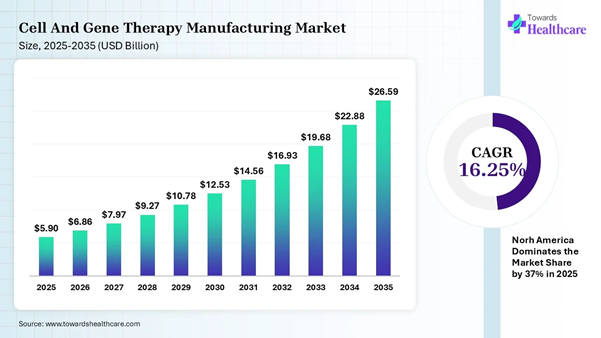
The global cell and gene therapy manufacturing market size is calculated at USD 5.90 billion in 2025, grew to USD 6.86 billion in 2026, and is projected to reach around USD 26.59 billion by 2035. The market is expanding at a CAGR of 16.25% between 2025 and 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/5024
The growing prevalence of a wide range of diseases, including cancer, neurodegenerative diseases, obesity, chronic conditions, diabetes, asthma, nervous system disorders, and many others, has raised the need for robust research and development of targeted therapies. The cell and gene therapies include CAR-T cell therapies, stem cell therapies, gene editing, and many other therapies that aim to treat cancers, genetic diseases, and tissue regeneration and repair.
The U.S. FDA-Approved Cellular and Gene Therapy Products in 2025
|
Sr. No. |
Name of the Product |
Manufacturer |
|
|
ABECMA |
Celgene Corporation, a Bristol-Myers Squibb Company |
|
|
ADSTILADRIN |
Ferring Pharmaceuticals A/S |
|
|
ALLOCORD |
SSM Cardinal Glennon Children's Medical Center |
|
|
AMTAGVI |
Iovance Biotherapeutics, Inc. |
|
|
AUCATZYL |
Autolus Limited |
|
|
BEQVEZ |
Pfizer, Inc. |
|
|
BREYANZI |
Juno Therapeutics, Inc., a Bristol-Myers Squibb Company |
|
|
CARVYKTI |
Janssen Biotech, Inc. |
|
|
CASGEVY |
Vertex Pharmaceuticals Incorporated |
|
|
CLEVECORD |
Cleveland Cord Blood Center |
The Cell And Gene Therapy Manufacturing
Market: Highlights ➔
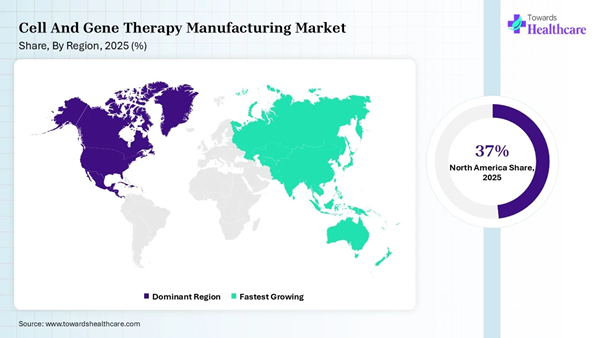
North America dominated the cell and gene therapy
manufacturing market share by 37% in 2025. ➔
Asia-Pacific is anticipated to grow at the fastest rate
in the market during the forecast period. ➔
By therapy, the allogeneic cell therapy segment held a
dominant presence in the market share by 62% in 2025. ➔
By therapy, the autologous cell therapy segment is
expected to be the fastest-growing in the studied years. ➔
By technology, the somatic cell technology segment led
the global market share by 28% in 2025. ➔
By technology, the viral vector technology segment is
expected to grow at the fastest rate in the coming years. ➔
By source, the induced pluripotent stem cells segment
registered its dominance over the global market by 34% in 2025. ➔
By source, the bone marrow segment is expected to grow
rapidly during 2025-2035. ➔
By application, the musculoskeletal segment held the
largest share by 26% of the market in 2025. ➔
By application, the oncology segment is expected to
grow fastest over the projected period. Market Overview: Promising Healthcare
and Transparency in Decision-Making The cell and gene therapy manufacturing
market revolves around excellent scientific research and development strategies of the leading pharmaceutical and biotechnology companies. Some of the prominent players in the
manufacturing of cell and gene therapies are Thermo
Fisher Scientific Inc., Sanofi, Bio-Rad Laboratories, Novartis, GE Healthcare,
Pfizer, Samsung Biologics, Catalent, Roche, and Merck. They aim to expand their
businesses across the globe, particularly leading in the North America and the
Asia Pacific regions. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Cell and Gene Therapy Manufacturing
Solutions: Major Footprint for Biologics The globally leading companies in the cell
and gene therapy manufacturing market, like Thermo Fisher Scientific Inc., are
dedicated to expanding the manufacturing of cell and gene therapy products
through clinical trials and commercial
approval. Apart from reagents and instrumentation, this company provides deep
expertise, custom solutions, technical consultation, innovative technologies,
and services to the manufacturing partners. Thermo Fisher Scientific Inc.
