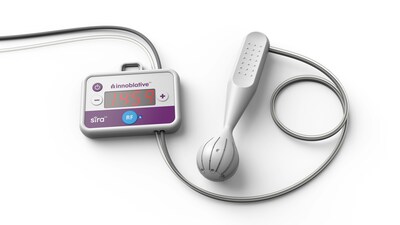
- The company's SIRA® RFA Electrosurgical Device designed specifically for patients undergoing breast-conserving surgery (BCS)
- Breast cancer is the second most common cancer worldwide with over 2.3M new cases each year1
CHICAGO, July 16, 2024 /PRNewswire/ -- Innoblative Designs, Inc. (Innoblative), a private medical device company addressing clinical unmet needs for patients with breast cancer, announced today that it has initiated its first-in-human clinical experience, successfully treating a 64-year-old patient with stage II luminal A breast cancer. The initial lumpectomy case was performed by Dr. Cem Yilmaz, Breast Surgeon and Founding Director of the Istanbul Oncology Hospital in Istanbul, Turkey.
"I am thrilled to perform the first clinical case with the SIRA device and am impressed by its performance. It was extremely easy to use, and I was able to completely ablate the cavity post-lumpectomy without complications," stated Dr. Yilmaz. "This is incredibly exciting as it allowed me to easily address residual cancer in the surrounding tissue at the time of the initial procedure, eliminating the need for my patient to undergo subsequent radiation therapy or reoperations. I believe this will be a game-changer with the potential to make breast conservation surgery a more palatable option for patients."
Breast cancer is a significant global health concern with documented impact in all countries.1 Once diagnosed, patients are faced with difficult decisions regarding available treatment options. Breast conservation therapy (BCT), which includes breast conserving surgery (BCS) plus radiation therapy, may be recommended. However, BCS is associated with a high rate of re-operation with nearly one-in-five patients requiring a follow up procedure within weeks of the initial excision to address residual cancer.2 In addition, subsequent radiation therapy can be cumbersome with frequent treatments over the course of several weeks or months and the potential for a range of unpleasant side effects. Many patients opt for a more drastic approach and undergo a mastectomy to avoid these concerns and alleviate fears of future reoccurrence.
Innoblative's SIRA device aims to overcome these challenges and offer a better treatment option for BCS candidates. Its single-use, disposable applicator is designed specifically for intraoperative ablation of soft tissue, including potential residual cancer post-lumpectomy. With a unique spherical shape, the SIRA electrode circumferentially delivers radiofrequency (RF) energy to the entire cavity and yields reproducible ablation depths to provide greater confidence of a consistent thermal effect. Radiofrequency (RF) ablation has been shown in multiple long-term clinical studies to reduce reoperations and may reduce local recurrence in breast cancer treatment. However, conventional RF devices are not optimized to treat lumpectomy cavities and can lead to variable treatment depths and incomplete ablations.
"Thank you to Dr. Yilmaz and his team for the seamless completion of this first-in-human case," commented Richard Stark, CEO of Innoblative. "This is an exciting time as Innoblative transitions to a commercial company. We look forward to expanding upon this early clinical experience which brings us one step closer to delivering this meaningful technology to breast cancer patients."
About Innoblative
Innoblative is a medical device company dedicated to innovating advanced-energy solutions that improve the way physicians treat breast cancer and other soft tissue diseases. For more information, visit Innoblative.com.
The SIRA® Device received FDA Breakthrough Designation for the use in BCS and is not yet available for sale in the United States for use in BCS.
1Global Cancer Incidence: World Health Organization. GLOBOCAN 2018. https://gco.iarc.fr/today/home. Accessed 9/9/2019.
2Klimberg VS, Ochoa D, Henry-Tillman R, Hardee M, Boneti C, Adkins LL, McCarthy M, Tummel E, Lee J, Malak S, Makhoul I, Korourian S. Long-term results of phase II ablation after breast lumpectomy added to extend intraoperative margins (ABLATE l) trial. Journal of the American College of Surgeons. 2014;218(4):741-9. doi: 10.1016/j.jamcollsurg.2013.12.032. PubMed PMID: 24655863.
Media Contact:
Charlene Herndon
Sprig Consulting
charlene@sprigconsulting.com
![]() View original content to download multimedia:https://www.prnewswire.com/news-releases/innoblative-announces-successful-first-in-human-use-of-its-novel-electrosurgical-device-for-breast-cancer-treatment-302197523.html
View original content to download multimedia:https://www.prnewswire.com/news-releases/innoblative-announces-successful-first-in-human-use-of-its-novel-electrosurgical-device-for-breast-cancer-treatment-302197523.html
SOURCE Innoblative Designs, Inc.