Cancer
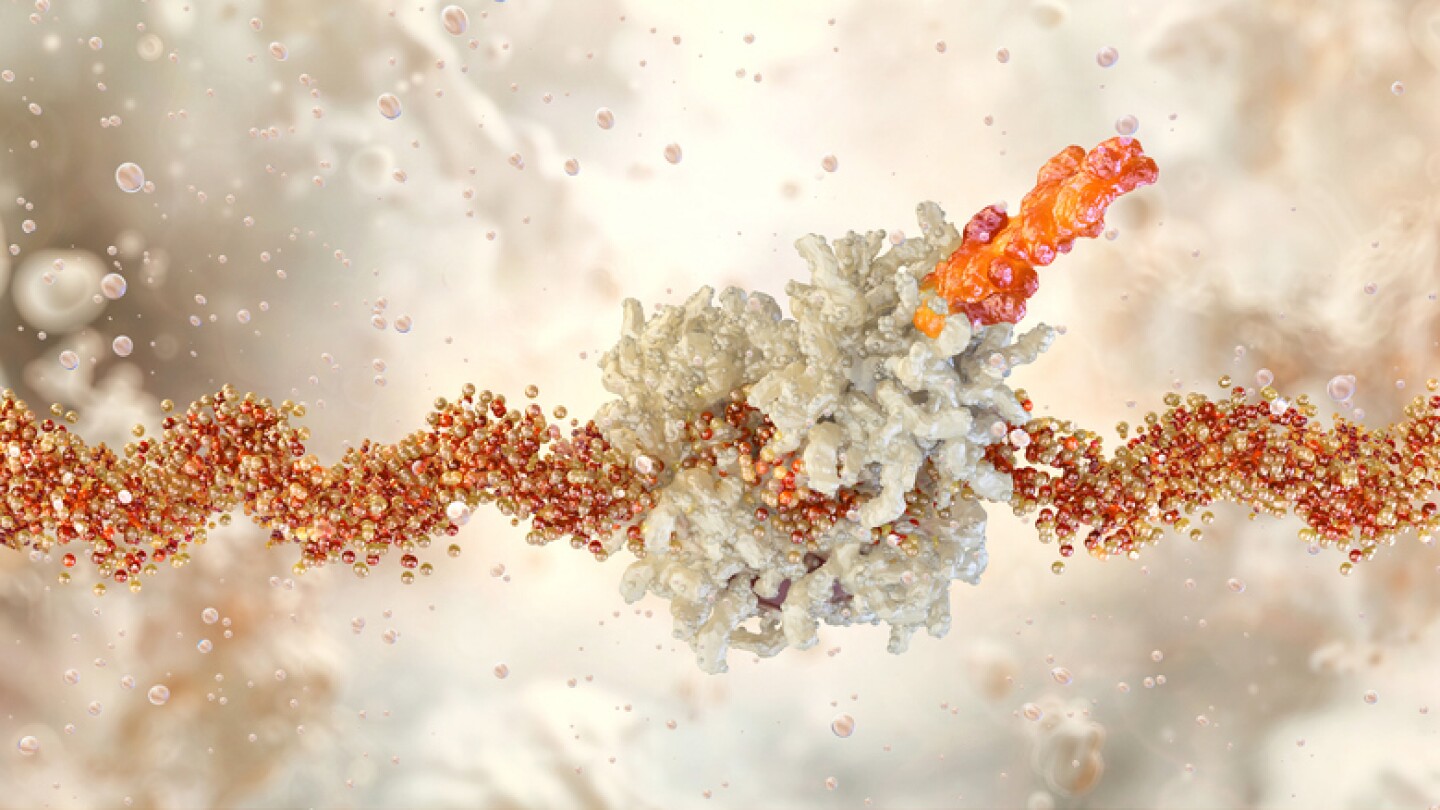
Phase Ib data show Hernexeos can elicit a confirmed objective response rate of 44% in patients with HER2-mutated NSCLC who had previously been treated with a directed antibody-drug conjugate.
The recent announcement of RFK Jr.’s termination of mRNA vaccine contracts is the latest effort to undermine this promising technology at the federal level. Pharmaceutical companies and private investors must fill the gap and ensure that research into this critical resource continues.
Arguably the FDA’s most anticipated decision this month is for a subcutaneous induction formulation of Biogen and Eisai’s Alzheimer’s drug Leqembi, which, according to Eisai, could “help reduce the burden on healthcare professionals and patients.”
Strand Therapeutics’ lead asset is STX-001, an intra-tumor self-replicating mRNA therapy that carries a payload expressing the immunomodulatory protein IL-12.
