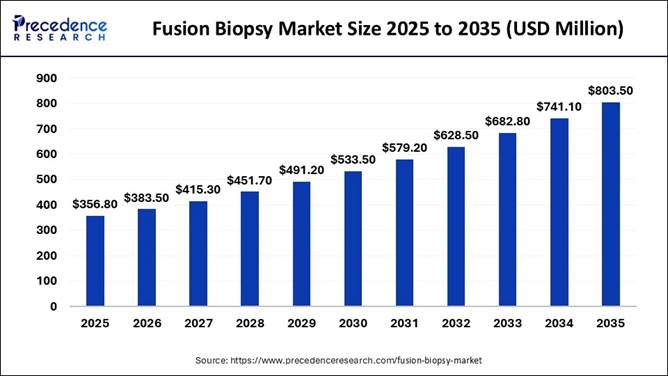
The global fusion biopsy market size is expected to be worth nearly USD 803.50 million by 2035, increasing from USD 383.50 million in 2026, driven by rising prostate cancer cases, growing adoption of AI-powered MRI-ultrasound fusion technologies, and increasing demand for minimally invasive precision cancer diagnostics across hospitals and imaging centers.
The fusion biopsy market is poised to grow at a compound annual growth rate (CAGR) of 8.60% from 2026 to 2035. From centralized, hospital-based imaging to accessible outpatient and ambulatory care centers, the fusion biopsy market is undergoing a significant transformation primarily driven by the need for improved detection accuracy in prostate cancer.
The market is moving away from random, systematic sampling towards AI-enhanced MRI-ultrasound fusion techniques. By integrating high-resolution MRI data with real-time ultrasound imaging, clinicians can now target suspicious lesions with greater precision. The rapid adoption of advanced, minimally invasive navigation platforms is redefining the standard of care for early cancer detection.
Where Data Meets Strategic Clarity 📥 View Sample Pages of the Complete Report 👉 https://www.precedenceresearch.com/sample/5350
Key Takeaways
🔹North America dominated the global fusion biopsy market in 2025, accounting for 41% of the total market share due to advanced healthcare infrastructure and high adoption of MRI-guided biopsy technologies.
🔹Asia-Pacific is projected to grow at the fastest CAGR from 2026 to 2035, driven by rising healthcare investments and increasing awareness of early cancer diagnosis.
🔹By product type, the consumables and accessories segment held the largest market share in 2025 owing to the recurring demand for biopsy needles, probes, and procedural accessories.
🔹By product type, the software segment is expected to witness the fastest growth between 2026 and 2035 due to advancements in AI-powered imaging and fusion guidance technologies.
🔹By biopsy route, the transrectal (TRUS-MRI fusion) segment accounted for the highest market share in 2025 because of its widespread clinical adoption and procedural familiarity.
🔹By biopsy route, the transperineal (TP-MRI fusion) segment is anticipated to grow at a strong CAGR through 2035 owing to lower infection risks and improved biopsy accuracy.
🔹By fusion technique, the software-based fusion segment captured a major market share in 2025 due to its efficiency in combining MRI and ultrasound imaging for targeted biopsies.
🔹By fusion technique, the in-bore MRI fusion segment is projected to expand notably from 2026 to 2035 driven by increasing demand for highly precise lesion targeting.
🔹By application, the prostate cancer diagnosis segment held the largest market share of 41% in 2025 due to the growing prevalence of prostate cancer globally.
🔹By application, the renal (kidney) lesion evaluation segment is expected to grow at a healthy CAGR between 2026 and 2035 as fusion imaging gains traction in kidney diagnostics.
🔹By end user, hospitals and tertiary care centers contributed the highest market share in 2025 due to access to advanced imaging systems and specialized healthcare professionals.
🔹By end user, ambulatory surgical centers (ASCs) are expected to grow at a strong CAGR from 2026 to 2035 because of the increasing preference for cost-effective outpatient procedures.
🔹By distribution channel, the direct sales segment held a dominant market share in 2025 supported by strong manufacturer relationships with healthcare institutions.
🔹By distribution channel, the indirect sales segment is projected to expand at a notable CAGR through 2035 due to growing distributor networks and expanding reach in emerging markets.
