- MALDI Biotyper® (MBT): Launch of proprietary MBT Easy T® Kit for consistent sample transfer to MALDI target plates
- MALDI Biotyper®: Expansion of reference libraries to more than 5,300 species, spanning bacteria, yeasts, filamentous fungi, and mycobacteria
- MALDI Biotyper®: Early KOL access to a novel cloud‑based identification solution, enabling identification against several specialist, expert‑curated libraries
-
MALDI Biotyper®: Ongoing broad, multi‑year clinical and translational study programs advancing automation, potential future claims, and next‑generation analytics, including:
- IVDR-compliant clinical validation studies evaluating automated sample preparation workflows, covering both colony‑based cultures (MBT PrepMatic™) and positive blood cultures (MBT SepsiMatic®)
- U.S. clinical studies intended to support upcoming identification claims for mycobacteria and filamentous fungi, with FDA submission targeted for 2026
- Ongoing clinical evaluation of MBT FAST, a rapid phenotypic antimicrobial susceptibility testing approach, aiming to enable IVDR-compliant same‑day results from positive blood cultures and agar colonies for Enterobacteriaceae
- Exploratory, data‑driven research studies presented at ESCMID, including AI‑based antibiotic resistance prediction for Staphylococcus aureus, derived from large MALDI Biotyper spectral datasets
- IR Biotyper®: Enhanced outbreak detection with IR Tracker for surveillance of hospital‑acquired infections (HAIs) and new classifiers for Salmonella Typhi, STEC, and Shigella sp. detection
- MBioSEQ™ Ridom Typer: New software version, including TB‑Profiler antimicrobial resistance (AMR) determination, and GAMBIT bacterial species identification supporting MALDI / IR Biotyper®-triggered WGS reflex testing workflows, with result application performed under laboratory responsibility
- Introduction of Molzym as a part of Bruker Microbiology and Infection Diagnostics
MUNICH--(BUSINESS WIRE)--$BRKR #BRKR--At ESCMID Global 2026, Bruker announces important portfolio enhancements across microbial identification, outbreak analysis, and reflex next‑generation sequencing (NGS) typing. These latest developments underscore Bruker’s continued commitment to advancing integrated, high‑performance solutions that support laboratories in addressing evolving infectious disease challenges.
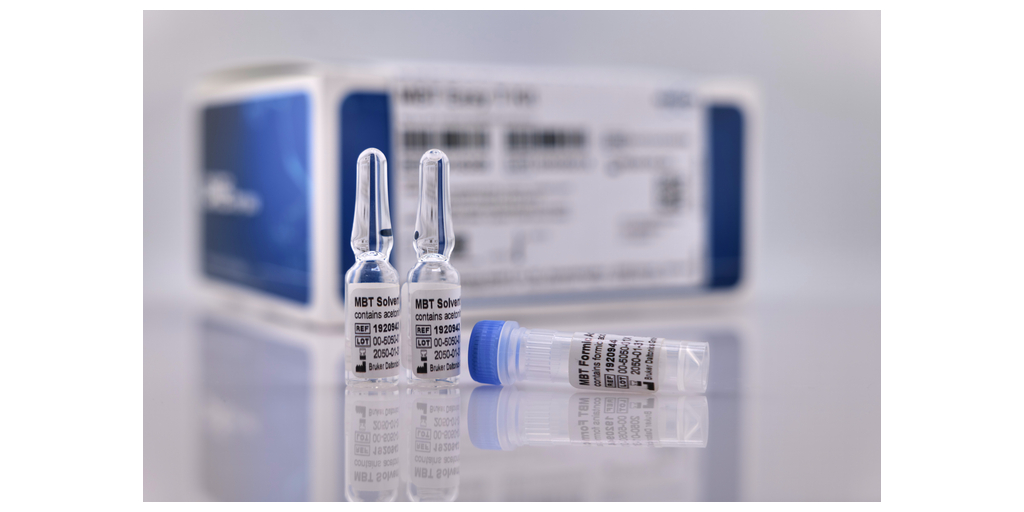


MALDI Biotyper® Workflow and Identification Enhancements
To further streamline routine MALDI‑TOF workflows, Bruker introduces the proprietary MBT Easy T® Kit (RUO and IVDR), a consumables kit designed to simplify and standardize sample transfer steps to MALDI target plates. The kit includes ready-to-use reagents and applicators, supporting up to 2,000 samples, while enabling room temperature storage and reducing chemical waste.
In parallel, Bruker expands its MALDI Biotyper® reference libraries to more than 5,300 species, including bacteria, yeasts, filamentous fungi, and mycobacteria (RUO). The latest library updates add more than 600 new species and significantly strengthen species coverage, particularly for the dedicated filamentous fungi MyT workflow which benefits from ~30% stronger species coverage. Corresponding IVDR‑registered library expansions are currently in progress.
To extend access to specialized identification content beyond local installations, Bruker is offering early access to a cloud‑based MALDI identification solution (RUO) for selected key opinion leaders and expert laboratories. This approach enables research use identification using specialist libraries curated by domain experts, within the MBT Compass HT environment. Initial expert libraries include yeast collections, anaerobic bacteria libraries developed by academic partners, and mosquito libraries supporting malaria vector monitoring and research. The cloud infrastructure allows visibility of library provenance and provides a foundation for future capabilities such as exclusive libraries, online system monitoring, and software updates.
