Ampio Pharmaceuticals, Inc. (NYSE American: AMPE), today released the following letter to stockholders from its Chairman, Kevin Buchi and Chief Executive Officer, Mike Martino.
|
ENGLEWOOD, Colo., Aug. 3, 2022 /PRNewswire/ -- Ampio Pharmaceuticals, Inc. (NYSE American: AMPE), today released the following letter to stockholders from its Chairman, Kevin Buchi and Chief Executive Officer, Mike Martino. Dear Fellow Stockholders, On behalf of the Board and management team, we are writing to provide an update on the previously announced internal investigations, the clinical development of Ampion and how this impacts our strategic alternatives process, and near-term strategic business alternatives. Finally, we would like to take this opportunity to address the importance of your vote at the 2022 Annual Meeting of Stockholders to be held on Wednesday, August 10, 2022, at 11 am MT. As we announced on May 16, 2022, an independent special committee of the Ampio Board of Directors (the "Special Committee") has been conducting internal investigations focused primarily on (1) the statistical analysis of Ampio's AP-013 clinical trial and (2) unauthorized provision of Ampion, an investigational drug that is not approved by the Food and Drug Administration ("FDA"), for use by individuals not participating in clinical trials ("unauthorized use"). These investigations are now completed and the investigations are summarized below. Through outside counsel, the Special Committee has engaged in extensive fact finding, including interviewing numerous individuals and reviewing documents and emails relating to the AP-013 clinical trial. In connection with the unauthorized use investigation, the Special Committee also reviewed documents, conducted interviews, and reviewed other information regarding the provision and unauthorized use of Ampion. The Special Committee committed significant resources to these endeavors and did not impose material limitations on the investigations' scope, timing, or access to information. As it relates to the AP-013 clinical trial, the Special Committee's primary findings include that certain former Ampio executive officers and senior staff were aware, at the time of the per-protocol interim analysis in March 2020, that the AP-013 trial did not demonstrate efficacy for Ampion on its co-primary endpoints of pain and function; and that these former Ampio executive officers and senior staff did not fully report the results of the AP-013 trial and the timing of unblinding of data from the AP-013 trial. The Special Committee has determined that the Company's current executive officers were not made aware of the unblinding events in March 2020. Through counsel, the Company and the Special Committee have informed the Securities and Exchange Commission of the investigations. The Company and the Special Committee also have contacted the FDA regarding this investigation. As it relates to the unauthorized use of Ampion, the Special Committee's primary finding is that certain Ampio personnel, including a former executive officer and certain former directors, facilitated the provision of Ampion for unauthorized use. Ampio has worked to resolve this issue, including through the conduct of safety surveillance activities relating to the provision of Ampion for unauthorized use, implementation of appropriate mitigation measures (described below), and self-reporting to FDA. The results of the safety surveillance activities have not changed the Company's view of the safety profile of Ampion. The Special Committee has determined that the Company's current executive officers were not involved in the provision of Ampion for unauthorized use. The Company has taken the following actions based in part upon the investigations and the findings of the Special Committee:
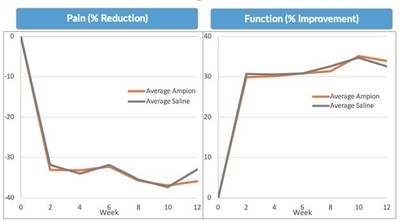
Regarding the status of Ampion™ development, in total, we have conducted seven Phase 2/3 trials, comprising more than 1,500 Ampion-treated patients and more than 1,400 saline-treated patients. Ampion has consistently demonstrated a 30 percent reduction in pain and a 30 percent improvement in function across these trials. Unfortunately, so has the control element, saline. The saline response in our trials is consistent with the response in other published materials on osteoarthritis of the knee ("OAK") trials. As illustrated in the graphs below, Ampion has simply not demonstrated a sufficient therapeutic benefit versus saline to support another superiority trial and a noninferiority trial versus saline would not be commercially competitive. Over the past several months we have undertaken an extensive meta-analysis of a merged database containing all clinical, laboratory and outcome data from all Ampion trials. This analysis has not identified either a sub-population of patients or combination of inclusion/exclusion factors that predicts a trial design where there would be a reasonable expectation for Ampion to outperform saline control. Additionally, we evaluated trial designs, techniques, and tools that might provide an opportunity to highlight the differential benefits of Ampion vs a saline control given our current knowledge, including the following:
