- First Presentation of Clinical Data for CX-2029, a Probody Drug Conjugate, Directed Against CD71: A Previously Undruggable Target - - Data Updates for CX-2009 Support Phase 2 Investigations in Breast Cancer - - Favorable Tolerability Profile Demonstrated for Anti-PD-L1 and Anti-CTLA-4 Probody Therapeutics - - Company to Host Webcast Today, -
- First Presentation of Clinical Data for CX-2029, a Probody Drug Conjugate,
Directed Against CD71: A Previously Undruggable Target -
- Data Updates for CX-2009 Support Phase 2 Investigations in Breast Cancer -
- Favorable Tolerability Profile Demonstrated for Anti-PD-L1 and Anti-CTLA-4 Probody Therapeutics -
- Company to Host Webcast Today, May 29, 2020 at 5:00 p.m. ET/ 2:00 p.m. PT -
SOUTH SAN FRANCISCO, Calif., May 29, 2020 (GLOBE NEWSWIRE) -- CytomX Therapeutics, Inc.. (Nasdaq: CTMX), a clinical-stage oncology-focused biopharmaceutical company pioneering a novel class of investigational antibody therapeutics based on its Probody® therapeutic technology platform, today announced the availability of oral and poster presentations at the American Society of Clinical Oncology’s (ASCO) ASCO20 Virtual Scientific Program taking place from May 29 - May 31, 2020.
“A comprehensive body of evidence was presented today at ASCO 2020 that continues to validate our approach to conditional antibody activation and therapeutic target engagement with the Probody platform,” said Amy Peterson, M.D., chief development officer of CytomX Therapeutics. “The seven presentations collectively highlight the potential of the Probody platform to enable successful engagement of previously undruggable targets, like CD71 and CD166, and create next generation immune-checkpoint inhibitors such as the anti-PD-L1 Probody therapeutic, CX-072, and BMS-986249, a Probody version of ipilimumab. The findings underpin the advancement of all four drug candidates into Phase 2 and our commitment to bringing meaningful advances to patients living with cancer.”
CX-2029: Validating CD71 As A First-in-Class Oncology Target
In the oral abstract 3502, Dr. Melissa Johnson of the Sarah Cannon Research Institute at Tennessee Oncology, presented preliminary clinical data from the first-in-human, dose-escalation, monotherapy Phase 1 study of CX-2029, a Probody drug conjugate (PDC) targeting CD71 (transferrin receptor). CX-2029 is conjugated to the cytotoxic payload MMAE and is being developed by CytomX in partnership with AbbVie. As of an April 20, 2020 data cutoff, 45 patients with advanced solid tumors were enrolled into 8 escalating dose cohorts between 0.1 mg/kg – 5 mg/kg CX-2029 administered intravenously every three weeks.
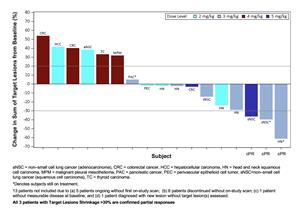
- Evidence of target lesion reduction was seen, principally in patients with tumors of squamous histology. (Figure 1)
° 3 confirmed partial responses were observed in 17 response-evaluable patients treated at doses ≥2 mg/kg of CX-2029, 2 in patients with squamous non-small cell lung cancer (SqNSCLC) and 1 in a patient with head and neck squamous cell cancer (HNSCC).
° 2 of the partial responses (both at the 3 mg/kg dose) were confirmed after the April 20th cutoff date.
CX-2029 Waterfall Plot (Doses 2–5 mg/kg)
Figure 1 is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/51129ea3-a850-4d2f-9c41-4da88807669f
Safety Profile Supports Recommended Phase 2 Dose of 3 mg/kg, Every Three Weeks
- CX-2029 was generally well tolerated at doses up to 3 mg/kg.
- At doses of 0.25 – 5 mg/kg, CX-2029 circulated predominantly as the intact species (>90%).
- The most common treatment related adverse events (TRAE) were infusion related reactions, anemia, and neutropenia/leukopenia.
° Infusion related reactions were mostly Grade 1/2, occurred at the first dose, were not dose dependent and resolved upon initiation of supportive care.
° Hematologic TRAEs Grade ≥ 3 were dose dependent.
• Anemia and neutropenia are commonly observed with the MMAE payload.
