GERMANTOWN, MD / ACCESS Newswire / March 16, 2026 / uBriGene Biosciences (uBriGene), a global CDMO leader, is excited to announce that its strategic partner, Grit Biotechnologies, has received FDA clearance for its Investigational New Drug (IND) application on GT307, a next-generation gene-edited Tumor-Infiltrating Lymphocytes (TIL) therapy.
uBriGene played a key role in enabling this milestone, providing tech transfer, process development, GMP manufacturing, and regulatory support to advance GT307 from development to clinical readiness.
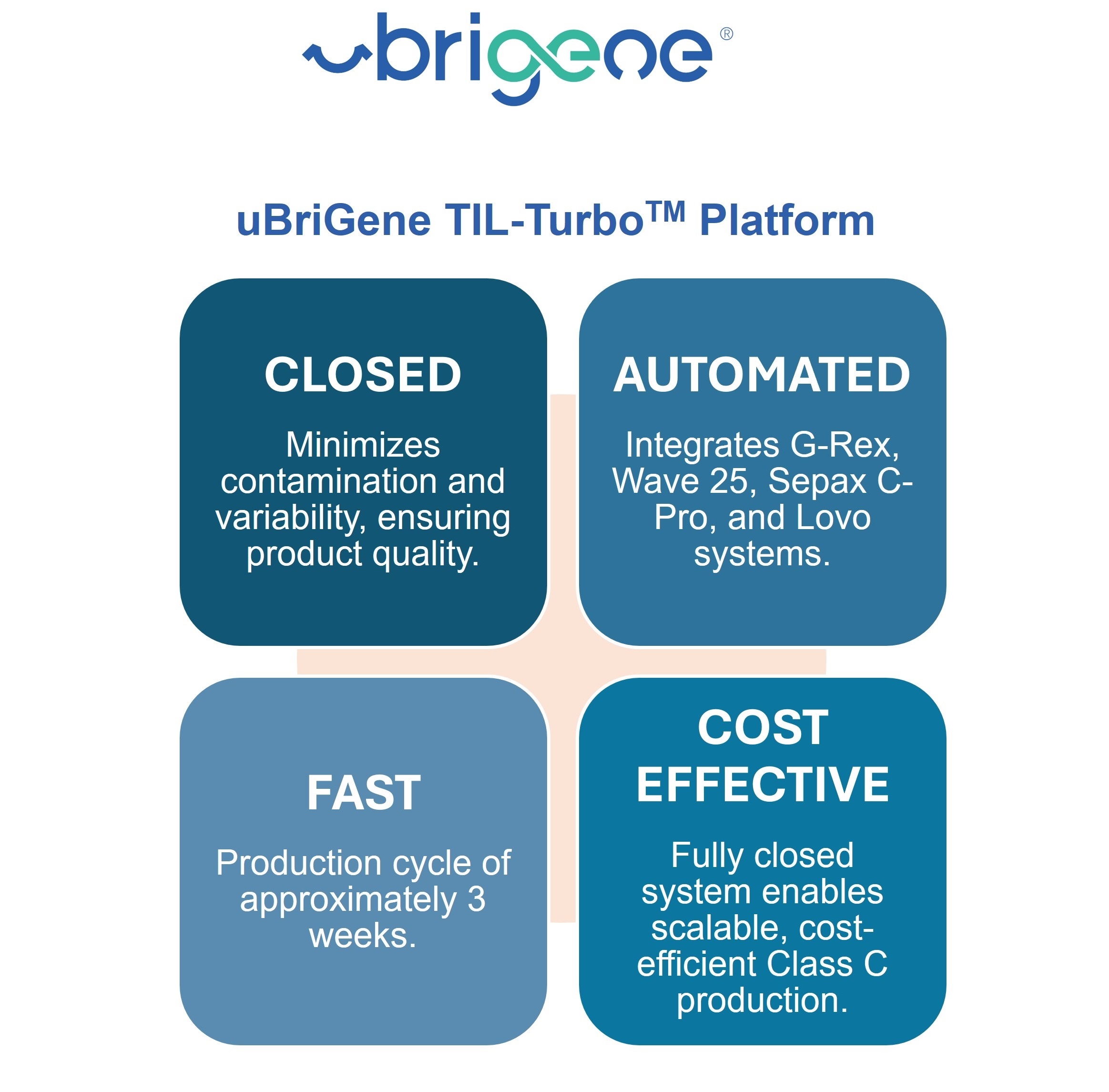
"uBriGene leveraged its TIL-TurboTM production platform, a fully closed and automated system that minimizes variability, reduces contamination risk, and ensures reproducible cell expansion," said Xiulian Sun, CTO, uBriGene Biosciences. "Using this platform, we successfully completed process transfer, process verification, and GMP manufacturing at our Maryland facility, delivering high-quality material ready for first-in-human studies."
"GT307 employs high-fidelity CRISPR/AaCas12bMAX gene editing to knock out key immune regulatory genes, designed to address key bottlenecks of conventional TIL therapies, such as TIL exhaustion within the tumor microenvironment and insufficient persistence in vivo," said Dr. Yarong Liu, CEO, Grit Biotechnologies. "This precise gene-editing strategy, combined with the extensive cell and gene therapy manufacturing experience of our partner uBriGene, allows us to advance next-generation TIL therapies efficiently and safely into the clinic."
The platform has successfully completed more than ten GMP TIL manufacturing runs and enables robust, high-yield expansion within approximately three weeks, consistently achieving cell yields that meet clinical-grade requirements.
About Grit Biotechnologies
Founded in 2019, Grit Biotechnologies is an innovative immunotherapy company focused on TIL and off-the-shelf cell therapies. Its GT101 is China's first TIL therapy approved for clinical trials, now in pivotal Phase II studies. The company's GT201, a membrane-bound IL-15 complex-engineered TIL therapy, has completed dual submissions in China and the U.S.
Grit Biotechnologies' core platforms include StemTexp® for TIL expansion, StaViral® viral stable line technology, ImmuT Finder® immune target discovery, and KOReTIL® high-efficiency gene knockout system. Leveraging these capabilities, the company is advancing next-generation gene-edited TIL therapies into clinical studies for patients in difficult-to-treat solid tumors.
About uBriGene Biosciences
Founded in 2015, uBriGene Biosciences is a leading Contract Development and Manufacturing Organization for advanced therapeutic medicinal products (ATMPs). The company provides integrated CDMO and CRO solutions, encompassing services for cell therapy products, viral vectors, and RNA-related products, with in-house QC testing and regulatory IND filing. Our global GMP Centers of Excellence, AAV-Turbo manufacturing platform, LVV Turbo manufacturing platform, TIL-Turbo manufacturing platform, iPSC Reprogramming technologies are propelling many biotech and pharmaceutical companies forward successfully.
Contact
Mingjuan Liu
Director of Marketing
contact@ubrigene.com
240-551-5140
SOURCE: UBRIGENE BIOSCIENCES INC
View the original press release on ACCESS Newswire




