Data validate the efficacy, safety, and tolerability of elismetrep, an investigational first-in-class TRPM8 migraine-associated channel blocker (MACB), as a novel approach for treating migraine
Potential for a new mechanism to help address the debilitating impact of migraine
Registrational program anticipated to initiate in mid-2026
NEW YORK--(BUSINESS WIRE)--Kallyope is a late-stage biotechnology company focused on developing innovative migraine and metabolic disease therapies for health challenges faced by hundreds of millions of people globally. Today, the Company presented results from a Phase 2b dose-ranging and proof of concept study of elismetrep, a novel TRPM8 channel blocker and the only agent known to be in late-stage development for the acute treatment of migraine. The data were reported during an oral presentation titled, “TRPM8 Antagonism with Elismetrep: A Novel Approach for Treating Migraine,” at the 2026 American Academy of Neurology (AAN) Annual Meeting underway in Chicago, IL.



“Migraine is a highly disabling neurological disorder with attacks of severe throbbing pain, light sensitivity, nausea, and other debilitating symptoms that routinely force people to put their lives on hold without notice. Many migraine patients remain dissatisfied with currently available therapeutic options and regularly switch between medicines to seek relief,” said Peter J. Goadsby, MD, PhD, FRS, King Abdullah University of Science and Technology, Saudi Arabia. “Elismetrep has the potential to introduce an entirely new class of medicines, the TRPM8 migraine-associated channel blockers (MACB), to the millions of people suffering from migraine in need of more effective relief.”
Phase 2b Study Overview and Key Data
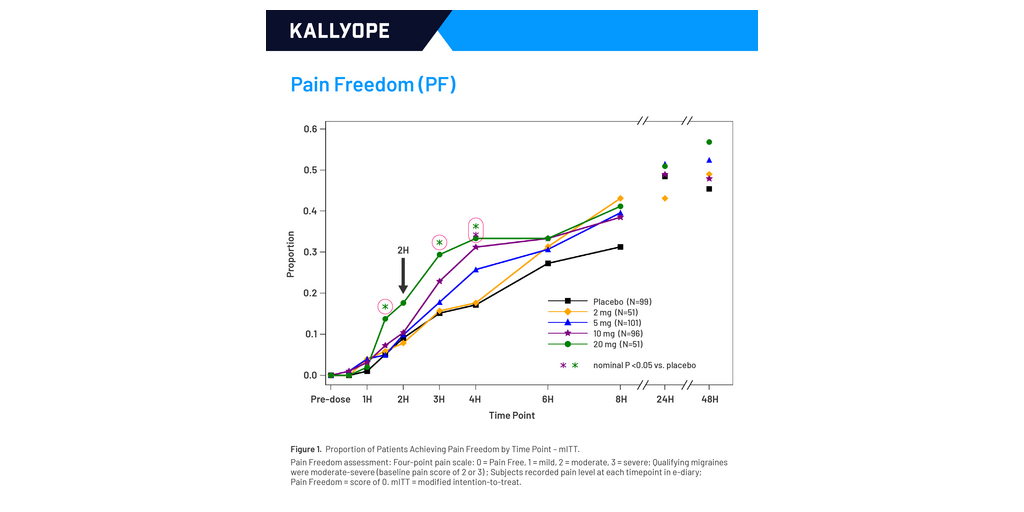
The study was a randomized, placebo-controlled, double-blind Phase 2b study (NCT06848075). Participants with 2-10 migraine attacks/month were randomized 2:1:2:2:1 to treat one migraine of moderate-severe pain with placebo or 2, 5, 10, or 20 mg elismetrep. Randomization was stratified by use of migraine preventive agents. Efficacy endpoints, assessed at 2h, were pain freedom (primary), freedom from most bothersome symptom (secondary), and pain relief (secondary). Efficacy data were recorded in real time in a patient-reported electronic-diary (e-Diary). Two-hour data were also captured by interview for those subjects with missing 2h e-Diary entries (All Data). Primary analyses used only e-Diary data, and a pre-specified supporting analysis of the primary endpoint used All Data.
Of 431 participants randomized, 398 treated a qualifying migraine (mean age 46 years; 87% female; 35% on migraine preventive agents; 31% triptan resistant). Overall, elismetrep showed dose- and time-dependent efficacy with the 20 mg dose providing the greatest effects (see graphs).