• Anemia was managed with transfusions and supportive care. - No CX-2029 treatment related deaths were reported and late onset Grade 3/4 TRAEs were predominately anemia and neutropenia.
- The etiology of anemia is under investigation and is likely multifactorial, including MMAE-related and CD71 expression on red blood cell precursors.
- CytomX is preparing to advance the dose of 3 mg/kg of CX-2029 administered every 3 weeks into 4 dose-expansion cohorts: HNSCC, SqNSCLC, esophageal carcinoma and diffuse large B cell lymphoma.
“Targeting CD71 with this novel approach has the potential to address areas of unmet medical need in difficult to treat tumor types, improving patient benefit,” said Melissa L. Johnson, M.D., CX-2029 principal study investigator and associate director of lung cancer research at Sarah Cannon Research Institute at Tennessee Oncology. "This first-in-human data of CX-2029 establishes the transferrin receptor as a high potential anticancer target addressable with CytomX’s Probody technology.”
CX-2009: Encouraging Clinical Activity Supports Advancement in HER2 Negative Breast Cancer
In Poster 18, Dr. Valentina Boni of START Madrid-CIOCC, presented updated data on CX-2009, a PDC targeting CD166 and conjugated to the cytotoxic payload DM4. As of an April 20, 2020 data cutoff, 96 patients were enrolled into the dose escalation Phase 1 study and received CX-2009 at escalating doses of 0.25 - 10 mg/kg every 3 weeks (86 patients) or 4 - 6 mg/kg every 2 weeks (10 patients).
Durable Clinical Activity Observed in HER2 Negative (HER2-) Breast Cancer
- Evidence of target lesion reduction was observed at doses or dose equivalents of ≥4 mg/kg every 3 weeks across 68 evaluable patients including those with HER2- breast cancer, ovarian cancer, NSCLC and HNSCC.
° HER2- breast cancer patients were heavily pretreated with a median of 7 prior lines of therapy. - 26 patients with HER2– breast cancer who received ≥4 mg/kg of CX-2009 were response-evaluable:
° 2 confirmed partial responses were observed, both in patients with hormone receptor positive (HR+) breast cancer.
° 3 unconfirmed responses were observed in patients with triple negative breast cancer (TNBC).
° Clinical benefit rates of 39% and 35% were observed at 16 and 24 weeks (CBR16 and CBR24, respectively).
• All 4 TNBC patients who achieved CBR16 also achieved CBR24.
CX-2009 Waterfall Plot and Spider Plot: HER2- Breast Cancer (≥4 mg/kg Every 3 Weeks)
Figure 2 is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/3d8523cb-3517-4ae7-b428-19700c46a749
Safety Profile Supports Recommended Phase 2 Dose of 7mg/kg, Every 3 Weeks
- CX-2009 was generally well tolerated at doses up to 7 mg/kg administered every three weeks.
- No dose limiting toxicities (DLTs) were reported at doses up to 7 mg/kg.
- DM4-related toxicities, including ocular, neuropathic, and hepatic were higher in frequency at dose equivalents greater than 7 mg/kg dosed at every three weeks compared to 7 mg/kg or lower.
° Occurrence and severity of ocular adverse events were dose dependent: One Grade 3+ event was observed in a patient treated at 5 mg/kg, none at 7 mg/kg.
° 20% of patients dosed at ≥8 mg/kg experienced Grade 3+ ocular adverse events. - Preliminary pharmacokinetic (PK) data showed that CX-2009 circulates predominantly intact at all doses and PK is not strongly influenced by target-mediated drug disposition or anti-drug antibodies (ADAs). (Poster 329)
In December 2019, CytomX announced the initiation of a Phase 2 expansion study of CX-2009 monotherapy at 7 mg/kg administered every three weeks in up to 40 patients with hormone receptor (ER, PR) positive, HER2 negative breast cancer. In March 2020, CytomX announced the decision to temporarily pause new patient enrollment and new site activation in this study due to the impact of the COVID-19 pandemic. CytomX continues to closely monitor emerging Health Authority guidance and IRB/Ethics Committee recommendations and intends to resume the CX-2009 clinical program as soon as practicable.