In addition, we evaluated the data from AP-008 (n = 342), in which patients were dosed with Ampion every two weeks for up to three doses versus a single dose of saline and confirmed this trial did not yield statistically significant results. Finally, we evaluated conducting trials with alternative controls, such as hyaluronic acid and corticosteroids and concluded that these trials are not feasible or advisable for several reasons. This includes the diminishing use of hyaluronic acid in clinical practice and the magnitude of the corticosteroid efficacy signal. In addition to evaluating the continued development of Ampion, we have also been developing a new and improved formulation of Ampion. While the in vitro and preclinical data are encouraging, we do not believe that redefining Ampio as a preclinical company is in the best interests of stockholders. Based on this extensive analysis, we do not believe that conducting an eighth, and likely a confirmatory ninth, pivotal trial for Ampion is a good use of the Company's cash and resources. With this context in mind, we have been pursuing other strategic alternatives for Ampio with the goal of maximizing value for Ampio stockholders. This exercise includes the potential for Ampio to acquire a product/pipeline and/or execute a reverse merger with a company that is seeking an NYSE American listing and has a development candidate and/or pipeline that we believe would represent a better value proposition for Ampio stockholders. With the assistance of a financial advisor, we have identified several potential acquisition and reverse merger candidates, as well as received inbound interest from potential counterparties for several transformative transactions. We are diligently evaluating all opportunities in terms of value returned to the Ampio stockholders, but this selection process takes time. With that fact in mind, we wish to emphasize the importance of the upcoming vote at the 2022 Annual Meeting of Stockholders and, specifically Proposal No. 3 regarding the reverse stock split. Our goal is to maximize stockholder value through the process we have just outlined. A key component of maximizing stockholder value is maintaining the listing of our stock on the NYSE American. De-listing would likely result in one or more of the following:
We believe it is important to position Ampio for maximum attractiveness as a counterparty and to permit maximum flexibility in the structure of a transaction that we believe will provide maximum value to stockholders. As we reported, Ampio is not currently in compliance with NYSE American listing requirements due to its low stock price. NYSE American requires Ampio to regain compliance with their listing requirements relating to share price by December 23, 2022. A reverse stock split is a means to regain compliance with the NYSE American listing requirements. We want to be clear, stockholder approval of Proposal No. 3 only permits but does not require the Ampio Board to effect a reverse stock split. This proposal gives us the ability to act in a timely manner to regain compliance with the NYSE American requirements, if need be. Stockholder approval at the 2022 annual meeting on August 10, 2022, will give the Board the ability to eliminate the uncertainty regarding future delisting action and help preserve value for the Ampio stockholders in the stock exchange listing. We urge Ampio stockholders to approve Proposal No. 3. Sincerely, Michael A. Martino Forward-Looking Statements This press release contains forward-looking statements that are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995. Any statements contained in this press release that are not statements of historical fact may be deemed to be forward-looking statements. Without limiting the foregoing, words such as "may," "will," "expect," "believe," "anticipate," or "estimate" or comparable terminology are intended to identify forward-looking statements. Such forward-looking statements include, for example, statements about the potential outcomes or timing of the strategic alternatives process being led by the Ampio board of directors. Forward-looking statements are subject to various risks and uncertainties that could cause actual results to differ materially from those expressed or implied in such statements including, among others:
The forward-looking statements in this press release speak only as of the date of this press release. Except as required by law, Ampio assumes no obligation to update or revise these forward-looking statements for any reason, except as required by law. Media Contact:
SOURCE Ampio Pharmaceuticals, Inc. |
||
Company Codes: AMEX:AMPE |