provides single-use technologies and closed systems to offer the flexibility to
innovate new processes and technologies. The robust, sustainable, and
repeatable processes are designed to accelerate production and avoid delays in
manufacturing to transfer. Significance of Single-Use Technology: A single-use technology reduces
contamination risk and minimizes turnover time for equipment. It enables cell
and gene therapy developers to establish
replicable processes and control platforms. It helps them to scale up their
solutions through robust clinical trials and commercial manufacturing. The
single-use consumables include cell factory systems, fluid transfer assemblies,
and bottles. The single-use bioprocessing equipment,
including mixers and bioreactors, is used from bench scale to production to
meet specific cell and gene therapy needs that further expand the cell and gene
therapy manufacturing market globally. Significance of Closed Manufacturing
Solutions: Closed manufacturing systems are used in
cell therapy to reduce contamination risks and reduce ISO cleanroom
requirements. These systems are combined with digital connectivity, which
further enables trackable, repeatable, and GMP-compliant manufacturing
processes. Potential Limitations Revolving around
Scalability, Automation, and Cost The cell and gene therapy manufacturing
market imposes major challenges from laboratory to commercial production.
Accordingly, living cells are hard to grow inside the laboratory in large
batches using heavy equipment. Researchers need to implement specialized
automation approaches for each laboratory step of biological processes,
including cell isolation, cell expansion, genetic modification,
and quality control. The cell and gene therapy manufacturing leads to effective
pricing from hundreds of thousands or millions of dollars per treatment, which
limits their access to patients and commercial expansion. Become a valued research partner with us
- https://www.towardshealthcare.com/schedule-meeting Cell And Gene Therapy Manufacturing
Market: Regional Analysis North America dominated the market share by
37% in 2025, owing to surging FDA approvals, massive investments in
infrastructure, and a shift to contract manufacturing. In September 2025, the
Advanced Research Projects Agency for Health (ARPA-H) launched a program and a
funding opportunity to advance innovative biomanufacturing for cell and gene
therapy. This program aims to build and position the U.S. in advanced
manufacturing methods for high-quality genetic medicines through cutting-edge
production technologies. This program ensures a distributed manufacturing
process combined with automated quality assurance. For instance, ➔ In
April 2025, Cellares made a partnership with the University of Wisconsin
School of Medicine and Public Health to support its clinical production and
regulatory advancement of CRISPR-edited GD2 CAR-T investigational therapy for
solid tumors. The GD2 CAR-T program is designed to modify patients’ T cells
through electroporation by using CRISPR gene editing. U.S. Market Trends The U.S. witnesses the expansion of
clinical research and development, manufacturing innovation and automation, and
increased outsourcing to CDMOs that expand the cell and gene therapy manufacturing market. The Advanced Research Projects Agency for Health (ARPA-H) aims to
produce novel kinds of genetic medicines to make revolutionary treatments
accessible, affordable, and ready to dose within a week of diagnosis. The
