New York
Quotient Therapeutics’ platform targets somatic mutations, which the startup contends can help identify a broader scope of genes potentially associated with disease phenotypes. Wednesday’s agreement is part of an existing partnership between Pfizer and Flagship Pioneering.
The company joins Eli Lilly in offering its own digital platform to more directly connect to patients, allowing them easier access to healthcare providers and prescription drugs.
With the FDA’s rejection of Ordspono in March, Monday’s green light from the European Commission marks the first approval worldwide and the first regulatory victory for Regeneron’s bispecific antibody platform.
The regulator’s Complete Response Letter flagged problems at a third-party fill/finish site, which Regeneron says it has already addressed. The manufacturing facility is currently awaiting reinspection.
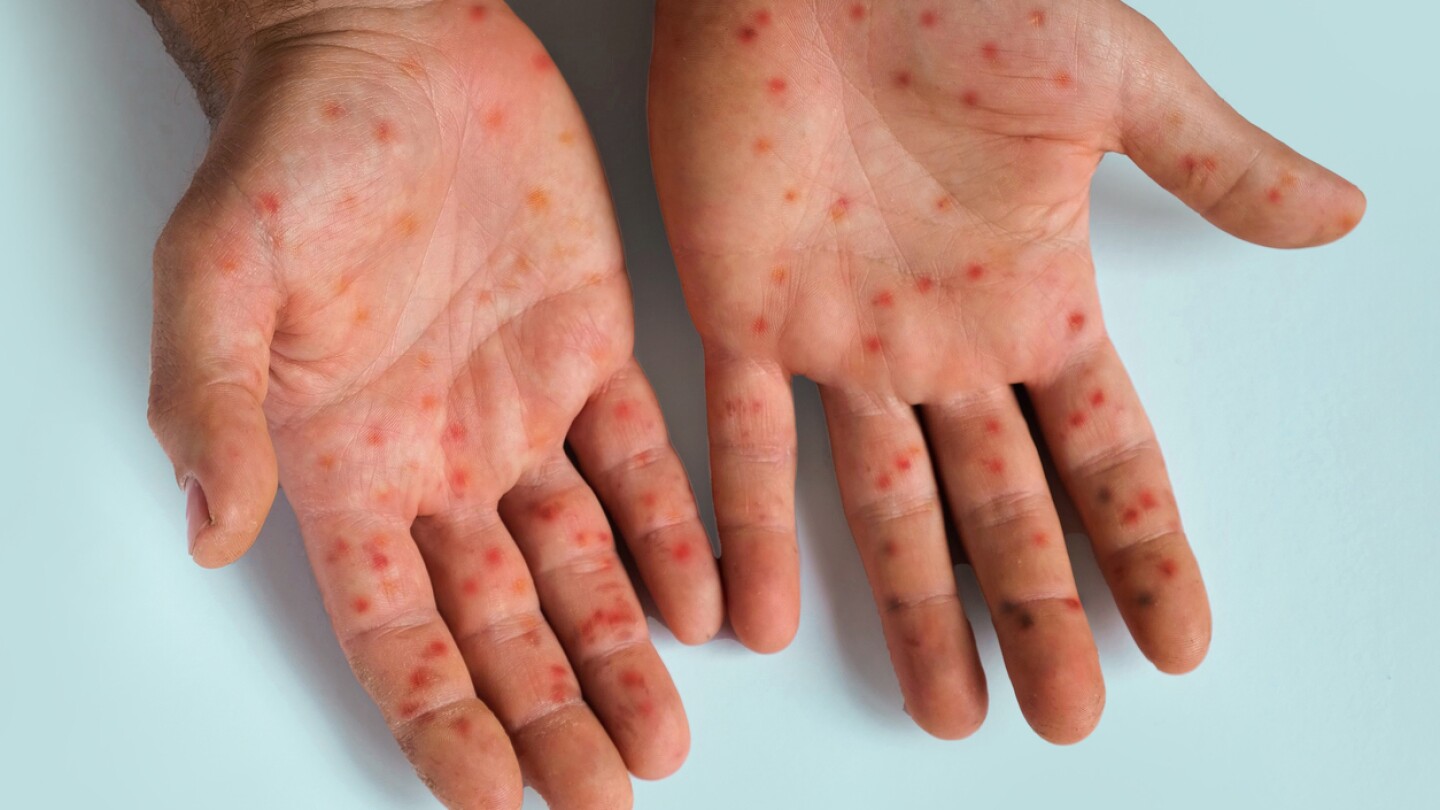
SIGA Technologies’ TPOXX did not outperform placebo at resolving lesions in patients with clade I mpox, the new strain that has spread through parts of Africa and is reaching beyond the continent.
