NANOBIOTIX announced new data from Study 1100, a US Phase 1 dose escalation and dose expansion study evaluating radiotherapy-activated NBTXR3 followed by anti-PD-1 immune checkpoint inhibitors as a second-or-later line therapy for patients with advanced solid and metastatic tumors.
- Data show favorable safety and feasibility in 68 heavily pre-treated patients with R/M-HNSCC (Intention-to-Treat population; “ITT”) who received RT-activated NBTXR3 followed by anti-PD-1 as a second-or-later line treatment
- 48% ORR in evaluable anti-PD-1 naïve patients (n=25); 28% ORR in evaluable anti-PD-1 resistant patients (n=25) as per RECIST 1.1
- 76% DCR in evaluable naïve patients; 68% DCR in evaluable resistant patients as per RECIST 1.1
- Preliminary review of survival data in ITT anti-PD-1 naïve patients (n=33) showed mPFS of 7.3 months and mOS of 26.2 months
- ITT anti-PD-1 resistant patients (n=35) showed mPFS of 4.2 months and mOS of 7.8 months
- Following the ASCO presentation, Nanobiotix will host an investor event on Sunday, June 2nd at 12:00 PM EDT / 6:00 PM CEST to review the presented results
PARIS and CAMBRIDGE, Mass., June 02, 2024 (GLOBE NEWSWIRE) -- NANOBIOTIX (Euronext: NANO –– NASDAQ: NBTX – the ‘‘Company’’), a late-clinical stage biotechnology company pioneering nanoparticle-based therapeutic approaches to expand treatment possibilities for patients with cancer and other major diseases, today announced new data from Study 1100, a US Phase 1 dose escalation and dose expansion study evaluating radiotherapy-activated NBTXR3 followed by anti-PD-1 immune checkpoint inhibitors (“ICIs”) as a second-or-later line (“2L+”) therapy for patients with advanced solid and metastatic tumors. These data were presented by Study 1100 Coordinating Investigator Colette Shen, MD, PhD, at the 2024 Annual Meeting of the American Society for Clinical Oncology (“ASCO 2024”).
“Novel approaches to improving response rates and reversing resistance to anti-PD-1 are an urgent unmet need for patients with recurrent or metastatic head and neck cancer,” said Study 1100 Coordinating Investigator Ari Rosenberg, MD. “The opportunity to ‘prime’ immune activity prior to the administration of anti-PD-1 through radiotherapy-activated NBTXR3’s unique mechanism of action represents a promising potential new treatment approach for patients.”
Abstract #6035: Early signs of efficacy in patients with anti-PD-1 naïve and anti-PD-1 resistant HNSCC treated with NBTXR3/SBRT in combination with nivolumab and pembrolizumab in the phase 1 trial Study 1100
Colette Shen11, Jessica Frakes2, Trevor Hackman1, Jiaxin Niu3, Jared Weiss1, Jimmy Caudell2, George Yang2, Tanguy Seiwert4, Paul Chang5, Septimiu Murgu5, Siddharth Sheth1, Shetal Patel1, Kedar Kirtane2, David Rolando6, Pavel Tyan6, Omar I. Vivar6, Zhen Gooi5, Aditya Joolori5, Ari Rosenberg5
Consistently Favorable Safety and Injection Feasibility
At the data cutoff, NBTXR3 injection followed by standard RT and anti-PD-1 therapy was feasible and well tolerated in 68 heavily pretreated patients with 2L+ R/M-HNSCC (Intention-to-Treat population; “ITT”)
- Serious Grade 3+ adverse events related to the combined therapeutic regimen (injection procedure, RT, NBTXR3, or anti-PD-1) occurred in 8.8% (6/68) of patients.