“Patients with advanced breast cancer continue to need treatment options, this is especially true for patients with hormone receptor positive and HER2 negative breast cancer that is refractory to hormonal based therapies,” said Alison L. Hannah, M.D., chief medical officer of CytomX Therapeutics. “We believe that targeting CD166, previously considered undruggable as an oncology target, using our Probody drug conjugate platform, may provide a unique treatment opportunity for this patient population. The data in patients with triple negative breast cancer are equally interesting and support the advancement of CX-2009 monotherapy into Phase 2 where we will also evaluate CX-2009 in combination with CX-072, our Probody therapeutic directed against PD-L1.”
CX-072: Anti-PD-L1 Probody Checkpoint Inhibitor
In the oral presentation of Abstract 3005 by Dr. Fiona Thistlethwaite of The Christie NHS Foundation Trust at the University of Manchester, updated data were presented from the Phase 1/2 trial of PROCLAIM-CX-072 monotherapy and CX-072 in combination with ipilimumab with a focus on patients who received long-term treatment, defined as ≥ 6 months of treatment. The CX-072 10 mg/kg monotherapy expansion arm enrolled 114 patients in seven tumor types.
- As of an April 20, 2020 data cutoff, CX-072 monotherapy continued to demonstrate durable anti-tumor activity in patients with IO sensitive tumors such as TNBC, anal squamous cell carcinoma (aSCC), cutaneous squamous cell carcinoma (cSCC) and tumors with high mutational burden (hTMB).
- As of the same data cutoff, CX-072 in combination with ipilimumab had been administered to 27 patients with advanced solid tumors. Durable anti-cancer activity was observed including one complete response in a patient with aSCC who remains on study more than two years after first dose.
- Of the 141 patients across the monotherapy and combination arms: 34 patients received long term treatment in the monotherapy arm (median 11.3 months), and 6 patients received long term treatment in the combination arm (median 21.3 months).
- Grade 3/4 TRAEs were 10% and 5.9% for who received monotherapy < 6 months and ≥ 6 months, respectively, and 33% in each group in the combination arm.
- Long term patients experienced fewer irAEs and had no grade 3+ irAEs suggesting that tolerability early on can impact duration of treatment.
- Preliminary clinical PK data (Poster 332) and translational analyses of pre- and on-treatment biopsies (Poster 172) were supportive of the Probody mechanism of action.
CX-072 Monotherapy Waterfall Plots and Spider Plots (10 mg/kg)
Figure 3 is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/daf244be-f486-43cc-9cf1-b50b60a8dce9
Continued Dr. Hannah “Our ASCO20 clinical, translational, and pharmacokinetic presentations on CX-072 reinforce previously presented data and show clear evidence of anti-cancer activity and favorable tolerability for this unique checkpoint inhibitor. These integrated data support a differentiated profile for CX-072 that we believe can be of potential utility as a unique combination partner for other anti-cancer agents, including CX-2009.”
BMS-986249: Anti-CTLA-4 Probody Demonstrates Encouraging Safety Profile in Phase 1 Trial
Bristol Myers Squibb presented dose escalation data from their Phase 1/2 trial of BMS-986249, a Probody version of the anti-CTLA-4 antibody ipilimumab in Abstract 3508. This trial assessed the safety, pharmacokinetics and pharmacodynamics of escalating doses of BMS-986249 as monotherapy or in combination with the anti PD-1 antibody nivolumab in patients with advanced cancers. The doses of BMS-986249 ranged from 240 mg to 2400 mg (approximately 3 - 30 mg/kg). BMS-986249 was generally well tolerated as monotherapy and in combination with nivolumab. Bristol Myers Squibb has initiated a randomized clinical trial to explore various doses of BMS-986249 in combination with nivolumab in patients with advanced melanoma.
ASCO20 Virtual Scientific Program - Posters and Presentations
Copies of the ASCO20 presentations and posters are available under the Scientific Publications section of the CytomX website at www.CytomX.com.
Conference Call and Webcast
CytomX senior management will host a conference call and live webcast with slides today, Friday, May 29, 2020, from 5:00 p.m. – 6:00 p.m. ET/ 2:00 p.m. – 3:00 p.m. PT to discuss these presentations. This event can be accessed in three ways:
- From the CytomX website: http://ir.cytomx.com/events-and-presentations. Please access the website 15 minutes prior to the start of the call to download and install any necessary audio software.
- By telephone: Participants can access the call by dialing 1-877-809-6037 (United States) or 1-615-247-0221 (International) referencing Conference ID 4267278.
