RNA caught the public’s imagination in the form of mRNA vaccines against COVID-19, but RNA can be used in other ways, too, to make therapeutic proteins.
“RNA therapeutics are experiencing a Renaissance,” Edward Kaye M.D., CEO of Stoke Therapeutics, told BioSpace. “RNA is a way to provide genetic therapy without altering the human genome.”
RNA caught the public’s imagination in the form of mRNA vaccines against COVID-19, but RNA can be used in other ways, too, to make therapeutic proteins. For example, “RNA splicing happens all the time. It’s a critical cell function. There are roughly 3 billion pieces of DNA and only about 30,000 proteins, so those proteins are manipulated in lots of different ways that give the body more flexibility with these limited pieces of information.”
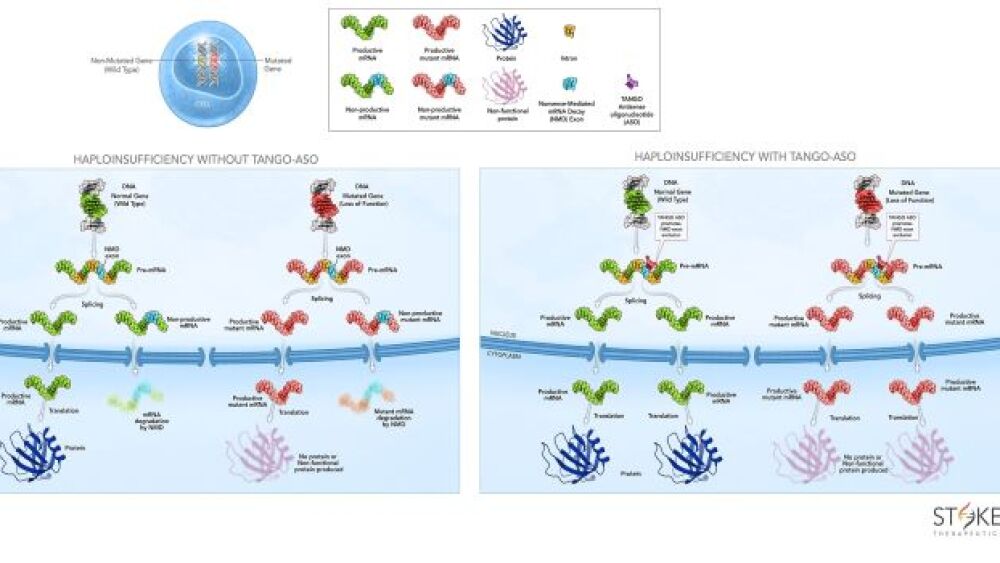
Stoke Therapeutics is using RNA splicing to upregulate protein in the body to target genetic diseases in which half the protein is missing because of a mutation in one copy of a gene, known as haploinsufficiencies. In this way, the body uses its own production system to increase the production of specific, too-low proteins to near-normal levels.
“We’re targeting conditions in which there is one normal and one abnormal copy of the gene and are upregulating the normal copy,” he said. “In animal models, we have shown that upregulating the missing protein to near normal levels corrects the underlying genetic disorder. Unlike gene therapy, this RNA therapy may need to be maintained for life.”
Gene therapy has a “one-and-done” reputation, but it isn’t as simple as that implies. Specifically, Kaye asked, “How long will the transgene continue to work? Eventually it may quit and you can’t redose because your body developed immunity to the viral vector used to deliver the transgene.”
RNA splicing overcomes the targeting issue, too.
“We let the cell upregulate its own mRNA,” he said. Therefore, “Cells can only upregulate protein production if they have the message to make the specific protein of interest.”
That serves as a safety limit to protein production.
“In about 50% of the genes, there are retained pieces of RNA that get stuck and aren’t spliced out.” Therefore, “only half of the targets will make full-length mRNA, to create more protein,” he explained.
Stoke Therapeutics’ technology, targeted augmentation of nuclear gene output (TANGO), has the potential to treat a broad spectrum of genetic diseases, designing antisense oligonucleotides that bind to pre-mRNA to help the target genes stoke protein production.
The lead program focuses on genetic epilepsy – specifically, Dravet syndrome.
“This disorder begins in infancy, in which children are missing a sodium channel that is essential for normal electrical function of the brain,” Kaye said. “Ion channels – sodium, potassium, and chloride – regulate energy and electrical transmission within the cell, so if those channels are missing certain proteins, it becomes critical.”
In Dravet syndrome, seizures are associated first with fevers, then occur without fevers, and increase until they may happen hundreds of times per day. Intellectual disability, developmental delays, move and balance issues, language and speech disturbances, growth defects, sleep abnormalities, chronic infections, disruptions of the autonomic nervous system and mood disorders also ensue. Approximately 20% of these patients die before reaching their teenage years.
“We’re trying to treat the underlying cause,” Kaye said.
The company also recently presented preclinical data for autosomal dominant optic atrophy (ADOA), showing that antisense oligonucleotides increased production of the protein OPA1, thus reducing the deficiency that is the main cause of ADOA. Additional programs are looking at addressing haploinsufficient diseases of the central nervous system, eye, kidney and liver.
Notably, splice technology can tackle diseases that mRNA can’t address. One of the challenges, Kaye explained, is that mRNA lasts a very short time – no more than minutes because it is degraded by ribonucleases (RNases).
“We use a fully modified backbone that resists degradation by RNases,” he said. Its longevity varies, but when administered to the vitreous for certain ophthalmic diseases, it lasts as long as a year. In other diseases, it may function for four to six months. “Then it is excreted naturally,” Kaye explained.
“The challenge that all antisense oligonucleotides have is that certain organs are more resistant to taking it up than others,” he continued.
For example, when injected into the vitreous, it is taken up by the retina, but not when it is administered intravenously. For the central nervous system, it is administered intrathecally, but when administered systemically, it won’t pass through the brain-blood barrier. Adding peptides or antibodies, for example, to the oligo can overcome some of those challenges so it can be delivered to the organ where it’s needed.