Early Signals of Efficacy
Anti-PD-1 Naïve Patients
Anti-PD-1 Naïve Population Evaluable for Tumor Response (n=25)
- 48.0% (12/25) overall response rate (“ORR”) as per RECIST 1.1(3 CR; 9 PR)
- 76.0% (19/25) disease control rate (“DCR”) as per RECIST 1.1
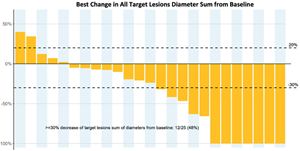
- 48.0% (12/25) all target lesions response (≥ 30% change in target lesion sum of diameters; see below)
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/efeea2d8-b14d-4d5e-910a-c00b6a139cce
Anti-PD-1 Naïve ITT Population for Preliminary Survival Analysis (n=33)
- 7.3 months mPFS
- 26.2 months mOS
- Median follow up of 99 days at data cutoff
Notable Baseline Characteristics in the Anti-PD-1 Naïve Population
- 75.0% of anti-PD-1 naïve patients for whom CPS testing data were available (12/16) had CPS < 20
- 10 anti-PD-1 naïve patients for whom HPV status data were available had HPV-positive R/M-HNSCC of the oropharynx
- At least 33.3% (11/33) of anti-PD-1 naïve patients had at least 2 prior lines of therapy
Anti-PD-1 Resistant Patients
Anti-PD-1 Resistant Population Evaluable for Tumor Response (n=25)
- 28.0% (7/25) ORR as per RECIST 1.1 (2 CR; 5 PR)
- 68.0% (17/25) DCR as per RECIST 1.1
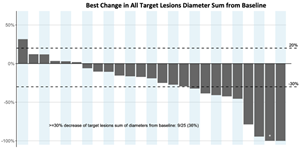
- 36.0% (9/25) all target lesions response (see below)
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/ec2a6ae8-756b-4e60-8827-d78245fa55c7
*One patient is in pathological complete response and has been included as a complete response in this figure
Anti-PD-1 Resistant ITT Population for Preliminary Survival Analysis (n=35):
- 4.2 months mPFS
- 7.8 months mOS
- 31.8 months mOS2 (OS from first anti-PD-1 dose prior to joining Study 1100)
- Median follow up of 90 days at data cutoff
Notable Baseline Characteristics in the Anti-PD-1 Resistant Population
- 57.7% of anti-PD-1 resistant patients for whom CPS testing data were available (15/26) had CPS < 20
- 12 anti-PD-1 resistant patients for whom HPV status data were available had HPV-positive R/M-HNSCC of the oropharynx
- At least 88.6% (31/35) of anti-PD-1 resistant patients had at least 2 prior lines of therapy
- At least 83.0% (29/35) of anti-PD-1 resistant patients had progressive disease when entering Study 1100
“We are excited to see the emergence of several innovative therapeutic approaches to improving treatment outcomes for patients with recurrent or metastatic head and neck cancer in clinical trials. However, it is clear that many of these new product candidates are prioritizing patients in specific settings, while NBTXR3 remains active regardless of several prior prognostic factors such as lines of therapy, CPS score, HPV status, and resistance to therapy,” said Louis Kayitalire, Nanobiotix chief medical officer. “In Study 1100, NBTXR3’s unique mechanism of action could enable a broader population of patients to potentially benefit from therapy, and we look forward to continuing to develop radiotherapy-activated NBTXR3 followed by anti-PD-1 for the treatment of recurrent or metastatic head and neck cancer.”
Investigators concluded that promising early signals of efficacy were observed in Study 1100 patients with naïve or resistant 2L+ R/M-HNSCC who received RT-activated NBTXR3 followed by anti-PD-1. Disease control was observed in both naïve and resistant R/M-HNSCC patients, highlighting the potential for NBTXR3 in this population. Overall, these results warrant further exploration in randomized trials for both naïve and resistant R/M-HNSCC patients.
“These new data from Study 1100 continue to provide encouraging signals that radiotherapy-activated NBTXR3 followed by anti-PD-1 could potentially improve response rates and reverse resistance to anti-PD-1 in patients with recurrent or metastatic head and neck cancer,” said Study 1100 Coordinating Investigator Colette Shen, MD, PhD. “The safety, feasibility, and early signals of efficacy we have observed to date support further evaluation in randomized clinical trials.”
Nanobiotix expects to provide first data from cohort 3 of the Study 1100 dose expansion part (advanced cancers other than R/M-HNSCC with lung, liver, or soft tissue metastases) in 2025.
Nanobiotix Investor Call
Nanobiotix will host a conference call and webcast featuring Nanobiotix chief executive officer and chairman of the executive board, Laurent Levy, following the poster session on Sunday June 2nd, 2024, at 12:00 PM EDT / 6:00 PM CEST.
Details for the call are as follows:
Audio-only dial-in link: click here
Webcast link: click here
Participants can use the audio-only link above to register and obtain dial-in instructions to listen to the presentation via phone and ask questions during the Q&A session, or participants can use the webcast link to register and listen and watch the slide presentation online; the replay version will be available under the same webcast link shortly after the presentation and will be archived on the Company’s website at www.nanobiotix.com. It is recommended to join 10 minutes prior to the event start. Participants are invited to email their questions in advance to investors@nanobiotix.com.
About NBTXR3
NBTXR3 is a novel, potentially first-in-class oncology product composed of functionalized hafnium oxide nanoparticles that is administered via one-time intratumoral injection and activated by radiotherapy. Its proof-of-concept was achieved in soft tissue sarcomas for which the product received a European CE mark in 2019. The product candidate’s physical mechanism of action (MoA) is designed to induce significant tumor cell death in the injected tumor when activated by radiotherapy, subsequently triggering adaptive immune response and long-term anti-cancer memory. Given the physical MoA, Nanobiotix believes that NBTXR3 could be scalable across any solid tumor that can be treated with radiotherapy and across any therapeutic combination, particularly immune checkpoint inhibitors.