domestic manufacturing in the U.S. ensures accessibility of the latest
therapeutic capabilities to researchers and patients. For instance, ➔ In January 2026, the
U.S. Food and Drug Administration (FDA) announced that it had increased
flexibility for the manufacturers of cell and gene therapies through clinical
and commercial quality control tests. The agency presented its new requirements
and reforms that will help researchers address the unique characteristics of
cell and gene therapies and boost innovation. In 2025, the Asia Pacific dominated the
cell and gene therapy manufacturing market due to advancements in manufacturing
technology, expansion of biomanufacturing hubs, and regulatory reforms. Cell
and gene therapy studies are gaining traction in the Asia Pacific region due to
the growing patient demand for life-saving drugs and therapies, and novel
innovations in healthcare. China witnessed the rising prevalence of solid
tumors, which has raised the urgent need for advanced therapies. There is an
increased government and regulatory support for cell and gene therapy trials,
along with a surge in investments in cell and gene therapy products. For instance, ➔ In August 2025, Tony
Acciarito, President of Thermo Fisher Scientific Inc. for the Asia Pacific and
the Middle East and Africa, proclaimed that the company’s integrated solutions
from research to manufacturing will help customers innovate faster, maintain
the highest quality standards, and increase productivity. He also reported that
India is one of the company’s fastest-growing markets, and it is building
capacity and capabilities through crucial investments in India. India Market Analysis India is advancing in the cell and gene
therapy manufacturing market due to the affordability and accessibility of
treatments, government funding and policies, and the growth of contract
development and manufacturing organizations (CDMOs). Research suggests that
India’s biopharmaceutical market was $8.1 billion in size in 2024, and is
expected to double to $15.9 billion by 2030. Studies implied that Bharat
Biotech in India has expanded its business from vaccine production to cell and
gene therapy production, which is expected to grow from $21.28 billion in 2024
and will reach over five-fold to $117.46 billion by 2034. For instance, ➔ In March 2025, Bharat
Biotech, the vaccine maker in India, launched a cell and gene therapy facility
in the southern Indian state of Telangana through the investment of $75 million
in its first CGT facility. It also expects that its new advanced therapies will
gain momentum in the market in the next three years. Get the latest insights on life science
industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership Cell And Gene Therapy Manufacturing
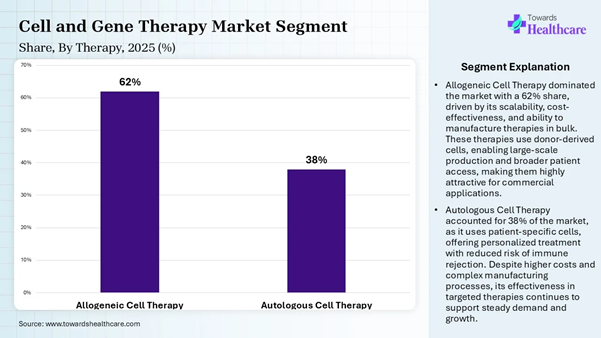
Market: Segmentation Analysis Therapy Insights Therapy Type Market Share (%) Allogeneic Cell Therapy 62% Autologous Cell Therapy 38%
The allogeneic cell therapy segment
dominated the market by 62% share in 2025, owing to its role in manufacturing,
which enables industrial scalability and reduces the cost per patient. It
standardizes quality control and eliminates manufacturing bottlenecks. Allogeneic
therapy is critical for acute or rapidly progressing diseases, which reduces
the wait of patients for custom manufacturing. On the other hand, the autologous cell
therapy segment is expected to grow at the fastest rate in the cell and gene
therapy manufacturing market due to the safety and efficacy associated with
