TVAX Biomedical (TVAX), is a cancer immunotherapy company that is moving into its pivotal glioblastoma study for FDA regulatory approval. TVAX Immunotherapy® could fundamentally change the way cancer is treated as T cells can kill all cancer cells, including cancer stem cells, without the toxicity of current treatments. With early
|
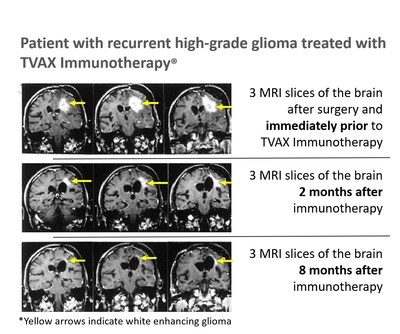
OLATHE, Kan., Dec. 9, 2020 /PRNewswire/ -- TVAX Biomedical (TVAX), is a cancer immunotherapy company that is moving into its pivotal glioblastoma study for FDA regulatory approval. TVAX Immunotherapy® could fundamentally change the way cancer is treated as T cells can kill all cancer cells, including cancer stem cells, without the toxicity of current treatments. With early promising outcomes, TVAX received Fast Track Designation and Orphan Product Designation from the FDA. Glioblastoma is an incurable brain cancer affecting >12,000 people annually in the US. This form of brain cancer killed Beau Biden, the son of President-elect Biden, and Senators John McCain and Ted Kennedy and countless others. TVAX is working to change that. TVAX Immunotherapy is a cutting-edge approach to cellular therapy. The individualized therapy first helps the patient develop immunity against their cancer and the unique "neoantigens" that cancer cells produce. Next, T cells are removed from patient's blood and activated in TVAX's specialized manufacturing facility. Patients are then treated with billions of their own "killer" T cells that attack their cancer. Dr. Wayne Carter, TVAX Biomedical's CEO, explains it like this, "TVAX's technology uses 'killer' T cells that function like billions of smart bombs hunting and destroying cancer cells throughout the body. TVAX Immunotherapy has very few side effects because 'killer' T cells kill cancer cells, not normal cells." This therapy has shown promising results for four-legged friends too. The ability to treat multiple types of cancer prompted TVAX to spin-off its animal health division into Elias Animal Health, an affiliate pursuing similar treatments in dogs. Elias is using the technology to treat osteosarcoma, an incurable bone cancer and is currently enrolling dogs for their registration study with the USDA. TVAX Immunotherapy has been administered to more than 120 patients and has demonstrated efficacy against several cancers, producing numerous long-lasting complete and partial responses with none of the "off-target" short and long-term toxicity seen with CAR-T cell therapy, checkpoint inhibitors, and chemotherapies.
TVAX worked closely with the FDA to plan its critical glioblastoma registration study. Fast Track Designation affords many advantages, including a pathway to accelerated marketing approval. TVAX Immunotherapy has dramatically lower manufacturing costs compared to other cellular immunotherapies such as CAR-T therapies and manufacturing can be easily automated to facilitate large-scale commercialization. About TVAX Biomedical TVAX Biomedical is a clinical stage development company advancing its proprietary personalized T cell-based cancer immunotherapy. The company's lead candidate is supported by positive Phase 2 clinical data, as well as extensive preclinical and Phase 1 safety studies. TVAX plans to initiate a registration study in newly diagnosed glioblastoma patients in 2021. The US brain cancer market is $1.5 billion with many follow-on market opportunities in other types of cancer. As part of its ongoing strategy, the company is seeking investors and corporate alliances to support the development.
SOURCE TVAX Biomedical |