Psychedelic drug developers are undeterred by the FDA’s Complete Response Letter for the company’s MDMA therapy for PTSD, and experts expect Lykos will ultimately obtain approval.
Regeneron, Akouos and Mass Eye and Ear are testing therapies that can reverse genetic protein deficiency to restore hearing, with promising early results.
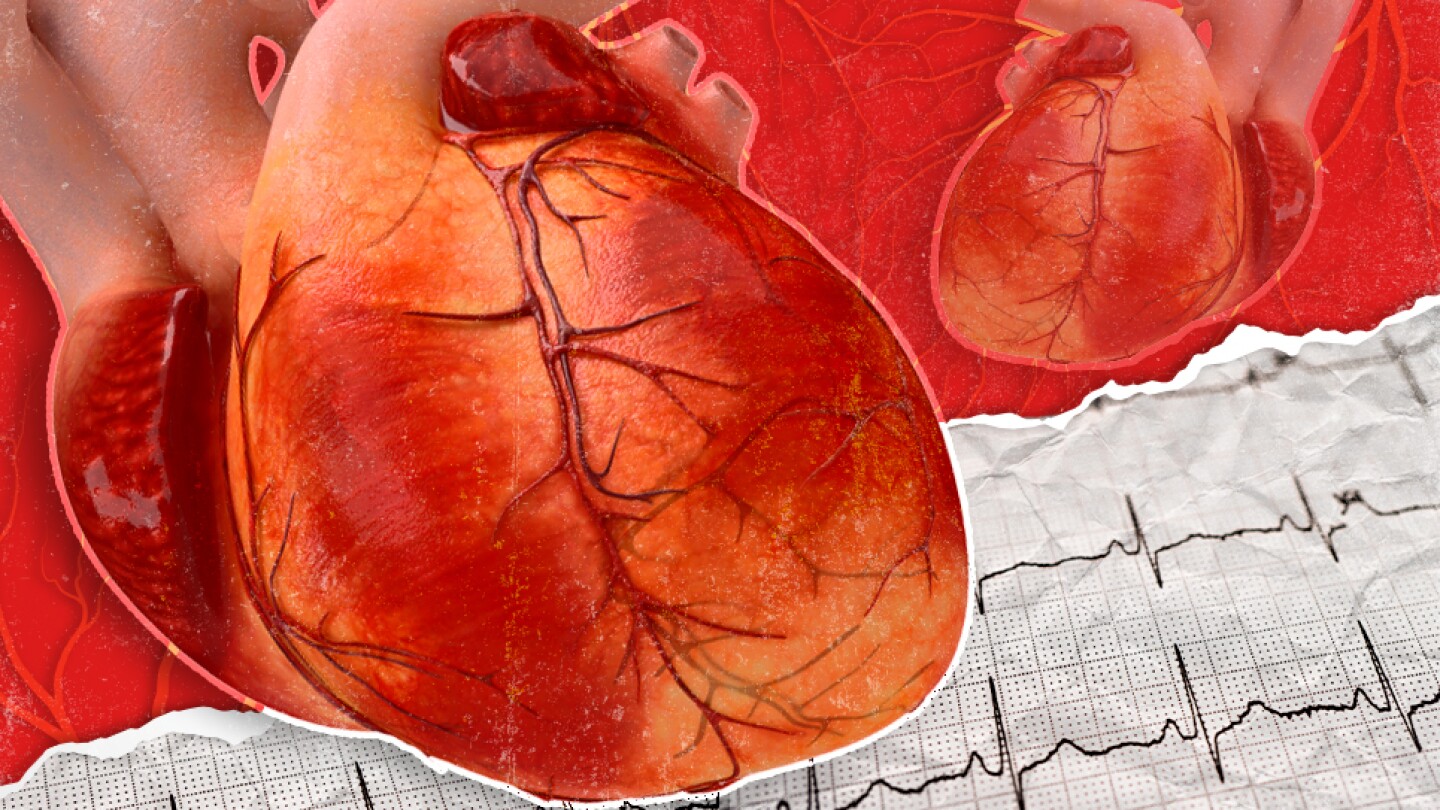
The entry of new players and new approaches into the ATTR-CM space could help bring down the cost of treatment, experts say.
Pfizer on Monday reported a “strong neutralizing response” against both subtypes of respiratory syncytial virus across all cohorts and age groups, according to topline data.
In the next two weeks, the FDA will hand down its verdicts for three drug applications, including ones for multiple myeloma and hypoparathyroidism therapies.
PRESS RELEASES