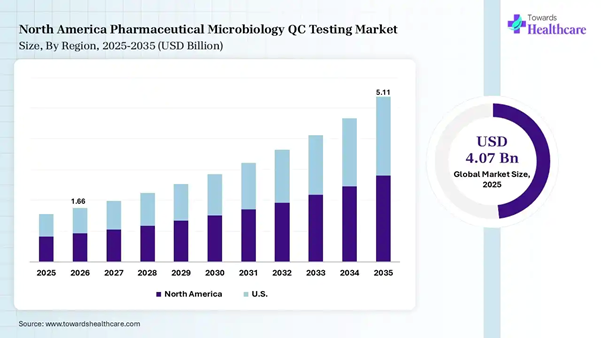
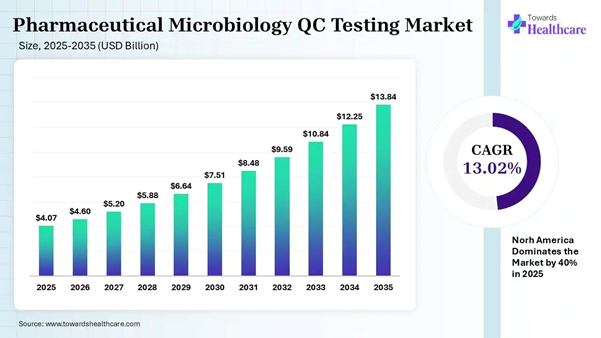
The global pharmaceutical microbiology QC testing market size was estimated at USD 4.07 billion in 2025 and is predicted to increase from USD 4.6 billion in 2026 to approximately USD 13.84 billion by 2035, expanding at a CAGR of 13.02% from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6763
Microbial quality control (QC) testing plays an integral role in pharmaceutical manufacturing, which ensures consistency, product safety, and regulatory compliance. The rigorous standards for microbial testing, validated assays, and precise reference materials as controls are required to ensure transparency in pharmaceutical production. The innovative technologies, such as automated platforms and single-use microbial QC controls in quantitated forms, are accelerating time-to-results for rapid microbiology QC testing. Automated imaging platforms, like the Rapid Micro Biosystems Growth Direct System, help resolve challenges in conventional QC workflows.
The Pharmaceutical Microbiology QC Testing Market: Highlights
• Pharmaceutical microbiology QC testing sector pushed the market to USD 4.6 billion by 2026.
• Long-term projections show USD 13.84 billion valuation by 2035.
• Growth is expected at a steady CAGR of 13.02% in between 2026 to 2035.
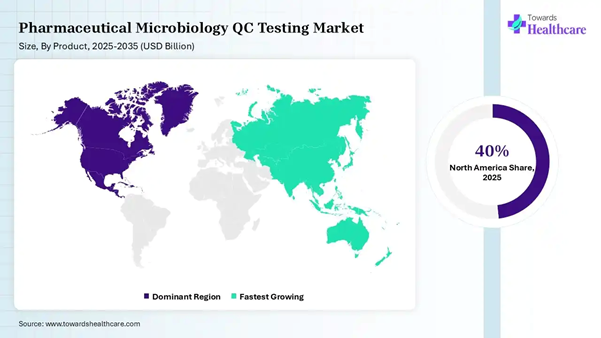
• North America dominated the pharmaceutical microbiology QC testing market by 40% in 2025.
• Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
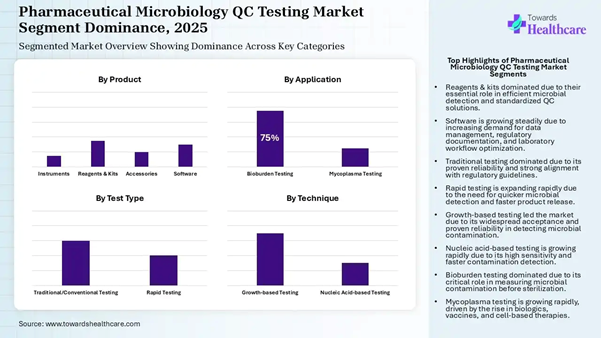
• By product, the reagents & kits segment holds a dominant position in the market by 15% in 2025.
• By product, the software segment is expected to grow at a notable CAGR in the market during the forecast period.