The fusion biopsy market is undergoing a significant transition from conventional imaging-assisted diagnostics toward AI-enabled precision oncology ecosystems. Advances in MRI-ultrasound fusion, robotic-assisted targeting, and real-time lesion mapping are improving diagnostic confidence while supporting minimally invasive cancer care pathways. As healthcare systems continue emphasizing early detection and personalized treatment strategies, fusion biopsy technologies are expected to play an increasingly important role in the future of precision cancer diagnostics.
Market Overview: The New Era of Fusion Biopsy Technology
The fusion biopsy market is experiencing a rapid and high-precision transformation, with projections indicating significant growth as it revolutionizes prostate cancer detection. By expertly combining 3D MRI data with live ultrasound, this innovative technology allows doctors to locate suspicious lesions with unparalleled accuracy, surpassing the limitations of traditional blind biopsies.
The growing global burden of prostate cancer continues to remain one of the major factors driving demand for advanced diagnostic technologies. According to global cancer health statistics, prostate cancer remains among the most commonly diagnosed cancers in men, particularly across aging populations in North America, Europe, and parts of Asia-Pacific. Rising awareness regarding early cancer detection, combined with increasing adoption of precision oncology practices, is encouraging healthcare providers to transition toward targeted biopsy procedures capable of improving diagnostic accuracy while minimizing unnecessary interventions.
This market is expanding due to the rising prevalence of prostate cancer, a high demand for minimally invasive, AI-guided procedures, and favorable reimbursement policies, establishing it as a crucial and growing force in targeted oncology care.
➡️ Become a valued research partner with us ☎ https://www.precedenceresearch.com/schedule-meeting
Integrating AI-Enhanced Autonomous Fusion Platforms: Major Potential
The integration of artificial intelligence (AI) and machine learning into MRI-ultrasound fusion platforms is transforming prostate cancer diagnostics from manual to autonomous processes. This shift improves precision, accelerates workflows, and reduces operator dependence. Technologies such as Koelis Trinity 3D and Esaote UroFusion can minimize registration errors to under 2-4 mm, resulting in higher detection rates of clinically significant prostate cancer and fewer false positives. The field is rapidly advancing towards full, end-to-end AI automation.
The Cost of Superior Care: Major Limitation
Despite the advantages of advanced fusion biopsy systems, the industry faces significant adoption challenges due to high upfront capital costs, specialized training requirements, and inconsistent insurance reimbursement. This financial burden creates a barrier to access, preventing smaller hospitals and low-to-middle-income regions from obtaining superior diagnostic capabilities. Consequently, the industry remains reliant on less accurate traditional transrectal ultrasound methods, widening the global gap in prostate cancer care.
Discover What’s Driving the Market Forward👉 https://www.precedenceresearch.com/fusion-biopsy-market
Fusion Biopsy Market Challenges
In addition to high equipment costs, the market also faces challenges related to limited MRI accessibility, shortage of trained imaging specialists, and workflow integration complexities within smaller healthcare facilities.
The adoption of advanced fusion biopsy systems often requires specialized physician training, software compatibility, and high-end imaging infrastructure, which can slow implementation across resource-constrained regions. Variability in reimbursement coverage and limited awareness in developing healthcare systems may also continue to impact adoption rates in certain markets.
Regulatory and Reimbursement Landscape Supporting the Expansion of Fusion Biopsy Technologies
The global fusion biopsy market is gaining strong momentum as healthcare systems increasingly recognize the clinical advantages of MRI-guided targeted biopsy procedures over conventional random sampling techniques. Regulatory bodies, reimbursement agencies, and medical associations are actively supporting the transition toward more accurate, minimally invasive diagnostic approaches for prostate cancer detection and monitoring.
In the United States, continued approvals and clearances from the U.S. Food and Drug Administration (FDA) are encouraging wider adoption of advanced MRI-ultrasound fusion platforms, AI-enabled lesion detection software, and robotic-assisted biopsy systems. These developments are helping healthcare providers improve targeting precision, reduce unnecessary repeat biopsies, and enhance overall diagnostic confidence in prostate cancer management.
Across Europe, the growing availability of CE-certified fusion biopsy technologies is supporting adoption across hospitals, imaging centers, and specialized oncology institutions. Healthcare providers in the region are increasingly shifting toward transperineal fusion biopsy procedures due to their lower infection risk, improved patient safety profile, and ability to deliver more accurate lesion targeting compared to traditional approaches.