Bruker continues to advance automation across sample preparation workflows with MBT PrepMatic™ and MBT SepsiMatic® systems, both currently under evaluation in IVDR-compliant clinical validation studies. MBT PrepMatic™ is designed to enable semi‑ to fully automated colony picking from Petri dish cultures and preparation of MALDI target plates. MBT SepsiMatic® is designed to support a fully automated workflow from positive blood culture bottles to prepared MALDI target plates, with the potential to produce purified samples containing viable cells for downstream analysis.
Advanced Clinical and Research Applications
Bruker is progressing multiple advanced applications for MALDI‑based microbiology. In the United States, clinical studies are underway to support upcoming claims for mycobacteria and filamentous fungi identification, with FDA submissions planned for 2026.
Clinical validation studies are underway on rapid phenotypic antimicrobial susceptibility testing (AST) using MALDI‑TOF MS, aimed at enabling same‑day results from positive blood cultures and agar colonies for Enterobacteriaceae. This approach explores the use of machine‑learning‑based growth assessment and aligns conceptually with CLSI‑ and EUCAST‑guided broth microdilution principles.
At ESCMID Global 2026, Bruker is also presenting poster data from exploratory research on AI‑driven antibiotic resistance prediction for Staphylococcus aureus, with AUROC >0.8, based on more than 2,600 MALDI Biotyper® spectra (RUO). Further studies with expanded scope are ongoing.
Hospital Hygiene and Outbreak Management
In the area of hospital hygiene and epidemiology, Bruker expands IR Biotyper® applications (GP) with IR Tracker for surveillance of hospital‑acquired infections (HAIs) and new classifiers for clinically and epidemiologically significant organisms. Newly supported targets include Salmonella Typhi, Shiga toxin‑producing E. coli (STEC), and Shigella species - pathogens associated with specific treatment requirements, high infectivity, and public health reporting obligations. These enhancements support rapid differentiation and classification to aid outbreak detection and infection control efforts.
Reflex NGS and Culture‑Independent Diagnostics
Bruker introduces a new software version of its MBioSEQ™ Ridom Typer (RUO), featuring short and long read whole genome sequencing (WGS) data analysis for research and expert laboratory applications. The software now includes TB-Profiler-based antimicrobial resistance (AMR) determination and GAMBIT bacterial species identification, supporting MALDI / IR Biotyper®-triggered WGS reflex workflows as part of laboratory‑defined analysis processes.
In addition, Bruker announces the integration of Molzym following its acquisition in late 2025, further strengthening the company’s Microbiology & Infection Diagnostics portfolio. Molzym brings expertise in culture‑independent pathogen diagnostics, including its unique and patented MolYsis™ host‑DNA depletion technology, which selectively removes host DNA to enable highly sensitive detection of microbial DNA in low‑biomass and blood samples for subsequent sequencing analysis.
“In routine MALDI‑TOF identification, having critical reagents readily available in a standardized format is essential,” said Kirsten Schönfeld, Diplom-Biologin, Institut Dr. Nowak GmbH, Germany. “The MBT Easy T® Kit provides pre‑filled, ready‑to‑use solvent ampoules, including formic acid, which will simplify logistics and daily laboratory operations. This helps maintain a robust and streamlined MALDI Biotyper workflow in a clinical setting.”
Dr. Wolfgang Pusch, President of Bruker’s Microbiology & Infection Diagnostics Division, added: “Feedback like this underscores why we continue to invest in practical innovations that strengthen everyday laboratory workflows. Across microbial identification and outbreak analysis, Bruker is pursuing a broad and sustained innovation initiative - significantly expanding our reference libraries, introducing new sample preparation options to enable the highest level of standardization, and advancing exploratory artificial‑intelligence‑based approaches for antibiotic resistance prediction from MALDI spectra. In parallel, we are driving the development of automation for sample preparation from both colonies and positive blood cultures, initiating clinical studies for MALDI Biotyper®‑based rapid antimicrobial susceptibility testing, and further expanding the analytical capabilities of the IR Biotyper®. At the same time, we continue to refine our NGS‑based MBioSEQ™ Ridom Typer , including ongoing, research‑use‑focused enhancements for tuberculosis antimicrobial resistance profiling.”
About Bruker Corporation – Leader of the Post-Genomic Era (Nasdaq: BRKR)
Bruker is enabling scientists and engineers to make breakthrough post-genomic discoveries and develop new applications that improve the quality of human life. Bruker’s high performance scientific instruments and high value analytical and diagnostic solutions enable scientists to explore life and materials at molecular, cellular, and microscopic levels. In close cooperation with our customers, Bruker is enabling innovation, improved productivity, and customer success in post-genomic life science molecular and cell biology research, in applied and biopharma applications, in microscopy and nanoanalysis, as well as in industrial and cleantech research, and next-gen semiconductor metrology in support of AI. Bruker offers differentiated, high-value life science and diagnostics systems and solutions in preclinical imaging, clinical phenomics research, proteomics and multiomics, spatial and single-cell biology, functional structural and condensate biology, as well as in clinical microbiology and molecular diagnostics. For more information, please visit www.bruker.com.
Contacts
Trade Press & Customer Contact:
Alessandro Abramo
Director Marketing & Product Management
Bruker Microbiology & Infection Diagnostics Division
T: +39-348-360-9058
E: alessandro.abramo@bruker.com
Investor Contact:
Joe Kostka
Director, Investor Relations & Corporate Development
Bruker Corporation
T: +1 (978) 313-5800
E: Investor.Relations@bruker.com