• By test type, the traditional testing segment registered dominance in the market by 60% in 2025.
• By test type, the rapid testing segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By technique, the growth-based testing segment led the pharmaceutical microbiology QC testing market by 70% in 2025.
• By technique, the nucleic acid-based testing segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By application, the bioburden testing segment dominated the market by 75% in 2025.
• By application, the mycoplasma testing segment is expected to grow at the fastest CAGR in the market during the forecast period.
Market Overview
Microbiological QC Testing: Driving Modern Manufacturing
The pharmaceutical microbiology QC testing market is accelerated by the critical and promising nature of microbiological quality control testing in pharmaceutical manufacturing. It ensures patient safety and product integrity, and contributes to sterility testing and environmental monitoring. It also ensures reliability and consistency, which avoids failures that further lead to regulatory action, recalls, and serious risk to patients. The routine QC testing activities across sterile and non-sterile manufacturing are supported by trusted formulations, such as TSB in single- and double-strength formats, specialist sterility testing kits, and TSA with neutralisers.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Challenges in Traditional Microbial QC Workflows
The pharmaceutical microbiology QC testing market revolves around workflows, including time-consuming steps that often extend over one to two weeks, due to the dependency of conventional workflows on laboratory-grade frozen cultures. A technical expertise and hands-on time are needed for these processes to ensure consistency across batches. Results bring variability from differences in media performance, incubation conditions, or a shift to strain phenotype. Long-term low-temperature storage and careful tracking are required for traditional reference material preparation.
Quick Summary Table
|
Table |
Scope |
|
Market Size in 2026 |
USD 4.6 Billion |
|
Projected Market Size in 2035 |
USD 13.84 Billion |
|
CAGR (2026 - 2035) |
13.02% |
|
Leading Region |
North America by 40% |
|
Historical Data |
2020 - 2023 |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Measurable Values |
USD Millions/Units/Volume |
|
Market Segmentation |
By Product, By Test Type, By Technique, By Application, By Region |
|
Top Key Players |
Thermo Fisher Scientific, Inc., Merck KGaA, BD, bioMérieux, Danaher Corporation, Sartorius AG, Rapid Micro Biosystems, Charles River Laboratories |
The Pharmaceutical Microbiology QC
Testing Market: Regional Analysis North America dominated the market share by
40% in 2025, owing to a surge in the production
of biologics, advanced therapies, vaccines, biosimilars,
and other complex products, which require high-throughput microbial quality
control testing. Manufacturers continue to collaborate with policymakers to
advance a manufacturing strategy. The prominent players like Merck have
invested across Delaware, North Carolina, Virginia, and Kansas, which have
created more than 1,600 new American jobs. A state-of-the-art pharmaceutical
Center of Excellence launched by Merck includes both Drug Product investment
and Active
Pharmaceutical Ingredient that will support both the manufacturing
and testing of small molecules. It will also create 8,000 construction jobs and
more than 500 full-time roles. For instance, • In May 2024,
Merck launched its first M-Trace Software & Mobile App, which is a
detailed data tracking solution to digitize microbial quality control and
sterility testing. This software can automatically document data increase
overall process safety for every step of the testing process. U.S. Market Trends The US pharmaceutical microbiology QC
testing market size to reach USD 5.11 billion by 2035. U.S. FDA has enhanced
drug quality assurance inspections and introduced the FDA PreCheck program to
streamline drug manufacturing in the U.S. The leading
pharmaceutical companies in the pharmaceutical microbiology QC testing
market, like Merck, are committed to investing in U.S. innovation to improve
lives and develop and offer life-saving medicines and vaccines
to treat diseases in both humans and animals. For instance, • In October
2025, Merck announced the investment of $3 billion for the construction of the
center of excellence for a pharmaceutical manufacturing facility at Elkton,
Virginia. It aims to expand research and development, and domestic
manufacturing to drive the company’s long-term growth and strengthen the
position of the U.S. as a global
leader in biopharmaceutical innovation. Get the latest insights on life science
industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership Asia Pacific is expected to grow at the
fastest rate in the market during the forecast period due to its emergence as
the global manufacturing hub and adoption of automation and rapid
microbiological methods to improve the pharmaceutical supply chain. Merck, a
leading science and technology company, reported that quality plays a
significant role in the purpose of pharmaceutical companies to impact life and
health with science. Accordingly, Merck has set quality, compliance, and
business support as its crucial approaches to deliver life
science products, services, and digital offerings. For instance, • In June 2024,
Merck invested € 62 million in a new quality control facility in Darmstadt,
Germany, aiming to provide quality control services for the pharmaceutical
production. This establishment will expand the Life Science business and unite
135 employees and multiple functions in one building. Germany Pharmaceutical Microbiology QC
Testing Market Analysis The German quality control facility built
by Merck features special safety requirements for biology laboratories, genetic
engineering, and dust-free laboratories that need specific hygiene protocols.