Reimbursement support continues to play an important role in market expansion, particularly in developed healthcare systems. In the U.S., Medicare and private insurers have gradually expanded coverage pathways for MRI-guided and fusion-assisted biopsy procedures, improving patient access to advanced diagnostic solutions. Favorable reimbursement structures are also encouraging hospitals and outpatient facilities to invest in next-generation imaging and biopsy technologies.
In addition, growing endorsement from leading clinical organizations is reinforcing physician confidence in fusion-guided diagnostic workflows. Medical bodies such as the National Comprehensive Cancer Network (NCCN) and the American Urological Association (AUA) increasingly emphasize the importance of multiparametric MRI and targeted biopsy approaches for patients with elevated prostate cancer risk, previous negative biopsy findings, and active surveillance requirements.
As regulatory frameworks continue to evolve and reimbursement access improves across major healthcare markets, fusion biopsy technologies are expected to become increasingly integrated into routine precision oncology and advanced cancer diagnostic practices worldwide.
Fusion Biopsy Market Report Coverage
|
Market Scope |
Details |
|
Market Size in 2025 |
USD 356.80 Million |
|
Market Size in 2026 |
USD 383.50 Million |
|
Market Size in 2035 |
USD 803.50 Million |
|
Market Growth Rate (2026–2035) |
CAGR of 8.60% |
|
Largest Regional Market |
North America |
|
Fastest Growing Region |
Asia-Pacific |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2035 |
|
Key Growth Driver |
Rising prevalence of prostate cancer and growing demand for precision diagnostics |
|
Major Technology Trend |
AI-powered MRI-ultrasound fusion and real-time 3D imaging platforms |
|
Leading Biopsy Route |
Transrectal (TRUS-MRI Fusion) |
|
Fastest Growing Biopsy Route |
Transperineal (TP-MRI Fusion) |
|
Dominant Fusion Technique |
Software-Based Fusion |
|
Emerging Technology Segment |
In-Bore MRI Fusion |
|
Largest Application Segment |
Prostate Cancer Diagnosis |
|
Fastest Growing Application |
Renal (Kidney) Lesion Evaluation |
|
Leading End User |
Hospitals & Tertiary Care Centers |
|
Fastest Growing End User |
Ambulatory Surgical Centers (ASCs) |
|
Key Market Opportunity |
AI-enabled autonomous fusion platforms improving lesion targeting accuracy |
|
Major Market Challenge |
High equipment costs and limited reimbursement in developing regions |
|
Primary Clinical Benefit |
Higher detection accuracy with fewer unnecessary biopsies |
|
Segments Covered |
Product Type, Biopsy Route, Fusion Technique, Application, End User, Distribution Channel, and Regions |
|
Regions Covered |
North America, Europe, Asia-Pacific, Latin America, and Middle East & Africa |
|
Key Companies Profiled |
Philips, KOELIS, ESAOTE, Eigen, Hitachi, Exact Imaging, Biobot Surgical, BK Medical, Focal Healthcare |
|
Notable Industry Trend |
Increasing adoption of minimally invasive and AI-assisted biopsy procedures |
|
Innovation Focus Areas |
Robotic-assisted targeting, AI lesion mapping, deformable fusion imaging, and transperineal biopsy systems |
🔓 Instant Access. Zero Waiting. 📥 Buy the Premium Market Research Report
Now 👉 https://www.precedenceresearch.com/checkout/5350
Liquid Biopsy Market Strengthening the Future of
Precision Cancer Diagnostics The global Liquid Biopsy Market size is valued at USD 8.07 billion in
2026 and is predicted to grow from USD 9.24 billion in 2027 to USD
22.69 billion by 2034 with a strong CAGR of 13.91% from 2025 to 2034.
The market is witnessing substantial momentum due to the growing demand for
minimally invasive cancer diagnostics, rising adoption of precision oncology,
and increasing utilization of biomarker-based screening technologies across
healthcare systems worldwide. The rapid advancement of next-generation
sequencing (NGS),
circulating tumor DNA (ctDNA) analysis, AI-powered diagnostic platforms, and
real-time cancer monitoring solutions is transforming the global oncology
diagnostics landscape.
