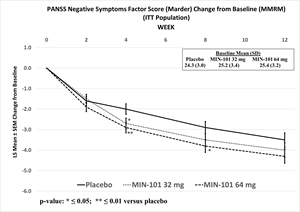
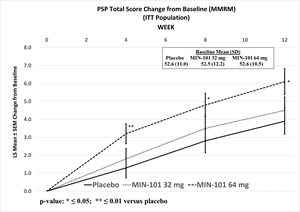
The 64 mg and 32 mg doses were not statistically significantly different from placebo at Week 12 on the primary endpoint, the PANSS Marder Negative Symptoms Factor Score (p ≤0.064 and 0.259, respectively), or the key secondary endpoint, the Personal and Social Performance Scale Total Score (p ≤0.021 and p ≤0.542, respectively) Roluperidone separated from placebo on both primary and key secondary endpoints at Weeks 4, 8 and 12 Roluperidone was generally well tolerated with a safety profile
- The 64 mg and 32 mg doses were not statistically significantly different from placebo at Week 12 on the primary endpoint, the PANSS Marder Negative Symptoms Factor Score (p ≤0.064 and 0.259, respectively), or the key secondary endpoint, the Personal and Social Performance Scale Total Score (p ≤0.021 and p ≤0.542, respectively)
- Roluperidone separated from placebo on both primary and key secondary endpoints at Weeks 4, 8 and 12
- Roluperidone was generally well tolerated with a safety profile comparable to placebo
WALTHAM, Mass., May 29, 2020 (GLOBE NEWSWIRE) --Minerva Neurosciences, Inc.. (NASDAQ: NERV), a clinical-stage biopharmaceutical company focused on the development of therapies to treat central nervous system disorders, announced today that the Phase 3 trial of roluperidone to treat negative symptoms in schizophrenia did not meet its primary (reduction in PANSS Marder Negative Symptoms Factor Score or NSFS) and key secondary (improvement in the Personal and Social Performance Scale Total Score or PSP) endpoints.
Trial parameters and top-line results
In total, 515 patients were enrolled into the trial, and 513 patients received treatment and were included in the safety and Intent-To-Treat population. The trial was conducted in the USA, Europe and Israel. There were 172 patients who received placebo, 172 patients who received roluperidone 32 mg, and 171 patients who received roluperidone 64 mg. Demographic and baseline disease characteristics were comparable across all treatment arms.
The results for both roluperidone doses versus placebo across both the primary and the key secondary endpoints to Week 12 were corrected for multiplicity using the truncated Hochberg procedure.
The primary objective of the trial was to evaluate the change from baseline to Week 12 of NSFS with 32 mg and 64 mg doses of roluperidone compared to placebo in patients diagnosed with schizophrenia presenting with moderate to severe negative symptoms. Neither the 32 mg nor 64 mg dose of roluperidone showed a statistically significant separation from placebo (32 mg: p ≤0.256, effect size [ES]=0.1; 64 mg: p ≤0.064, ES=0.2).
Furthermore, neither dose showed a statistically significant separation from placebo on the key secondary endpoint, the change from baseline to Week 12 in PSP (32 mg: p ≤0.542, ES=0.1; 64 mg: nominal p ≤0.021, ES=0.3).
Although limited inferences can be drawn from this data, unadjusted statistically significant separations from placebo were observed in NSFS at Week 4 for both doses (32 mg: nominal p ≤0.036, ES=0.2; 64 mg: nominal p ≤0.007, ES=0.3), and at Week 8 for the 64 mg dose (nominal p ≤0.027, ES=0.3), and the 64 mg dose was statistically significantly different from placebo as measured by change in PSP at all other assessment timepoints (Week 4, nominal p ≤0.005, ES=0.3; Week 8: nominal p ≤0.018, ES=0.3).
Photos accompanying this announcement are available at
https://www.globenewswire.com/NewsRoom/AttachmentNg/55de50fe-e501-4ef7-a1af-c2a72c8ddc54
https://www.globenewswire.com/NewsRoom/AttachmentNg/ad64a83c-09ed-4727-919a-cf137154cda1
Overall, subgroup analyses by region (USA and rest of the world) and by age groups were similar.
Roluperidone was generally well tolerated, and the incidences of patients who reported treatment‑emergent adverse events over the duration of 12 weeks of treatment were 37% for the 64 mg group, 42% for the 32 mg group, and 33% for placebo. Only 42 patients discontinued from the study due to adverse events, 16 (9%) in 64 mg arm, 18 (10%) in 32 mg arm, and 8 (5%) in placebo arm. Two treatment-unrelated deaths were reported in the 32 mg treatment arm.
