Akebia Therapeutics, Inc. (Nasdaq: AKBA) today announced that it and Keryx Biopharmaceuticals, Inc. (Nasdaq: KERX) filed a definitive joint proxy statement with the U.S. Securities and Exchange Commission (“SEC”)
CAMBRIDGE, Mass.--(BUSINESS WIRE)-- Akebia Therapeutics, Inc. (Nasdaq: AKBA) today announced that it and Keryx Biopharmaceuticals Inc. (Nasdaq: KERX) filed a definitive joint proxy statement with the U.S. Securities and Exchange Commission (“SEC”) in connection with their respective special meetings of shareholders that have been called to approve shareholder proposals relating to the previously announced proposed merger of Akebia and Keryx. Akebia is mailing a letter together with its proxy materials to Akebia shareholders in connection with the special meeting of Akebia shareholders (the “Special Meeting”).
This press release features multimedia. View the full release here: https://www.businesswire.com/news/home/20181030006158/en/
The Special Meeting will be held at 11:00 a.m. Eastern Time on December 11, 2018, at the offices of Latham & Watkins LLP, which are located at 200 Clarendon Street, Boston, MA 02116. Holders of record of Akebia common stock as of 5:00 p.m. Eastern Time on October 22, 2018, will be entitled to vote at the Special Meeting.
The Akebia Board of Directors unanimously recommends that Akebia shareholders vote “FOR” the proposals relating to the proposed merger with Keryx.
In connection with the filing of the definitive proxy statement, Akebia will mail the following letter to shareholders:
Dear Fellow Akebia Shareholder:
As Chairperson of the Akebia Board of Directors, I am writing to encourage you to vote in the upcoming Special Meeting of Akebia shareholders to approve the shareholder proposals relating to the combination of Akebia with Keryx Biopharmaceuticals. The Akebia Board of Directors has been intensely focused on driving value for our shareholders. The Board unanimously believes this combination will provide substantial strategic, financial and operational benefits to Akebia shareholders.
We believe that this transaction will lower Akebia’s risk profile by providing it with a broader range of growth opportunities to enhance our market position, capital resources and operational efficiency, and increase the value of your investment in the near-term, mid-term and long-term.
In the near-term, Akebia shareholders will gain access to Auryxia® (ferric citrate), a U.S. Food and Drug Administration (FDA)-approved medicine to treat dialysis dependent chronic kidney disease (CKD) patients with hyperphosphatemia and non-dialysis dependent CKD patients with iron deficiency anemia. In the mid-term, Auryxia’s potential growth is expected to generate the cash to fund pro forma operations and cover the majority of Akebia’s capital needs beginning in 2020. In the long-term, Akebia shareholders remain positioned to participate in the potential benefits from vadadustat, an innovative Phase 3 product candidate with the potential to compete in a complementary multibillion-dollar market, subject to successful completion of its development program and regulatory approval, with an established commercial organization.
The Akebia Board of Directors recommends that all shareholders vote “FOR” the proposals relating to the proposed merger for the following reasons:
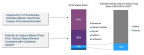
FULLY INTEGRATED RENAL COMPANY WITH A COMPLEMENTARY PORTFOLIO
- The merger will create a fully integrated renal company with a complementary portfolio comprising Keryx’s FDA-approved Auryxia and Akebia’s product candidate, vadadustat, which is in Phase 3 clinical trials
- Auryxia has substantial revenue growth potential
- In the second quarter of 2018, quarterly prescriptions for Auryxia doubled compared to the same quarter last year
- Auryxia’s second quarter 2018 market share was approximately 6% and has the potential for significant market share gains over the next several years
See image one.
- Physicians express favorable perception of Auryxia in iron deficiency anemia (IDA)
- Majority of physicians surveyed recognize benefits of Auryxia’s profile5
- Majority of surveyed nephrologists report satisfaction with Auryxia6
COMBINATION EXPECTED TO ENHANCE CAPITAL RESOURCES AND CASH FLOW
- The combined company will have a strong cash position with pro forma cash on hand as of June 30, 2018, of $452 million (unaudited), enabling further development of vadadustat while reducing the need for future share dilution
- Keryx’s ongoing revenue streams and potential for growth are expected to enhance Akebia’s profitability and cash flow as Auryxia’s revenues have the potential to continue to increase
See image two.