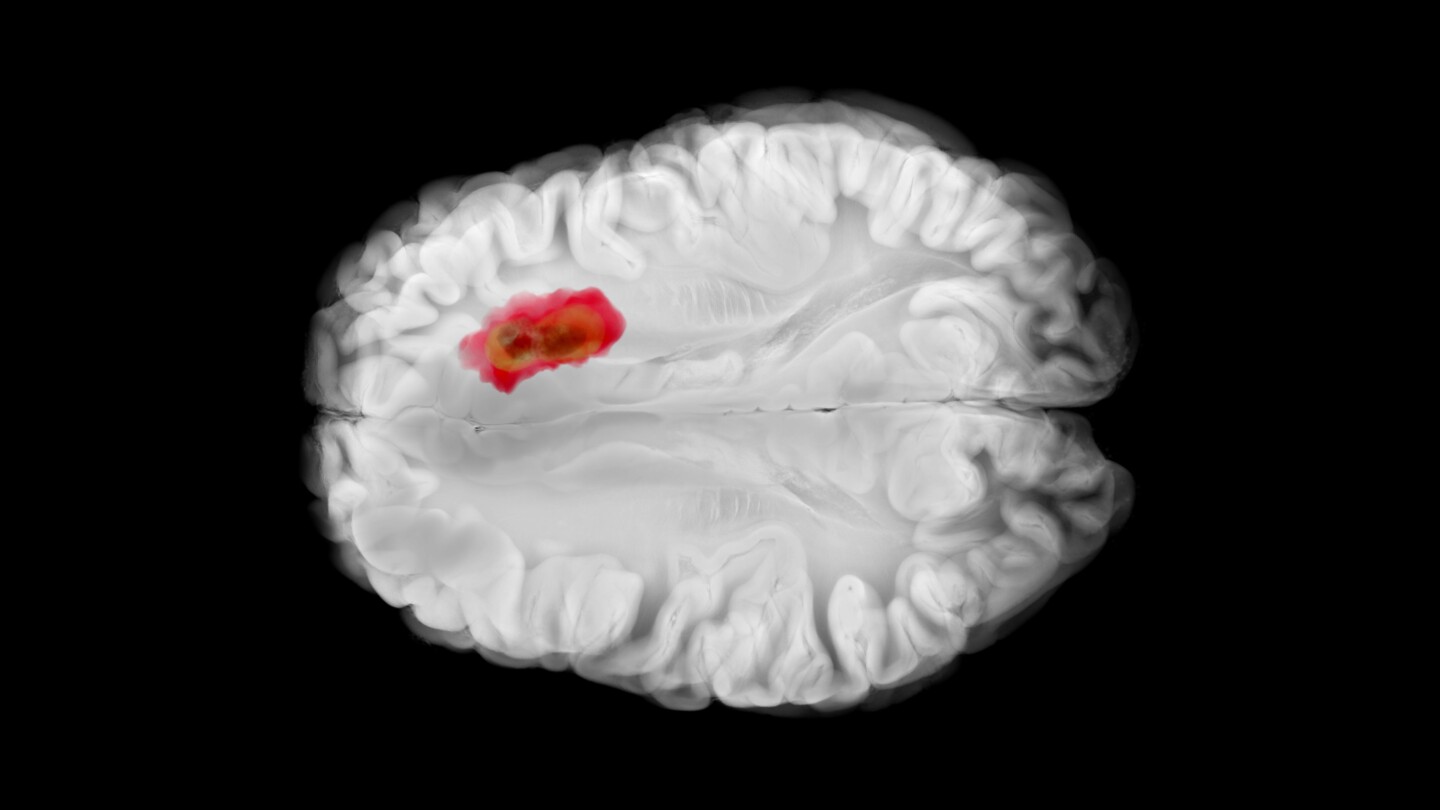
The small molecule drug, acquired by Jazz Pharmaceuticals in its $935 million Chimerix pick-up this spring, is intended for relapsed adult and pediatric patients with H3 K27M mutations.
Terns, once a rising star in obesity and the MASH space, will refocus on cancer and partner out a handful of obesity assets.
BioNTech also laid off 63 employees in June in conjunction with the discontinuation of its cell therapy manufacturing operations in Gaithersburg, Maryland.
George Tidmarsh takes over temporarily at CBER following Vinay Prasad’s abrupt departure; Replimmune trial leaders protest rejection reportedly driven by FDA’s top cancer regulator Richard Pazdur; Merck’s $3 billion savings push claims 6,000 jobs; and Pfizer CEO Albert Bourla addresses President Donald Trump’s new threats around Most Favored Nation drug pricing.
In an open letter, 22 experts who designed and ran Replimune’s Phase III IGNYTE trial answered the FDA’s issues, as outlined in the complete response letter for the melanoma candidate RP1.
According to reporting from multiple outlets, Richard Pazdur, head of the Oncology Center of Excellence at CDER, opposed the consensus opinion of CBER staff to approve the drug. Replimune’s stock has dropped precipitously since the rejection.
PRESS RELEASES