this therapy. It shows proven clinical success for breakthrough treatments,
specifically CAR-T cell therapies for hematologic cancers. This therapy is also
used in regenerative medicine to treat
cardiovascular diseases, neurodegenerative disorders, and autoimmune
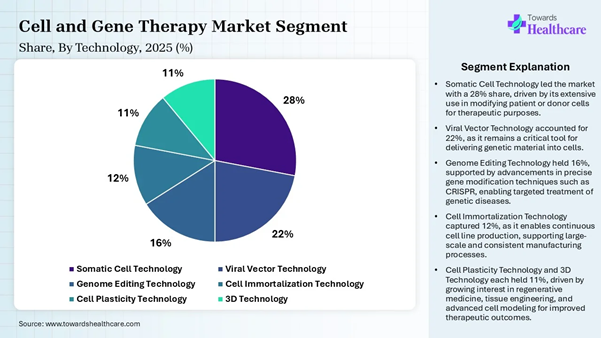
conditions. Technology Insights Technology Type Market Share (%) Somatic Cell Technology 28% Viral Vector Technology 22% Genome Editing Technology 16% Cell Immortalization
Technology 12% Cell Plasticity Technology 11% 3D Technology 11%
The somatic cell technology dominated the
market by 28% in 2025, owing to the increased focus of this technology on
creating delivery systems like lipid nanoparticles in some manufacturing
strategies. Somatic cells are ideal for regenerative medicine and are
reprogrammed to create functional tissues or organoids for drug testing or
transplantation. The manufacturing depends on single-use technologies and
closed-system processing to prevent contamination. The viral vector technology segment is
expected to grow fastest in the cell and gene therapy manufacturing market in
the coming years due to the common viral vectors used in gene therapy, which
include retroviral vectors, lentiviral vectors, Herpes simplex virus vectors,
adenovirus vectors, and adeno-associated virus. It shows an incredible
potential in gene therapy, which transforms the treatment of genetic disorders
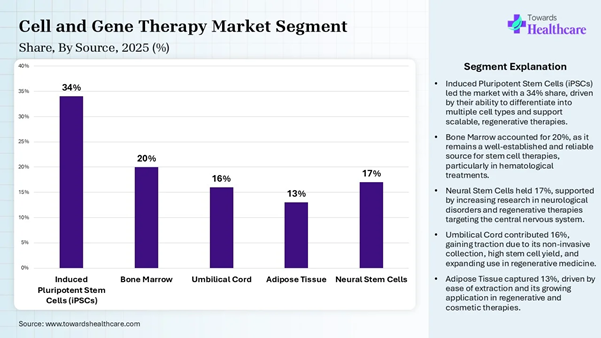
like cystic fibrosis. Source Insights Source Type Market Share (%) Induced Pluripotent Stem
Cells (iPSCs) 34% Bone Marrow 20% Umbilical Cord 16% Adipose Tissue 13% Neural Stem Cells 17%
The induced pluripotent stem cells
dominated the market by 34% in 2025, owing to their contribution in
transforming in vitro research and advancing regenerative medicine. They are
widely used for drug screening, modeling human
development and disease, and developing cell therapies. The
development of induced pluripotent stem cell technology has opened up wide
opportunities for in vitro modelling of human biology and cell therapy
applications. The bone marrow segment is expected to grow
rapidly in the cell and gene therapy manufacturing market due to the use of
bone marrow components to create biomimetic niches that support the expansion
of engineered cells ex vivo. Bone marrow clots also act as natural scaffolds to
deliver modified cells directly to injury sites. Hematopoietic stem cells are
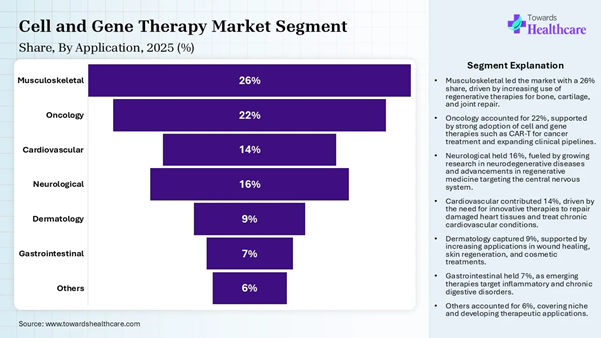
the most widely used targets in gene therapy trials. Application Insights Application Market Share (%) Musculoskeletal 26% Oncology 22% Cardiovascular 14% Neurological 16% Dermatology 9% Gastrointestinal 7% Others 6%
The musculoskeletal segment dominated the
market by 26% share in 2025, owing to the pivotal role of cell and gene therapy
manufacturing in overcoming the limited self-repair capacity of tissues like
tendons and cartilage. Cell and gene therapy has wide musculoskeletal
applications, such as regenerative medicine, gene delivery, gene modification,
biomaterial-assisted therapy, and immune system modulation. The manufacturing
processes, like ex vivo and in vivo methods, induce tissue-specific repair. The oncology segment is expected to grow at
the fastest rate in the cell and gene therapy manufacturing market during the
predicted timeframe due to the major oncology applications of cell
and gene therapy manufacturing. These include CAR-T cell production, gene
modification, oncolytic virotherapy, and process optimization for scale. The