EXPERIENCED RENAL MANAGEMENT TEAM AND SUBSTANTIAL ORGANIZATIONAL SYNERGIES EXPECTED TO CREATE COST SAVINGS AND LOWER RISK OF COMMERCIAL LAUNCH
- Together, Akebia and Keryx plan to leverage Keryx’s existing renal commercial infrastructure, eliminating the need for Akebia as a standalone to build its own salesforce ahead of launch
- The combined company will be led by a management team with a long track record of success developing, launching and commercializing products for patients with kidney disease, including Chief Executive Officer, John P. Butler, who led the renal business at Genzyme Corporation, growing the business from $150 million to over $1 billion in revenue
- We plan to leverage our leadership’s extensive expertise in the commercial renal market with the goal of maximizing sales of Auryxia while driving launch momentum for vadadustat in the United States, subject to its regulatory approval
- We estimate potential cost savings of greater than $250 million to be realized within five years following closing
VOTE FOR ENHANCED VALUE AND ACCELERATED GROWTH OPPORTUNITY
VOTE “FOR” THE PROPOSALS RELATING TO AKEBIA – KERYX MERGER TODAY
The Akebia Board and management team have been – and continue to be – highly focused on driving value for all our shareholders. We undertook a robust strategic evaluation prior to entering into the merger agreement with Keryx, and based on that evaluation, coupled with the fairness opinions received from independent financial advisors, we believe the merger is the optimal path for Akebia to build value for Akebia shareholders over time.
Accordingly, we recommend that shareholders vote today “FOR” the Akebia proposals set forth in the definitive proxy statement, including “FOR” the proposal to issue shares of Akebia common stock to Keryx shareholders in relation to the proposed merger.
Your vote is extremely important, no matter how many shares you own. Please take a moment to vote “FOR” the proposals set forth on the enclosed proxy card today – by internet, telephone toll-free or by signing, dating and returning the enclosed proxy card in the postage-paid envelope provided.
If you have any questions or need assistance voting your shares, please contact MacKenzie Partners, Inc., our proxy solicitor, by calling toll-free at (800) 322-2885 (from the U.S. and Canada) or (212) 929-5500 (call collect from other locations) or via email at proxy@mackenziepartners.com.
On behalf of the Akebia Board of Directors, thank you for your continued support of Akebia.
Sincerely,
Muneer A. Satter
Chairperson of the Akebia Board of Directors
About Akebia
Akebia Therapeutics, Inc. is a biopharmaceutical company headquartered in Cambridge, Massachusetts, focused on delivering innovative therapies to patients with kidney disease through hypoxia-inducible factor biology. For more information, please visit our website at www.akebia.com, which does not form a part of this release.
Additional Information and Where to Find It
In connection with the proposed merger, Akebia has filed with the SEC a Registration Statement on Form S-4, which, as amended, includes a final prospectus with respect to the shares of Akebia’s common stock to be issued in the proposed merger and a definitive joint proxy statement of Keryx and Akebia with respect to the proposed merger. The Registration Statement was declared effective by the SEC on October 30, 2018 and the definitive joint proxy statement was mailed or otherwise made available to Keryx’s and Akebia’s respective stockholders on or about October 31, 2018. BEFORE MAKING ANY VOTING DECISION, AKEBIA’S AND KERYX’S RESPECTIVE SHAREHOLDERS ARE URGED TO READ THE JOINT PROXY STATEMENT/PROSPECTUS IN ITS ENTIRETY AND ANY OTHER DOCUMENTS FILED BY EACH OF AKEBIA AND KERYX WITH THE SEC IN CONNECTION WITH THE PROPOSED MERGER OR INCORPORATED BY REFERENCE THEREIN BECAUSE THEY WILL CONTAIN IMPORTANT INFORMATION ABOUT THE PROPOSED TRANSACTION AND THE PARTIES TO THE PROPOSED TRANSACTION. Investors and shareholders can obtain a free copy of the joint proxy statement/prospectus and other documents containing important information about Akebia and Keryx filed with or furnished to the SEC, through the website maintained by the SEC at www.sec.gov. Akebia and Keryx make available free of charge at www.akebia.com (in the “Investors” section) and www.keryx.com, respectively (in the “Investors & Media” section), copies of materials they file with, or furnish to, the SEC.