It also ensures GMP compliance, and encompasses facilities for regenerative
energy generation and low-CO2 construction. For instance, • In June 2024,
Robert Nass, Head of Quality & Regulatory Management, Life Sciences
business of Merck, proclaimed that its new quality control facility will
reflect the company’s commitment to offer the highest quality and safest
products to its customers and patients. This facility was built to meet the
German Sustainable Building Council GOLD standard for sustainability. The Pharmaceutical Microbiology QC
Testing Market: Segmentation Analysis Become a valued research partner with us
- https://www.towardshealthcare.com/schedule-meeting Product Insights The reagents & kits
segment dominated the market in 2025, owing to their vital role in the
detection of microorganisms at all production stages, and rapid
decision-making. The specific kits, like PCR for rapid microbiological methods,
reduce detection times from days to hours. The kits and various reagents are
certified, ready-to-use, and validated, which helps laboratories adhere to
global manufacturing practice (GMP) and pharmacopeial standards. The software segment is expected to grow at
a notable rate in the pharmaceutical microbiology QC testing market during the
forecast period due to the core functions of data management software in
pharmaceutical quality control to streamline laboratory operations and ensure
regulatory compliance. The data management software helps laboratories to
resolve bottlenecks, avoid delayed release of batches, and eliminate the risk
of non-compliance. Advanced software enables automated data capture and
processing, robust audit trails, and centralized data storage. Test Type Insights The traditional testing
segment dominated the market in 2025, owing to the increased focus on
maintaining aseptic conditions, validating sterilization, and improving
environmental monitoring. The conventional QC testing is essential to ensure
the safety, sterility, and contaminant-free nature of drugs before they reach
the market. The traditional culture methods are ideal for industries and
laboratories due to their reliability and long-term regulatory acceptance. The rapid testing segment is expected to
grow at the fastest rate in the pharmaceutical microbiology QC testing market
during the forecast period due to accelerated batch release, real-time
contamination control, and enhanced precision supported by rapid QC testing. It
speeds up the supply chain and reduces inventory hold times. It allows
continuous and real-time monitoring of water and air systems. Technique Insights The growth-based testing segment dominated
the market in 2025, owing to the core roles of growth-based methods in
environmental monitoring, sterility testing, microbial enumeration, and
detection of microcolonies. It is considered the definitive method for
bioburden assessment, sterility assurance, and environmental monitoring. It is
also the standard method to ensure product safety despite the rise of rapid
alternatives. The nucleic acid-based testing segment is
expected to grow fastest in the pharmaceutical microbiology QC testing market
during the forecast period due to its essential role in testing cell
and gene therapies and the detection of mycoplasma. Real-time PCR is
ideal to detect Mycoplasma in biotechnological products and cell substrates.