Liquid biopsy technologies are increasingly complementing imaging-guided
procedures such as fusion biopsy by enabling earlier cancer detection, therapy
selection, treatment monitoring, and personalized patient management with
reduced procedural burden. As healthcare providers continue shifting toward
non-invasive and highly accurate diagnostic pathways, the growing integration
of liquid biopsy with MRI-ultrasound fusion imaging, molecular diagnostics, and
AI-assisted oncology workflows is expected to accelerate innovation across
precision cancer care and targeted biopsy technologies globally. The Full Study is Readily Available |
Download the Sample Pages of this Report@ https://www.precedenceresearch.com/sample/5350 Fusion Biopsy Market Regional Analysis
North
America was the leading market in 2025, primarily due to the high prevalence of
prostate cancer, advanced healthcare infrastructure, and rapid adoption of
minimally invasive technologies. The U.S. and Canada feature well-established
hospital networks with extensive MRI capabilities, facilitating the widespread
adoption of fusion biopsy units. Medicare and
private insurance in the U.S. have also established specific reimbursement
codes for fusion biopsy procedures. The region serves as a hub for medical
innovation, with major industry players like GE Healthcare and Eigen actively
developing AI-driven imaging and robotic fusion technologies. The U.S.
leads the market as a premier research and development center for AI-enhanced
imaging and robotic-assisted systems. This leadership is fueled by strong
clinical endorsements from organizations such as the AUA and NCCN, along with
favorable reimbursement policies and a dense network of cancer treatment
centers. 🔸 In April 2026, KOELIS® will enhance prostate care
at EAU 2026 with the launch of ProMap® Smart for the Trinity® system. This
AI-driven innovation offers instantaneous, automatic 3D prostate segmentation
to improve the efficiency and precision of the MRI/Ultrasound fusion workflow.
(Source: https://koelis.com/press-details/koelis-announces-innovative-products-at-eau-2026-enhancing-mri-fusion-biopsy-workflows-and-promoting-focal-therapy-in-prostate-cancer/) What is
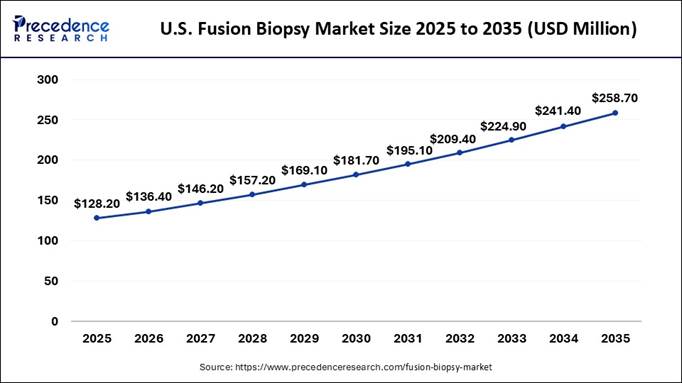
the U.S. Fusion Biopsy Market Size and Growth Rate? According to
Precedence Research, the U.S. fusion biopsy market size accounted for
USD 136.40 million in 2026 and is projected to reach approximately USD
258.7 million by 2035, growing at a CAGR of 7.4% from 2026 to 2035. Canada is
also accelerating the adoption of AI-driven fusion biopsy technology to achieve
higher precision diagnostics. Major institutions, such as the University of
Toronto, are pioneering advancements in image registration to meet the growing
demand for minimally invasive transperineal and transrectal techniques. Asia Pacific
region is expected to experience the fastest growth during the forecast period,
primarily driven by the rapid development of healthcare infrastructure,
increasing incidences of prostate cancer, and a rise in medical tourism. With
an aging population in countries like Japan and China, there is a significant
increase in prostate cancer cases that necessitate more precise diagnostics.
