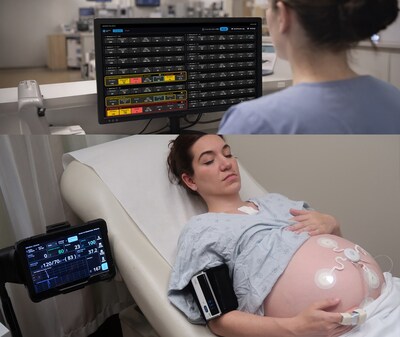
CHICAGO, April 9, 2026 /PRNewswire/ -- Sibel Health today announced FDA 510(k) clearance of ANNE® Maternal, the world's first fully wireless, comprehensive maternal-fetal monitoring platform. Designed for continuous, simultaneous monitoring of maternal and fetal health, ANNE® Maternal combines advanced wearable sensor technology with integrated clinical decision support — and represents a critical step toward making continuous maternal monitoring a universal standard of care. ANNE® Maternal is a sister product to ANNE® One monitoring platform already FDA-cleared for continuous monitoring of adolescents and adults in the home and hospital setting.
A New Standard in Maternal-Fetal Monitoring
ANNE® Maternal leverages soft, flexible epidermal wearable sensors that wirelessly stream continuous maternal vital signs — including heart rate, respiratory rate, blood oxygen saturation, and skin temperature — alongside wireless fetal heart rate monitoring and uterine contraction detection. Unlike conventional systems that restrict laboring patients to beds with cumbersome wired equipment, ANNE® Maternal is fully wireless, enabling patient mobility and comfort without sacrificing clinical rigor.
The platform delivers real-time bedside and central monitoring with visual and audio alarms, and incorporates an integrated electronic Modified Early Obstetric Warning System (MEOWS) — providing clinicians with automated signals when parameters warrant prompt evaluation. ANNE® Maternal is also compatible with wireless point-of-care ultrasound enabling a single unified interface for comprehensive perinatal assessment.
The clearance arrives at a critical moment for maternal health in the United States. The U.S. maternal mortality rate stands at 22 deaths per 100,000 live births — the highest of any high-income nation and more than double the rate of most comparable countries. For the fourth consecutive year, the March of Dimes awarded the U.S. a D+ in maternal and infant health, describing the system as "dangerously stalled." ANNE® Maternal directly addresses this crisis by enabling the kind of continuous, comprehensive monitoring for both intrapartum and postpartum settings that has long been standard in other domains of acute care but has remained largely absent from labor and delivery.
Foundational and Next Generation Technology Development Grants Facilitate Global Reach
The development of ANNE® Maternal has been anchored by a $17.5 million grant from the Gates Foundation, awarded in early 2024 to advance continuous maternal monitoring across low- and middle-income countries (LMICs). Sibel Health was originally spun out of Northwestern University with early seed support from the Gates Foundation — making today's FDA clearance a full-circle moment for the company's founding mission.
"What makes this platform truly unique is its dual mandate: it meets the rigorous clinical standards demanded by the FDA while also being designed for deployment in the world's most resource-limited settings. That convergence — high-income market validation and global health impact in a single platform — is what we've been building toward since day one. We are deeply grateful to the Gates Foundation, whose continued investment has made this product possible, and to our global partners whose clinical expertise and dedication to global health have been indispensable. Together, we are closer than ever to making continuous maternal monitoring a universal standard of care," said Steve Xu, MD, CEO and Co-Founder of Sibel Health.
ANNE® Maternal was featured in the 2026 Gates Foundation Annual Letter as a potential tool to achieve the Foundation's 2045 goal that no mother, baby, or child dies of a preventable cause. Sibel receives an additional $5 million grant to develop future AI-powered features for ANNE® Maternal designed specifically for low-resource settings and cost reductions. These capabilities build on Sibel Health's existing patient monitoring infrastructure and are expected to be integrated into future product releases.
Global Deployments and a Dual-Market Model
ANNE® Maternal is already deployed across four countries — India, Pakistan, Nigeria, and Rwanda — in a research partnership with the University of Edinburgh and the NIHR Global Health Research Unit on Global Surgery. Globally, approximately 700 women die every day in pregnancy or childbirth, the vast majority in settings without access to vital signs monitoring.
"The day of birth should be a joyful one, but it is also a time where mothers and babies can be vulnerable to devastating complications. The ANNE® Maternal system uses low-profile wearable sensors to allow birthing mothers comfort, mobility, and comprehensive state-of-the-art vital signs monitoring. Global researchers from Christian Medical College Ludhiana (Punjab, India), Aga Khan University (Karachi, Pakistan), Lagos University (Lagos, Nigeria), and University Teaching Hospital of Kigali at the University of Rwanda (Kigali, Rwanda) are using these sensors in places where the need is the greatest to understand how best to detect and prevent complications in childbirth, and support women laboring to give birth to the future," said Dr. Rosemary Townsend MBChB MRCOG MD, Senior Lecturer and Honorary Consultant in Obstetrics at the University of Edinburgh. "In our setting, the clinician-to-patient ratios are heavily constrained and early detection of obstetric complications could be life-saving at scale," adds Dr. Kavita Bhatti, Professor and Department Chair of Obstetrics and Gynecology of Christian Medical College and Hospital Ludhiana.
"FDA clearance of ANNE® Maternal reflects the kind of rigorous, science-driven review process that gives clinicians and patients confidence in the devices they rely on whether deployed in the U.S. or in low resource settings. We are grateful for the FDA's thorough engagement throughout the submission, and their commitment to advancing innovative digital health technologies," said Indira Malladi, Senior Regulatory Affairs Specialist and lead regulatory author of the ANNE® Maternal 510(k) submission.
About Sibel Health
Sibel Health is an award-winning digital health company with a mission to deliver Better Health Data for All®. Based in Chicago with an international office in Seoul, the company's FDA-cleared ANNE® platform includes advanced wearable sensors, AI-enabled data analytics, and an integrated mobile software and cloud platform. The company's Discovery platform is used by the world's leading pharmaceutical companies for clinical trials. For more information, visit www.sibelhealth.com, follow us on LinkedIn.
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/sibel-health-receives-fda-clearance-for-anne-maternal-a-comprehensive-and-fully-wireless-maternal-fetal-monitoring-platform-302737398.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/sibel-health-receives-fda-clearance-for-anne-maternal-a-comprehensive-and-fully-wireless-maternal-fetal-monitoring-platform-302737398.html
SOURCE Sibel Health