Radiotherapy-activated NBTXR3 is being evaluated across multiple solid tumor indications as a single agent or in combination with anti-PD-1 immune checkpoint inhibitors, including in NANORAY-312—a global, randomized Phase 3 study in locally advanced head and neck squamous cell cancers. In February 2020, the United States Food and Drug Administration granted regulatory Fast Track designation for the investigation of NBTXR3 activated by radiation therapy, with or without cetuximab, for the treatment of patients with locally advanced HNSCC who are not eligible for platinum-based chemotherapy—the same population being evaluated in the Phase 3 study.
Given the Company’s focus areas, and balanced against the scalable potential of NBTXR3, Nanobiotix has engaged in a collaboration strategy to expand development of the product candidate in parallel with its priority development pathways. Pursuant to this strategy, in 2019 Nanobiotix entered into a broad, comprehensive clinical research collaboration with The University of Texas MD Anderson Cancer Center to sponsor several Phase 1 and Phase 2 studies evaluating NBTXR3 across tumor types and therapeutic combinations. In 2023, Nanobiotix announced a license agreement for the global co-development and commercialization of NBTXR3 with Janssen Pharmaceutica NV.
About NANOBIOTIX
Nanobiotix is a late-stage clinical biotechnology company pioneering disruptive, physics-based therapeutic approaches to revolutionize treatment outcomes for millions of patients; supported by people committed to making a difference for humanity. The Company’s philosophy is rooted in the concept of pushing past the boundaries of what is known to expand possibilities for human life.
Incorporated in 2003, Nanobiotix is headquartered in Paris, France and is listed on Euronext Paris since 2012 and on the Nasdaq Global Select Market in New York City since December 2020. The Company has subsidiaries in Cambridge, Massachusetts (United States) amongst other locations.
Nanobiotix is the owner of more than 25 umbrella patents associated with three (3) nanotechnology platforms with applications in 1) oncology; 2) bioavailability and biodistribution; and 3) disorders of the central nervous system.
For more information about Nanobiotix, visit us at www.nanobiotix.com or follow us on LinkedIn and Twitter.
Disclaimer
This press release contains “forward-looking” statements within the meaning of the “safe harbor” provisions of the Private Securities Litigation Reform Act of 1995, including, but not limited to, statements regarding the use of proceed therefrom, and the period of time through which the Company’s anticipates its financial resources will be adequate to support operations. Words such as “expects”, “intends”, “can”, “could”, “may”, “might”, “plan”, “potential”, “should” and “will” or the negative of these and similar expressions are intended to identify forward-looking statements. These forward-looking statements, which are based on our management’s current expectations and assumptions and on information currently available to management. These forward-looking statements involve known and unknown risks, uncertainties and other factors that could cause actual results to differ materially from those implied by the forward-looking statements, including risks related to Nanobiotix’s business and financial performance, which include the risk that assumptions underlying the Company’s cash runway projections are not realized. Further information on the risk factors that may affect company business and financial performance is included in Nanobiotix’s Annual Report on Form 20-F filed with the SEC on April 24, 2024 under “Item 3.D. Risk Factors”, in Nanobiotix’s 2023 universal registration document filed with the AMF on April 24, 2024 and subsequent filings Nanobiotix makes with the SEC from time to time which are available on the SEC’s website at www.sec.gov. The forward-looking statements included in this press release speak only as of the date of this press release, and except as required by law, Nanobiotix assumes no obligation to update these forward-looking statements publicly.
Contacts
| Nanobiotix | ||
| Communications Department Brandon Owens VP, Communications +1 (617) 852-4835 contact@nanobiotix.com |
Investor Relations Department Craig West SVP, Investor Relations +1 (617) 583-0211 investors@nanobiotix.com |
|
| Media Relations | ||
| FR – Ulysse Communication Laurent Wormser + 33 (0)6 13 12 04 04 lwormser@ulysse-communication.com |
Global – LifeSci Advisors Kevin Gardner +1 (617) 283-2856 kgardner@lifesciadvisors.com |
|
1University of North Carolina School of Medicine, Chapel Hill, North Carolina, USA; 2Moffitt Cancer Center, Tampa, Florida, USA; 3Banner MD Anderson Cancer Center, Gilbert, Arizona, USA; 4Johns Hopkins Medicine, Baltimore, Maryland, USA; 5The University of Chicago, Chicago, Illinois, USA; 6Nanobiotix, SA, Paris, France
Attachments