manufacturing processes are central to developing personalized therapies. Access our comprehensive healthcare
dashboard for detailed market insights, segment breakdowns, regional
performance, and company profiles: https://www.towardshealthcare.com/access-dashboard Cell And Gene Therapy Manufacturing
Market Companies • Wuxi Advanced Therapies • Boehringer Ingelheim • Cellular Therapeutics • Miltenyi Biotec • Thermo Fisher Scientific • F. Hoffmann-La Roche Ltd • Bluebird Bio Inc. • Catalent Inc. • F. Hoffmann-La Roche Ltd. • Samsung Biologics •Novartis AG • Lonza • Merck KGaA • Bluebird Bio Inc. • Hitachi Chemical Co., Ltd. • Takara Bio Inc. Segments Covered in the Report By Therapy • Allogeneic Cell Therapy • Autologous Cell Therapy By Technology • Somatic Cell Technology • Cell Immortalization Technology • Viral Vector Technology • Genome Editing Technology • Cell Plasticity Technology • 3D Technology By Source • Induced Pluripotent Stem Cells • Umbilical Cord • Neural Stem • Bone Marrow • Adipose Tissue By Application • Musculoskeletal • Cardiovascular • Gastrointestinal • Neurological • Oncology • Dermatology • Others By Region • North America • Asia Pacific • Europe • Latin America • Middle East and Africa (MEA) Immediate Delivery Available | Buy This
Research Report Now @ https://www.towardshealthcare.com/checkout/5024 About Us Towards Healthcare is a leading global provider of technological solutions, clinical
research services, and advanced analytics, with a strong
emphasis on life science research. Dedicated to
advancing innovation in the life sciences sector, we build strategic
partnerships that generate actionable insights and transformative
breakthroughs. As a global strategy consulting firm, we empower life science
leaders to gain a competitive edge, drive research excellence, and accelerate
sustainable growth. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Europe Region: +44 778 256 0738 North America Region: +1 8044 4193 44 APAC Region:
+91 9356 9282 04 Web: https://www.towardshealthcare.com Our Trusted Data Partners Precedence
Research | Towards Packaging | Towards
Food and Beverages | Towards
Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest Browse More Insights of Towards
Healthcare: Cell
and Gene Therapy CRO Market Trends Shaping 2025 Future Cell
and Gene Overexpression Service Market Research Update Cell
and Gene Therapy Thawing Equipment Market Demand Rising Cell
and Gene Therapy Cold Chain Logistics Market AI Adoption Cell
and Gene Therapy Isolator Market Sees Surge in Innovation Cell
and Gene Supply Chain Solutions Market Expands with AI in 2025 Cell
and Gene Therapy Infrastructure Market Ignites Global Expansion Cell
and Gene Therapy Tools and Reagents Market Driving Future Growth Cell
and Gene Therapy Manufacturing QC Market Driving Personalized Care Cell
and Gene Therapy (CGT) Pharmaceuticals Market Expands with AI Boost Cell
and Gene Therapy Bioassay Services Market Driven by Outsourcing Surge Cell
and Gene Therapy Third-Party Logistics Market Supply Chain & Logistics Data Cell
and Gene Therapy Supply Chain and Logistics Market Powering Advanced Medicine Cell
and Gene Therapy Market Unlocks High-Value Opportunities in Precision Medicine
Era Cell
and Gene Therapy Drug Delivery Devices Market Accelerates Advanced Therapy
Adoption Cell
And Gene Therapy Services Market Transforms Clinical Pipelines Into Commercial
Success Cell
and Gene Therapy Bioanalytical Testing Services Market Evolving with Precision
Analytics