vendor-neutral products are FDA-cleared and CE-marked and are in clinical use in multiple centers in the US and abroad. About GBCAs The U.S. Food and Drug Administration (FDA) required a new class warning and other safety measures for all gadolinium-based contrast agents (GBCAs) for magnetic resonance imaging (MRI) concerning gadolinium remaining in patients' bodies, including the brain, for months to years after receiving these drugs. Many GBCAs have a black box warning linking them to the development of nephrogenic systemic fibrosis (NSF), with higher risk from repeat dosing. The more recent identification of gadolinium deposition within the brain and body has raised safety and liability concerns about the usage of GBCAs for both patients and imaging institutions. The long-term effects of gadolinium deposition are still unclear. About Subtle Medical Subtle Medical, Inc. is a healthcare technology company with deep learning solutions that increase the quality and efficiency of medical imaging. It was named CB Insights Top AI 100 and Digital Health 150 company in 2020 and is an Nvidia Inception Award Winner. Current clinical partners include UCSF, Hoag Hospital, Mount Sinai, Sickkids, Radnet, DASA, Tiantan Hospital, and Middlesex Hospital, among others. Subtle is currently in collaboration with top-tier medical device vendors, including Siemens Healthineers, Philips (China), and several pharmaceutical partners. For more information or to inquire about a clinical collaboration, please visit subtlemedical.com or email sales@subtlemedical.com. Press Contact: Anna Menyhart-Borroni Head of Marketing
SOURCE Subtle Medical, Inc. |