Moreover, multiplex
PCR allows for the simultaneous screening of multiple pathogens. Application Insights The bioburden testing segment dominated the
market in 2025, owing to the increased focus among researchers on detecting the
presence of viable microorganisms within a device, on a surface, or in a
portion of liquid before sterilization. Bioburden testing detects and
quantifies microbial contamination in a product at various stages of
production, from initial manufacturing through final distribution. It ensures
effective quality control and accurate test results that are needed to reduce
risks for consumers. The mycoplasma testing segment is expected
to grow at the fastest rate in the pharmaceutical microbiology QC testing
market during the forecast period due to the high sensitivity, specificity, and
efficiency of modern mycoplasma testing methods. The Applied Biosystems MycoSEQ
Mycoplasma Detection System of Thermo Fisher Scientific Inc. offers superior
sensitivity, detailed coverage, rapid time to detection, high specificity, and
reduced false positives. It also offers enhanced product safety, cost-efficiency,
and regulatory compliance. Access our comprehensive healthcare
dashboard for detailed market insights, segment breakdowns, regional
performance, and company profiles: https://www.towardshealthcare.com/access-dashboard Pharmaceutical Microbiology QC Testing
Market Companies • Thermo Fisher Scientific, Inc. • Merck KGaA • bioMérieux • Danaher Corporation • Sartorius AG • Rapid Micro Biosystems • Charles River Laboratories • Becton, Dickinson and Company • SGS Life Sciences • Eurofins Scientific • Redberry Segments Covered in the Report By Product • Instruments • Reagents & Kits • Accessories • Software By Test Type • Traditional/Conventional Testing • Rapid Testing By Technique • Growth-based Testing • Nucleic Acid-based Testing • Cellular Component-based Testing • Viability-based Testing • Other Techniques By Application • Bioburden testing • Sterility testing • Environmental monitoring • Endotoxin testing • Pyrogen testing • Mycoplasma testing • Microbial identification • Other applications By Region • North America • Latin America • Europe • Asia Pacific • MEA Immediate Delivery Available | Buy This
Research Report Now @ https://www.towardshealthcare.com/checkout/6763 About Us Towards Healthcare is a leading global provider of technological solutions, clinical
research services, and advanced analytics, with a strong
emphasis on life science research. Dedicated to
advancing innovation in the life sciences sector, we build strategic
partnerships that generate actionable insights and transformative
breakthroughs. As a global strategy consulting firm, we empower life science
leaders to gain a competitive edge, drive research excellence, and accelerate
sustainable growth. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Europe Region: +44 778 256 0738 North America Region: +1 8044 4193 44 APAC Region:
+91 9356 9282 04 Web: https://www.towardshealthcare.com Our Trusted Data Partners Precedence
Research | Towards Packaging | Towards
Food and Beverages | Towards
Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest Browse More Insights of Towards
Healthcare: ➡️The global cell
and gene therapy manufacturing QC market was valued at USD 2.66 billion
in 2024 and increased to USD 3.11 billion in 2025. It is projected to reach
approximately USD 12.35 billion by 2034, expanding at a CAGR of 16.89% during
the forecast period. ➡️The global microbiology
culture market reached US$ 9.38 billion in 2025 and is expected to grow
to US$ 9.93 billion in 2026, with projections indicating a market size of US$
16.55 billion by 2035. The market is anticipated to expand at a CAGR of 5.85%
from 2026 to 2035. ➡️The global microbiology
testing market was valued at USD 5.84 billion in 2024 and grew to USD
6.37 billion in 2025. It is forecasted to reach USD 13.89 billion by 2034,
registering a CAGR of 9.04% over the forecast period. ➡️The global industrial
microbiology market was valued at US$ 12.75 billion in 2024 and
increased to US$ 13.63 billion in 2025. It is projected to reach approximately
US$ 24.92 billion by 2034, growing at a CAGR of 6.93%. ➡️The global clinical
microbiology market is expected to grow from USD 5.12 billion in 2025
to around USD 8.2 billion by 2034, at a CAGR of 5.37%. ➡️The pharma
nitrosamine testing market was valued at US$ 157.9 million in 2025 and
is projected to reach US$ 168.38 million in 2026. It is expected to grow
further to approximately US$ 300.32 million by 2035, at a CAGR of 6.64%. ➡️The global endocrine
testing market reached USD 3.25 billion in 2025 and is expected to grow
to USD 3.52 billion in 2026. The market is projected to reach USD 7.36 billion
by 2035, expanding at a CAGR of 8.54%. ➡️The global syphilis
testing market was valued at US$ 2.29 billion in 2024 and is expected
to grow to US$ 2.45 billion in 2025. It is projected to reach nearly US$ 4.57
billion by 2034, with a CAGR of 6.85%. ➡️The global oncology
biomarker market was valued at US$ 34.16 billion in 2024 and increased
to US$ 38.62 billion in 2025. It is forecasted to reach approximately US$
113.54 billion by 2034, growing at a CAGR of 12.73%. ➡️The global diagnostic
testing market was valued at US$ 203.3 billion in 2024 and is expected
to reach US$ 209.48 billion in 2025. By 2034, it is projected to grow to
approximately US$ 274.53 billion, at a CAGR of 3.04%.