In the primary and supporting analyses for 2h pain freedom, response rates for elismetrep 20 mg vs. placebo were 17.6% vs. 9.1% (odds ratio [OR]=2.1, P=0.0650) (e-Diary only) and 19.6% vs. 9.1% (OR=2.4, nominal P=0.0344) (All Data) respectively. Response rates for 2h freedom from most bothersome symptom for elismetrep 20 mg vs. placebo were 38.3 % vs. 24.2% (OR=1.9, nominal P=0.0425). Response rates for 2h pain relief for elismetrep 20 mg vs. placebo were 58.8% vs. 42.4% (OR=1.9, nominal P=0.0293).
There were no serious adverse events (AEs). The incidence of AEs was dose dependent. At 20 mg, all AEs were mild (70%) or moderate. The most common AEs in the 20 mg group were oral paresthesia (9.8%), feeling hot (9.8%), paresthesia (7.8%), hot flush (7.8.%), and flushing (5.9%).
“These data are incredibly exciting for migraine patients, their physicians, and Kallyope. With only ~30% of migraine sufferers experiencing adequate and consistent relief with current therapeutics, there is a dire need for innovation,” said Brett Lauring, MD, PhD, Chief Medical Officer, Kallyope. “By targeting an entirely new part of the migraine cascade, we are hopeful that elismetrep can provide a safe and effective option for patients. We believe these data support the initiation of the registrational trials for elismetrep, anticipated to begin in mid-2026.”
New Elismetrep Formulation
Kallyope has developed a new liquid-filled softgel capsule (LSGC) formulation of elismetrep that results in substantially more rapid absorption compared to the formulation used in Phase 2b. The LSGC formulation will be used in future trials and is expected to result in earlier onset of migraine relief and enhanced efficacy at 2 hours (the timepoint where the primary endpoints for registrational trials are assessed). Full pharmacokinetic data from the LSGC formulation will be presented at an upcoming medical meeting in 2026.
“The opportunity to provide a new treatment option for millions of people who suffer the debilitating effects of migraine is unique and important. Kallyope is committed to advancing this program rapidly to bring this potentially meaningful new medicine to patients as quickly as possible,” said Jay Galeota, CEO and President, Kallyope.
About Transient Receptor Potential Melastatin 8 (TRPM8)
Migraine is increasingly understood as a disorder of trigeminal sensory neuron hyperexcitability, in which trigeminal sensory pathways become sensitized, leading to sensory hypersensitivity and amplified pain signaling. TRPM8 is a polymodal sensory ion channel expressed on a distinct subset of trigeminal sensory neurons relevant to migraine pain signaling, migraine-associated channels. In the context of migraine, TRPM8 activity is associated with trigeminal sensory neuron pathway activation, contributing to pain onset and heightened, sustained pain signaling. TRPM8 is a mechanistically distinct therapeutic target from current migraine treatments, including calcitonin gene-related peptide (CGRP) receptor antagonists and serotonergic (5-HT)-based agents, and is the only TRP channel with an established genetic association to migraine in genome-wide association studies.
About Elismetrep
Elismetrep (K-304) is an investigational, novel, oral, TRPM8 migraine-associated channel blocker (MACB) that is highly selective for TRPM8 migraine-associated channels versus other TRP channels. Specifically designed to block TRPM8 activity, elismetrep has been shown to reduce migraine pain and associated symptoms. The Phase 2b dose-ranging and proof of concept trial for elismetrep for the acute treatment of migraine in adults demonstrated clinically meaningful efficacy that is competitive with the current leading marketed therapies. Registrational trials for elismetrep for the acute treatment of migraine are planned to initiate by mid-2026. Elismetrep is an investigational product and has not been approved by the FDA or any other regulatory agency.
About Kallyope
Kallyope is a late-stage biotechnology company focused on developing innovative migraine and metabolic therapies for health challenges faced by hundreds of millions of people globally. Kallyope’s lead programs target previously unknown drivers of disease in neural signaling pathways. The Company’s most advanced candidate, elismetrep (K-304), is poised to begin registrational trials for the acute treatment of migraine in mid-2026. The metabolism pipeline includes agents that target a novel target identified and validated by the Company’s Klarity™ platform, as well as oral small molecule approaches to the highly validated amylin pathway for the treatment of obesity. Kallyope was founded by world-leading neuroscientists and continues to explore the role of neural circuits in driving disease.
For more information, visit www.kallyope.com.
Contacts
Media Contact:
Ten Bridge Communications
Michael Galfetti
TBCKallyope@tenbridgecommunications.com
Investor Contact:
Kallyope
ir@kallyope.com