Emerging economies in the region are investing heavily in upgrading hospitals
and diagnostic centers with advanced, high-end imaging technology, leading to a
strong shift towards AI-integrated and 3D imaging systems that enhance lesion
detection and procedural accuracy. India is
primed as the emerging market within the region, mainly fueled by a sharp rise
in cancer cases and an urgent, tech-driven shift toward early, minimally
invasive diagnostics. High-end private hospitals are accelerating this boom,
leading in AI-powered MR fusion biopsy adoption, while medical tourism is
rapidly turning India into a global hub for affordable, advanced oncology care. 🔸 In July 2024, Sir HN Reliance Foundation Hospital
introduced Maharashtra's first AI-powered MR Fusion Biopsy machine,
significantly enhancing prostate cancer detection accuracy to 90%. This
advanced technology blends MRI scans with real-time ultrasound, offering a
faster, safer, and more precise diagnostic approach. (Source: https://www.healthcareradius.in/features/surgery/reliance-hospital-installs-mr-fusion-biopsy-machine) China
dominates the Asia-Pacific fusion biopsy market, leveraging high-volume demand,
government-backed screenings, and advanced CT/bronchoscopic technology
adoption. As a growing medical device manufacturing hub, the country is also
reducing costs for advanced diagnostics, strengthening its position as both a
key consumer and supplier. Have questions or ready to move forward? Contact our experts
today @ sales@precedenceresearch.com | +1 804
441 9344
Fusion Biopsy Market Segmental Analysis By Product
Type Analysis The
consumables and accessories segment led the market in 2025. This was primarily due to the
high-volume recurring revenue and the rising incidence of prostate cancer,
which necessitated higher procedure volumes. As a result, there was a constant
demand for specialized disposable biopsy needles and tracking kits. The shift
towards transperineal biopsy, seen as a cleaner procedure with a lower risk of
sepsis, required specific, specialized accessories, further increasing the demand
for disposable items. Newer fusion platforms utilize proprietary accessories
and disposable sensors for precise real-time guidance, making these components
essential for the procedures. By Biopsy
Route Analysis The
transrectal (TRUS-MRI fusion) segment also dominated the market in 2025. Its success was largely attributed
to its established reputation as the gold standard, high familiarity among
urologists, and ease of use in office settings. As the
preferred technique for decades, transrectal ultrasound-guided biopsy is
well-known to many urologists for its accessibility and efficiency. Most
commercially available fusion biopsy systems were initially designed for the
transrectal approach, facilitating easy adoption and wider availability. This
method provides high-precision targeting of prostate cancer by fusing MRI data
with live ultrasound, allowing for effective, rapid, and often outpatient
procedures. By Fusion
Technique Analysis The
software-based fusion segment held significant market dominance in 2025. This was largely due to its
superior cost-efficiency, ability to integrate with existing ultrasound
systems, and enhanced learning curves for practitioners. Software solutions
provide a cheaper alternative to hardware-heavy in-bore setups, allowing
institutions to utilize existing ultrasound technology alongside MRI data. Software-based
fusion offers a quicker learning curve for medical professionals, enabling
faster training and consistent accuracy. It allows for precise, real-time 3D
reconstruction and tracking of MRI-defined lesions in ultrasound images, resulting in higher cancer
detection rates. By
Application Analysis The
prostate cancer diagnosis segment led the market in 2025, primarily due to the increasing
demand for greater accuracy in detecting clinically significant cancers over
traditional methods. Fusion biopsy combines magnetic resonance imaging with real-time ultrasound to target
specific suspicious areas, addressing the shortcomings of traditional random
sampling, which often misses high-grade tumors. Given that
prostate cancer is among the top five cancers globally, the emphasis on early
screening and heightened awareness has increased the demand for advanced,
targeted diagnostic technologies to prevent unnecessary overtreatment. By
End-User Analysis Hospitals
and tertiary care centers dominated the market in 2025, largely because of their high
purchasing power for expensive equipment, superior infrastructure, and capacity
to handle high patient volumes. These facilities possess the necessary MRI
scanners and high-end ultrasound systems essential for image fusion, facilitating
on-demand biopsies. As primary
care settings for cancer diagnostics, hospitals manage significant volumes of
cases, making them the main purchasers of high-cost fusion systems. They
provide comprehensive care, from initial imaging and targeted biopsies to
pathology, creating a seamless workflow for patient management. By
Distribution Channel Analysis The
direct sales segment accounted for significant market dominance in 2025, mainly due to its effective role in
managing the high-capital, high-tech nature of MRI-ultrasound fusion systems
and fostering relationships with major hospital networks. Fusion
biopsy systems are high-value, sophisticated technologies that require
specialized training and installation, making direct-to-hospital sales more efficient.