Participants in the Solicitation
Akebia, Keryx and their respective directors, executive officers and certain employees and other persons may be deemed to be participants in the solicitation of proxies from the shareholders of Akebia and Keryx in connection with the proposed merger. Security holders may obtain information regarding the names, affiliations and interests of Akebia’s directors and officers in Akebia’s Annual Report on Form 10-K for the fiscal year ended December 31, 2017, which was filed with the SEC on March 12, 2018 and its definitive proxy statement for the 2018 annual meeting of shareholders, which was filed with the SEC on April 30, 2018. Security holders may obtain information regarding the names, affiliations and interests of Keryx’s directors and officers in Keryx’s Annual Report on Form 10-K for the fiscal year ended December 31, 2017, which was filed with the SEC on February 21, 2018, and the Amendment No. 1 on Form 10-K/A, which was filed with the SEC on April 30, 2018, and its definitive proxy statement for the 2018 annual meeting of shareholders, which was filed with the SEC on May 31, 2018. To the extent the holdings of Akebia’s securities by Akebia’s directors and executive officers or the holdings of Keryx securities by Keryx’s directors and executive officers have changed since the amounts set forth in Akebia’s or Keryx’s respective proxy statement for its 2018 annual meeting of shareholders, such changes have been or will be reflected on Statements of Change in Ownership on Form 4 filed with the SEC. Additional information regarding the interests of such individuals in the proposed merger are included in the joint proxy statement/prospectus relating to the proposed merger that was filed with the SEC. These documents may be obtained free of charge from the SEC’s website at www.sec.gov, Akebia’s website at www.akebia.com and Keryx’s website at www.keryx.com.
This document does not constitute a solicitation of proxy, an offer to purchase or a solicitation of an offer to sell any securities.
Forward Looking Statements
These materials contain forward-looking statements within the meaning of the federal securities law. Such statements are based upon current plans, estimates and expectations that are subject to various risks and uncertainties. The inclusion of forward-looking statements should not be regarded as a representation that such plans, estimates and expectations will be achieved. Words such as “believe,” “build,” “create,” “drive,” “enhance,” “estimate,” “expect,” “goal,” “leverage,” “may,” “maximize,” “opportunity,” “optimal,” “plan,” “position,” “potential” “will,” “would,” and words and terms of similar substance used in connection with any discussion of future plans, actions or events identify forward-looking statements. All statements, other than historical facts, including statements regarding the ability of the parties to complete the merger considering the various closing conditions; the consummation of the merger; the potential benefits of the merger; creating shareholder value; the growth potential of Auryxia and the market potential of Auryxia and vadadustat are forward looking statements. Important factors that could cause actual results to differ materially from Akebia’s and Keryx’s plans, estimates or expectations could include, but are not limited to: (i) Akebia or Keryx may be unable to obtain stockholder approval as required for the merger; (ii) conditions to the closing of the merger may not be satisfied; (iii) the merger may involve unexpected costs, liabilities or delays; (iv) the effect of the announcement of the merger on the ability of Akebia or Keryx to retain and hire key personnel and maintain relationships with customers, suppliers and others with whom Akebia or Keryx does business, or on Akebia’s or Keryx’s operating results and business generally; (v) Akebia’s or Keryx’s respective businesses may suffer as a result of uncertainty surrounding the merger and disruption of management’s attention due to the merger; (vi) the outcome of any legal proceedings related to the merger; (vii) Akebia or Keryx may be adversely affected by other economic, business, and/or competitive factors; (viii) the occurrence of any event, change or other circumstances that could give rise to the termination of the merger agreement; (ix) risks that the merger disrupts current plans and operations and the potential difficulties in employee retention as a result of the merger; (x) the risk that Akebia or Keryx may be unable to obtain governmental and regulatory approvals required for the transaction, or that required governmental and regulatory approvals may delay the transaction or result in the imposition of conditions that could reduce the anticipated benefits from the proposed transaction or cause the parties to abandon the proposed transaction; (xi) risks that the anticipated benefits of the merger or other commercial opportunities may otherwise not be fully realized or may take longer to realize than expected; (xii) the impact of legislative, regulatory, competitive and technological changes, including the recent changes to reimbursement coverage for Auryxia that could have a material adverse effect on Auryxia sales and profitability; (xiii) expectations for future clinical trials, the timing and potential outcomes of clinical studies and interactions with regulatory authorities; and (xiv) other risks to the consummation of the merger, including the risk that the merger will not be consummated within the expected time period or at all. Additional factors that may affect the future results of Akebia and Keryx are set forth in their respective filings with the SEC, including each of Akebia’s and Keryx’s most recently filed Annual Report on Form 10-K, subsequent Quarterly Reports on Form 10-Q, Current Reports on Form 8-K, in the definitive joint proxy statement/prospectus filed by Akebia and Keryx and other filings with the SEC, which are available on the SEC’s website at www.sec.gov. See in particular “Risk Factors” in the definitive joint proxy statement/prospectus filed by Akebia and Keryx, Item 1A of Akebia’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2018 under the heading “Risk Factors,” and Item 1A of Keryx’s Quarterly Report on Form 10-Q for the quarter ended June 30, 2018 under the heading “Risk Factors.” The risks and uncertainties described above and in the definitive joint proxy statement/prospectus filed by Akebia and Keryx, Akebia’s most recent Quarterly Report on Form 10-Q and Keryx’s most recent Quarterly Report on Form 10-Q are not exclusive and further information concerning Akebia and Keryx and their respective businesses, including factors that potentially could materially affect their respective businesses, financial condition or operating results, may emerge from time to time. Readers are urged to consider these factors carefully in evaluating these forward-looking statements, and not to place undue reliance on any forward-looking statements. Readers should also carefully review the risk factors described in other documents that Akebia and Keryx file from time to time with the SEC. The forward-looking statements in these materials speak only as of the date of these materials. Except as required by law, Akebia and Keryx assume no obligation to update or revise these forward-looking statements for any reason, even if new information becomes available in the future.
