- Crinecerfont Demonstrates Meaningful Reductions in Key Disease Biomarkers After 14 Days of Treatment - Initiation of Global Registrational Study of Crinecerfont in Adults Planned for Second Half 2020 [08-June-2020] SAN DIEGO , June 8, 2020 /PRNewswire/ -- Neurocrine Biosciences, Inc. (Nasdaq: NBIX) today announced positive data from its completed op
|
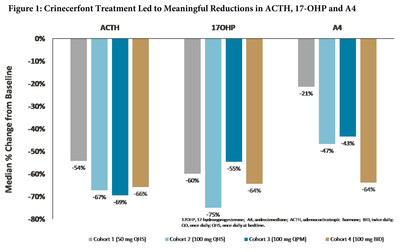
SAN DIEGO, June 8, 2020 /PRNewswire/ -- Neurocrine Biosciences, Inc.. (Nasdaq: NBIX) today announced positive data from its completed open-label, multiple-dose, dose-finding, Phase II clinical study of crinecerfont (NBI-74788), demonstrating meaningful reductions in all three key disease hormone markers in adult patients with classic congenital adrenal hyperplasia (CAH), a genetic disorder affecting the adrenal glands. Crinecerfont treatment produced meaningful reductions in elevated adrenocorticotropic hormone (ACTH) and 17-hydroxyprogesterone (17-OHP) levels (by 54% to 75%) at all doses studied, together with a dose-related decrease in androstenedione (A4) levels, ranging from 21% to 64% (Figure 1). At the highest dose of crinecerfont (100 mg twice daily), 75% of patients showed a response of at least 50% reduction from baseline for each of the three hormone markers at day 14 (Table 1). Treatment with crinecerfont was well tolerated with a favorable safety profile with no related serious adverse events reported. Adverse events reported in two or more participants included headache, upper respiratory tract infection, fatigue, contusion, insomnia and nausea. The full data set from the Phase II study assessing the safety, tolerability, pharmacokinetics and pharmacodynamics of crinecerfont, an investigational, oral, non-steroidal corticotropin-releasing factor type 1 (CRF1) receptor antagonist, is available as part of a recorded presentation at the Endocrine Society’s ENDO Online 2020 meeting. Table 1: Number and Percentage of Participants in Each Cohort Demonstrating a ≥ 50% Reduction from Baseline in ACTH, 17-OHP and Androstenedione (A4)
"There continues to be a need for effective treatment options that are well tolerated in patients with classic CAH. Patients with this genetic disorder require glucocorticoid replacement therapy, but often at high doses to manage their excessive adrenal androgen production. At the same time, side effects from chronic treatment with supraphysiologic amounts of glucocorticoids can cause serious long-term health consequences including bone loss and metabolic dysfunction," said Richard Auchus, M.D., Ph.D., the study's lead investigator and Professor of Internal Medicine, Division of Metabolism, Endocrinology & Diabetes at Michigan Medicine. "It is encouraging to see that crinecerfont, a non-steroidal therapy, provided meaningful reductions in three key disease biomarkers in patients with classic CAH. These data suggest that crinecerfont has the potential to improve CAH symptoms and to reduce the burden of daily glucocorticoid exposure for these patients. Hopefully, this approach might provide a new treatment option to better manage the androgen excess of classic CAH while mitigating the adverse consequences of current treatment schemes." Neurocrine Biosciences plans to initiate a single, global registrational study of crinecerfont in adult patients with classic CAH in the second half of 2020. Classic CAH is a genetic disorder, in which an enzyme deficiency alters the production of adrenal steroids. Because of this deficiency, the adrenal glands fail to produce enough cortisol and, sometimes, aldosterone, resulting in a potentially life-threatening condition. "We are pleased that crinecerfont was effective in producing a meaningful, dose-related reduction of adrenal androgens and other key biomarkers of disease in patients with classic CAH and was well tolerated in this study," said Eiry W. Roberts, M.D., Chief Medical Officer at Neurocrine Biosciences. "In addition, data from the study was successful in identifying an effective dosing regimen for further evaluation in a single, global registration study in adults with classic CAH. We hope to demonstrate that crinecerfont is a valuable, non-steroidal, treatment option for patients to manage the burdensome symptoms of classic CAH, while also reducing the need for chronic supraphysiologic dosing with glucocorticoids." Crinecerfont Phase II Study Design About Classic Congenital Adrenal Hyperplasia (CAH) Corticosteroids, the current standard of care, are used both to correct the endogenous cortisol deficiency and to reduce the high ACTH levels and androgen excess. However, the dose and duration of glucocorticoids required to suppress ACTH are often well above the normal physiological level of cortisol, which can result in serious complications typical of iatrogenic Cushing's syndrome, including metabolic issues, bone loss, growth impairment, and infection risk. Classic CAH is a disease that affects approximately 20,000 to 30,000 people in the United States and approximately 50,000 people in Europe. About Crinecerfont Neurocrine Biosciences plans to initiate a single, global registrational study of crinecerfont in adult patients with classic CAH in the second half of 2020 and recently restarted enrollment for the Phase IIa pediatric study in adolescents with classic CAH. About Neurocrine Biosciences Forward-Looking Statements
SOURCE Neurocrine Biosciences, Inc. |
||||||||||||||||||||||
Company Codes: NASDAQ-NMS:NBIX |