This approach allows for better long-term technical support, software upgrades,
and training for specialized staff, facilitating the adoption of AI-powered
imaging workflows. ✚ Related Topics You May Find Useful: ➡️ Cancer Biopsy Market: Discover the growing role
of advanced biopsy technologies in improving cancer diagnosis and treatment
planning ➡️ Interventional
Radiology Products Market: Analyze how minimally invasive procedures are increasing
demand for innovative radiology products worldwide ➡️ Biopsy Devices Market: Gain insight into
technological advancements driving efficiency and accuracy in tissue sampling
procedures ➡️ Breast
Biopsy Devices Market: See how rising breast cancer screening initiatives are
fueling demand for advanced biopsy solutions ➡️ Endomyocardial
Biopsy Market: Understand the increasing importance of cardiac biopsy
procedures in diagnosing complex heart conditions ➡️ Breast
Cancer Core Needle Biopsy Market: Track the adoption of core needle
biopsy techniques for accurate and minimally invasive breast cancer diagnosis Fusion
Biopsy Market Top Companies
➢ Koninklijke
Philips N.V. ➢ Hitachi Ltd. ➢ MedCom ➢ ESAOTE SPA ➢ KOELIS ➢ Focal Healthcare ➢ BK Medical
Holding Company, Inc. ➢ Exact Imaging ➢ Biobot Surgical
Pte Ltd. ➢ UC-Care Medical
Systems Ltd Fusion Biopsy Market Competitive Landscape Analysis The
fusion biopsy market is becoming increasingly competitive as leading medical
imaging, robotics, and AI-driven diagnostic companies continue investing in
precision oncology technologies. Market participants are focusing on improving
lesion targeting accuracy, workflow efficiency, procedural automation, and
real-time imaging integration to strengthen their competitive positioning. Companies
such as Philips and ESAOTE are leveraging their established imaging ecosystems
to expand advanced MRI-ultrasound fusion capabilities across hospital networks
and cancer diagnostic centers. KOELIS continues to strengthen its market
presence through AI-powered 3D prostate mapping and workflow optimization
technologies designed to improve targeting precision during biopsy procedures. Emerging
innovators including Biobot Surgical and Exact Imaging are accelerating
advancements in robotic-assisted targeting systems and high-resolution
micro-ultrasound imaging platforms. Meanwhile, several companies are
increasingly investing in deformable image fusion software, AI-assisted lesion
segmentation, and transperineal biopsy solutions to address growing demand for
minimally invasive and highly accurate diagnostic procedures. Strategic
collaborations between imaging technology providers, healthcare institutions,
and AI software developers are also expected to accelerate innovation across
next-generation fusion biopsy platforms globally. Key
Emerging Innovation in the Fusion Biopsy Market Innovation Focus Area Impact AI-Driven Auto-Segmentation Diagnostics Accuracy Automated 3D MRI/US lesion segmentation reduces
procedural time, operator dependency, and variability in tumor targeting. Robotic-Assisted Targeting Procedural Precision Robotic systems provide superior needle positioning
flexibility and stability for precise, complex targeting. Transperineal Fusion Patient Safety Shifting toward transperineal access with
specialized grids lowers infection rates compared to transrectal methods. Real-time 3D Deformable Fusion Image Guidance Advanced software accounts for prostate shape
changes during needle insertion, mapping images in real-time. AI-Assisted Biparametric MRI Decision Support AI analyzes bpMRI to identify suspicious areas,
reducing unnecessary biopsies by improving PI-RADS scoring accuracy.