-------------------------------------------------------------------------
| Footnotes: |
| 1. Keryx consolidated data based on data received from IMS and specialty pharmacies (Fresenius Rx, DaVita Rx) |
| 2. Definitive Proxy Statement/Prospectus filed by Akebia Therapeutics, Inc. with the U.S. Securities and Exchange Commission on October 30, 2018 (see “The Merger—Certain Akebia Management Unaudited Prospective Financial Information – Akebia Management Keryx Projections”). This estimate of peak sales is unaudited and was based upon Akebia assumptions made in preparation for the June 28, 2018, merger announcement, including upon publicly filed financial information of Keryx, certain financial information provided to Akebia management by Keryx, and certain assumptions made by the Akebia management, including estimates of revenue growth for U.S. sales of Auryxia and associated operational costs, and has not been updated since that time. Furthermore, this estimate of peak sales was not adjusted for a number of critical risks, including the recent changes to reimbursement coverage for Auryxia that could have a material adverse effect on Auryxia sales and profitability. See the Forward-Looking Statements section herein for additional information regarding risks. |
| 3. Akebia management internal estimates based on market research. This Auryxia market share estimate formed the basis of certain information included in the Definitive Proxy Statement/Prospectus filed by Akebia Therapeutics, Inc. with the U.S. Securities and Exchange Commission on October 30, 2018 (see “The Merger—Certain Akebia Management Unaudited Prospective Financial Information – Akebia Management Keryx Projections”) and was based upon Akebia assumptions made in preparation for the June 28, 2018, merger announcement, including upon publicly filed financial information of Keryx, certain financial information provided to Akebia management by Keryx, and certain assumptions made by the Akebia management, including estimates of revenue growth for U.S. sales of Auryxia and associated operational costs, and has not been updated since that time. Furthermore, this market share estimate was not adjusted for a number of critical risks, including the recent changes to reimbursement coverage for Auryxia that could have a material adverse effect on Auryxia sales and profitability. See the Forward-Looking Statements section herein for additional information regarding risks. |
| 4. Prevention and Treatment of Chronic Kidney Disease-Mineral and Bone Disorder (CKD_MBD); Vol. 7, Issue 1. July 2017 |
| 5. Reason Research Q3 Auryxia ATU |
| 6. Spherix Global Anemia 1Q Pulse (2018); aided awareness data |
| 7. Definitive Proxy Statement/Prospectus filed by Akebia Therapeutics, Inc. with the U.S. Securities and Exchange Commission on October 30, 2018 (see “The Merger—Certain Akebia Management Unaudited Prospective Financial Information”). These cash balance estimates are unaudited and were based upon Akebia assumptions made in preparation for the June 28, 2018, merger announcement, including assumptions related to timing for clinical trial completion and commercial launch, estimated operational costs, including R&D, manufacturing and general and administrative costs, and estimates of revenue growth for U.S. sales of Auryxia, and have not been updated since that time. Furthermore, these cash balance estimates are not adjusted for a number of critical risks, including the risks and probability of success of vadadustat, delays of any clinical trials or commercial launch, the financial implications of Akebia’s collaborations and other relationships with third parties, and the recent changes to reimbursement coverage for Auryxia that could have a material adverse effect on Auryxia sales and profitability. See the Forward-Looking Statements section herein for additional information regarding risks. |
View source version on businesswire.com: https://www.businesswire.com/news/home/20181030006158/en/
Contacts
Akebia Therapeutics
John Garabo, 617-844-6130
Director, Corporate Communications
jgarabo@akebia.com
Source: Akebia Therapeutics, Inc.