- By replay: A replay of the webcast will be located under the Investor Relations section of CytomX’s website approximately two hours after the conclusion of the live call and will be available for 30 days following the call.
About CytomX Therapeutics
CytomX is a clinical-stage, oncology-focused biopharmaceutical company with a vision of transforming lives with safer, more effective therapies. We are developing a novel class of investigational antibody therapeutics, based on our Probody® technology platform, for the treatment of cancer.
Probody therapeutics are designed to remain inactive until they are activated by proteases in the tumor microenvironment. As a result, Probody therapeutics are intended to bind selectively to tumors and decrease binding to healthy tissue, to minimize toxicity and potentially create safer, more effective therapies. As leaders in the field, our innovative technology is designed to turn previously undruggable targets into druggable targets and to enable more effective combination therapies. CytomX and its partners, comprised of leading biotechnology and pharmaceutical companies, have developed a robust pipeline of potential first-in-class therapeutic candidates against novel, difficult to drug targets and potential best-in-class immunotherapeutic candidates against clinically validated targets. The CytomX clinical stage pipeline includes first-in-class product candidates against previously undruggable targets, including a CD166-targeting Probody drug conjugate wholly owned by CytomX (CX-2009) and a CD71-targeting Probody drug conjugate partnered with AbbVie (CX-2029). CD166 and CD71 are among cancer targets that are considered to be inaccessible to conventional antibody drug conjugates due to their presence on many healthy tissues. The CytomX clinical stage pipeline also includes cancer immunotherapeutic candidates against validated targets such as the CTLA-4-targeting Probody therapeutics, BMS-986249 and BMS-986288, partnered with Bristol Myers Squibb. CytomX has strategic drug discovery and development collaborations with AbbVie, Amgen, Astellas and Bristol Myers Squibb. For additional information about CytomX Therapeutics, visit www.cytomx.com and follow us on LinkedIn and Twitter.
CytomX Therapeutics Forward-Looking Statements
This press release includes forward-looking statements. Such forward-looking statements involve known and unknown risks, uncertainties and other important factors that are difficult to predict, may be beyond our control, and may cause the actual results, performance or achievements to be materially different from any future results, performance or achievements expressed or implied in such statements. Accordingly, you should not rely on any of these forward-looking statements, including those relating to the potential benefits, safety and efficacy or progress of CytomX’s or any of its collaborative partners’ product candidates, the potential benefits or applications of CytomX’s Probody platform technology, and CytomX’s ability to develop and advance product candidates into and successfully complete clinical trials, including the ongoing and planned clinical trials of CX-2009 and CX-2029. Risks and uncertainties that contribute to the uncertain nature of the forward-looking statements include: the unproven nature of CytomX’s novel Probody Platform technology; CytomX’s clinical trial product candidates are in the initial stages of clinical development and its other product candidates are currently in preclinical development, and the process by which preclinical and clinical development could potentially lead to an approved product is long and subject to significant risks and uncertainties, including the risk that the COVID-19 worldwide pandemic may continue to negatively impact the business, research and clinical operations of CytomX or its partners, including the development of preclinical drug candidates due to delays in and disruption of research activities and the development of clinical drug candidates due to delays in or disruption of clinical trials, including impacts on the enrollment of patients in clinical trials or other clinical trial disruptions; the possibility that the results of early clinical trials may not be predictive of future results; the possibility that CytomX’s clinical trials will not be successful; the possibility that current pre-clinical research may not result in additional product candidates; CytomX’s dependence on the success of CX-2009, CX-2029, BMS-986249, BMS-986288, and CX-072; CytomX’s reliance on third parties for the manufacture of the company’s product candidates; and possible regulatory developments in the United States and foreign countries. Additional applicable risks and uncertainties include those relating to our preclinical research and development, clinical development, and other risks identified under the heading "Risk Factors" included in CytomX’s Quarterly Report on Form 10-Q filed with the SEC on May 7, 2020. The forward-looking statements contained in this press release are based on information currently available to CytomX and speak only as of the date on which they are made. CytomX does not undertake and specifically disclaims any obligation to update any forward-looking statements, whether as a result of any new information, future events, changed circumstances or otherwise.
Probody is a U.S. registered trademark of CytomX Therapeutics, Inc.
Investor and Media Contact:
Christopher Keenan
VP, Investor Relations and Corporate Communications
ckeenan@cytomx.com
650-383-0823