You can place an order or ask any questions, please feel free
to contact at sales@precedenceresearch.com | +1 804
441 9344 Major
Shifts in the Fusion Biopsy Market 🔸In February 2025, Hologic, Inc. announced that its
Affirm® Contrast Biopsy Software received CE marking. The software, which
enhances diagnostic capabilities and streamlines biopsy procedures, was
previously cleared by the FDA in October 2020. Designed to integrate with Selenia®
Dimensions® and 3Dimensions™ systems, it helps clinicians target tissue samples
in lesions identified through contrast-enhanced mammography, ensuring
compassionate patient care. (Source: https://www.nasdaq.com/press-release/hologic-obtains-ce-mark-affirmr-contrast-biopsy-software-2025-02-20) 🔸In January 2025, Biobot Surgical announced that its
Mona Lisa 2.0 platform, for robotic-assisted biopsy and tissue ablation in
prostate cancer, received European CE certification. Already approved in the
US, Australia, and Singapore, this platform features advanced positioning for
improved precision and real-time adjustments during procedures. Albert Lee, CEO
of Biobot Surgical, highlighted its potential to enhance prostate care across
Europe. (Source: https://www.urologytimes.com/view/eu-approves-mona-lisa-2-0-robotic-system-for-prostate-cancer) Fusion
Biopsy Market Segmentation By
Product Type 🔸Equipment (Systems) →
MRI–Ultrasound Fusion Platforms → Fusion-Enabled
Ultrasound Systems → Interventional
MRI Systems 🔸Software → Image
Registration and Fusion Software → AI-Driven
Lesion Mapping and Segmentation Tools → 3D
Visualization and Navigation Software → Post-Procedure
Reporting and Analytics Platforms 🔸Consumables and Accessories → Biopsy
Needles and Guidance Kits → Needle
Guides and Templates (Grid-based / Freehand) → Fiducial
Markers and Target Sensors → Disposable
Probes and Sheaths By Biopsy
Route 🔸Transrectal (TRUS–MRI Fusion) 🔸Transperineal (TP–MRI Fusion) By Fusion
Technique 🔸Software-Based Fusion 🔸Cognitive Fusion (Visual / Manual
Estimation) 🔸In-Bore MRI Fusion (Direct MRI-Guided
Biopsy) By
Application 🔸Prostate Cancer Diagnosis (Primary) 🔸Renal (Kidney) Lesion Evaluation 🔸Liver Tumor Targeting 🔸Active Surveillance Monitoring 🔸Focal Therapy Planning By
End-User 🔸Hospitals and Tertiary Care Centers 🔸Specialized Oncology Clinics 🔸Diagnostic Imaging Centers 🔸Ambulatory Surgical Centers (ASCs) 🔸Academic and Research Institutions By Distribution
Channel 🔸Direct Sales (Institutional
Procurement) 🔸Indirect Sales (Medical Device
Distributors) By Region 🔸North
America 🔸Asia Pacific 🔸Europe 🔸Latin America 🔸Middle East and Africa Thank you for reading. You can also
get individual chapter-wise sections or region-wise report versions, such as North
America, Europe, or Asia Pacific. Immediate Delivery Available | Buy This Premium Research
Report@ https://www.precedenceresearch.com/checkout/5350 Research Methodology 🔹Primary interviews with industry
experts 🔹Secondary research from company
reports, regulatory databases, journals 🔹Market forecasting using top-down and
bottom-up approaches 🔹Validation through triangulation
models About Us Precedence
Research is a global market intelligence and consulting powerhouse, dedicated
to unlocking deep strategic insights that drive innovation and transformation.
With a laser focus on the dynamic world of
life sciences,
we specialize in decoding the complexities of cell and
gene therapy,
drug development, and oncology markets, helping our clients stay ahead in
some of the most cutting-edge and high-stakes domains in healthcare. Our
expertise spans across the biotech and pharmaceutical ecosystem, serving
innovators, investors, and institutions that are redefining what’s possible in regenerative medicine, cancer care, precision
therapeutics, and beyond. Web: https://www.precedenceresearch.com Our
Trusted Data Partners: Towards Healthcare | Nova One Advisor | Market Stats Insight Get
Recent News: https://www.precedenceresearch.com/news For the
Latest Update Follow Us: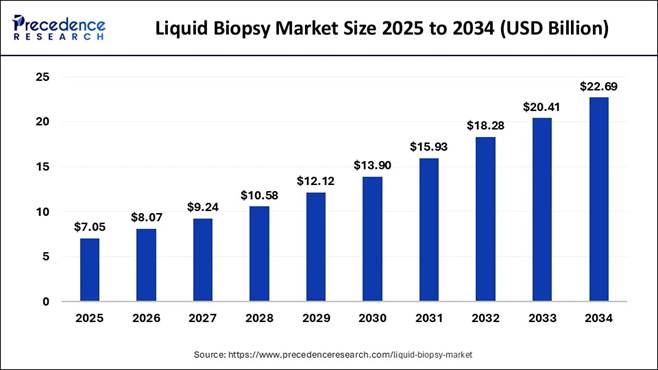
The Complete Market Intelligence Report is Available Now 📥 Get Instant Access to Sample Pages 👉 https://www.precedenceresearch.com/sample/5350
➡️ U.S. Liquid Biopsy Market: Explore how non-invasive
cancer diagnostics are transforming early detection and precision medicine in
the United States