“As someone who has spent his career studying everyday functioning in schizophrenia, I see disability as the most important treatment target for people with schizophrenia,” stated Philip Harvey, Ph.D., Leonard M. Miller Professor of Psychiatry and Director of the Division of Psychology at the University of Miami Miller School of Medicine. “The substantial improvements in the PSP scale with the 64 mg dose are tremendously encouraging. These study results represent a very important outcome in a study of a potential treatment of negative symptoms, one of the most important drivers of everyday disability and a critical unmet medical need for patients with schizophrenia. The consistency in treatment effects, in terms of overall negative symptoms and of the most important subtype, reduced emotional experience, between the previous Phase 2b study and the current one is encouraging. The increased placebo effect from the first to second study seems to be the only reason that the study did not meet its primary endpoint.”
“We are encouraged by the results obtained in this study which expand upon the outcome of the Phase 2b study that showed improvements in the primary endpoint and in multiple secondary endpoints,” said Dr. Remy Luthringer, Executive Chairman and Chief Executive Officer of Minerva. “Even though this study didn’t achieve its primary and key secondary endpoints, primarily due to a larger than expected placebo effect at Week 12, results obtained with the 64 mg dose including the early onset of effect and functional improvement as measured by PSP suggest roluperidone merits continued investigation for the treatment of primary negative symptoms. We intend to consult with the US FDA about the next steps in the development of roluperidone for this indication after we complete the analysis of the study data. I would like to express our sincere appreciation to all of the patients, caregivers, the investigators and their staff who participated in this trial.”
The company will hold a webcast event on Monday, June 1, 2020 at 8:30 a.m. to discuss additional data from this study and to provide further insight into the roluperidone Phase 3 trial.
About Minerva Neurosciences
Minerva’s proprietary compounds include: roluperidone (MIN-101), in clinical development for schizophrenia; seltorexant (MIN-202 or JNJ-42847922), in clinical development for insomnia and MDD; and MIN-301, in pre-clinical development for Parkinson’s disease. Minerva’s common stock is listed on the NASDAQ Global Market under the symbol “NERV.” For more information, please visit www.minervaneurosciences.com.
Forward-Looking Safe Harbor Statement
This press release contains forward-looking statements which are subject to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995, as amended. Forward-looking statements are statements that are not historical facts, reflect management’s expectations as of the date of this press release, and involve certain risks and uncertainties. Forward-looking statements include statements herein with respect to the timing and scope of future clinical trials and results of clinical trials with roluperidone (MIN-101); the clinical and therapeutic potential of this compound; the timing and outcomes of future interactions with U.S. and foreign regulatory bodies; our ability to successfully develop and commercialize our therapeutic products; the sufficiency of our current cash position to fund our operations; and management’s ability to successfully achieve its goals. These forward-looking statements are based on our current expectations and may differ materially from actual results due to a variety of factors including, without limitation, whether roluperidone will advance further in the clinical trials process and whether and when, if at all, it will receive final approval from the U.S. Food and Drug Administration or equivalent foreign regulatory agencies and for which indications; whether any of our therapeutic products will be successfully marketed if approved; whether any of our therapeutic product discovery and development efforts will be successful; management’s ability to successfully achieve its goals; our ability to raise additional capital to fund our operations on terms acceptable to us; and general economic conditions. These and other potential risks and uncertainties that could cause actual results to differ from the results predicted are more fully detailed under the caption “Risk Factors” in our filings with the Securities and Exchange Commission, including our Quarterly Report on Form 10-Q for the quarter ended March 31, 2020, filed with the Securities and Exchange Commission on May 4, 2020. Copies of reports filed with the SEC are posted on our website at www.minervaneurosciences.com. The forward-looking statements in this press release are based on information available to us as of the date hereof, and we disclaim any obligation to update any forward-looking statements, except as required by law.
Contact:
William B. Boni
VP, Investor Relations/
Corp. Communications
Minerva Neurosciences, Inc.
(617) 600-7376
PANSS Negative Symptoms Factor Score (Marder) Change from Baseline (MMRM) (ITT Population)
PANSS Negative Symptoms Factor Score (Marder) Change from Baseline (MMRM) (ITT Population)
PSP Total Score Change from Baseline (MMRM) (ITT Population)