Chinook Therapeutics, Inc. (Nasdaq: KDNY), a biopharmaceutical company focused on the discovery, development and commercialization of precision medicines for kidney diseases, today announced two posters and three informational posters being presented today at ASN Kidney Week 2022 being held virtually and live in Orlando, Florida.
- Atrasentan was well-tolerated with no treatment-related serious adverse events (SAEs)
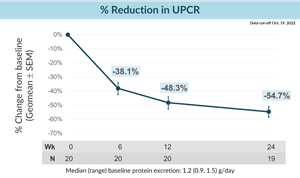
- Atrasentan demonstrated mean proteinuria reductions of 38.1% proteinuria at six weeks of treatment, 48.3% at 12 weeks of treatment and 54.7% at 24 weeks of treatment
- There were no meaningful changes in blood pressure or acute eGFR, suggesting proteinuria reductions were not primarily due to hemodynamic effects of atrasentan, and there were no increases in brain natriuretic peptide (BNP) or mean bodyweight, suggesting minimal fluid retention
- Preclinical data was presented on single-cell RNA-seq of a mouse model of IgAN, revealing a prominent expansion of failed repair proximal tubular epithelial cells, which was reversed by atrasentan but not ACE inhibition
- Three informational trial-in-progress posters were presented on the ongoing phase 3 ALIGN, phase 2 AFFINITY and phase 1/2 BION-1301 clinical trials
- Chinook to host live investor conference call, webcast and in-person event on Friday, November 4th at 6:30 pm EDT with Dr. Sreedhar A. Mandayam, Professor of Nephrology at the University of Texas MD Anderson Cancer Center and Baylor College of Medicine, and Laura Kooienga, MD, practicing nephrologist and director of research at Colorado Kidney Care
SEATTLE, Nov. 03, 2022 (GLOBE NEWSWIRE) -- Chinook Therapeutics, Inc., (Nasdaq: KDNY), a biopharmaceutical company focused on the discovery, development and commercialization of precision medicines for kidney diseases, today announced two posters and three informational posters being presented today at ASN Kidney Week 2022 being held virtually and live in Orlando, Florida.
“The updated interim data from the IgAN patient cohort of the atrasentan phase 2 AFFINITY basket trial continue to demonstrate highly consistent and clinically meaningful proteinuria reductions in patients with IgAN already on a maximally tolerated and stable dose of a RAS inhibitor,” said Eric Dobmeier, president and chief executive officer of Chinook Therapeutics. “We are encouraged by these results, and look forward to reporting topline proteinuria data from our ongoing phase 3 ALIGN trial in 2023.”
TH-PO497 – Atrasentan for the Treatment of IgA Nephropathy: Interim Results from the AFFINITY Study
Atrasentan is a potent and selective endothelin A (ETA) receptor antagonist that has the potential to provide benefit in multiple chronic kidney diseases by reducing proteinuria and having direct anti-inflammatory and anti-fibrotic effects to preserve kidney function. Chinook selected IgAN as the lead indication for atrasentan due to the role of ETA activation in driving proteinuria, mesangial cell activation, kidney inflammation and fibrosis, the hallmarks of IgAN disease progression.
The AFFINITY Study (see www.clinicaltrials.gov, identifier NCT04573920) is an ongoing global phase 2, open-label, basket study to evaluate the safety and efficacy of atrasentan in patients with proteinuric glomerular disease who are at risk of progressive loss of kidney function. The four AFFINITY cohorts consist of patients with: biopsy-proven IgAN with urine protein to creatinine ratio (UPCR) of 0.5 to 1.0 g/g, focal segmental glomerulosclerosis (FSGS), Alport syndrome and diabetic kidney disease (DKD) in combination with an SGLT2 inhibitor. The 20 patients enrolled in each cohort receive 0.75 mg oral atrasentan daily for 52 weeks while continuing to receive a maximally tolerated and stable dose of a RAS inhibitor as standard of care. The study is also evaluating the safety and efficacy of a higher 1.5 mg oral daily dose of atrasentan in patients with FSGS for whom the 0.75 mg dose is well tolerated.
Key highlights from the presentation include the following:
- In the IgAN cohort, median baseline 24-hour urine protein excretion was 1.2 g/day. Overall, 14 of 20 patients enrolled had baseline total proteinuria over one gram per day despite optimized RAS inhibitor treatment, representing an IgAN patient population at high risk for progression.
- As of the October 19, 2022 data cutoff, atrasentan was well-tolerated in patients with IgAN, with no treatment-related SAEs.
- Of the 20 patients enrolled, ten patients remained on study, nine patients completed 52 weeks of treatment and one patient discontinued after 13 weeks of treatment due to a headache. Mean time on treatment for all patients was 45 weeks.
- One patient had an unrelated serious adverse event of a traffic accident, but remained on study.
- Five patients had a treatment-related adverse event, all of which were considered mild or moderate.
- Two patients experienced peripheral edema - one mild and one moderate - which resolved in less than seven days with temporary use of low-dose diuretics.
- All other treatment-emergent adverse events have resolved.
- There were no meaningful changes in blood pressure or acute eGFR effects, suggesting proteinuria reductions were not primarily due to hemodynamic effects of atrasentan. There were no increases in BNP or mean bodyweight, suggesting minimal fluid retention.
- Atrasentan demonstrated mean reductions in 24-hour urine protein creatinine ratio (UPCR) of 38.1% at six weeks of treatment, 48.3% at 12 weeks of treatment and 54.7% at 24 weeks of treatment (see figure below). After 24 weeks of treatment, 15 of the 19 patients (79%) who had completed this visit had greater than a 40% reduction in UPCR.
TH-PO419 – Single Nuclei RNA-seq Reveals Cell-type Specific Responses to Disease and Enalapril in the gddY Mouse Model of IgAN
The gddY mouse model, a spontaneous model of early-onset IgAN, is characterized by IgA immune complex deposition in the mesangium of the kidney, leading to significant proteinuria, glomerular hypercellularity, mesangioproliferative glomerular lesions, glomerulosclerosis and reduced kidney function, all hallmarks of human IgAN. The gddY mouse was utilized as a model to create a high-resolution single-cell dataset to characterize the kidney cell-type specific transcriptional networks that drive IgAN, and to assess the impact of pharmacological intervention with ACE inhibition or atrasentan on these disease-driving transcriptional pathways.
Failed repair proximal tubular epithelial cells (FR PTEC) were identified as the most expanded kidney cell type in the gddY mouse model of IgAN, and may play a major role in tubulointerstitial inflammation and fibrosis leading to progressive kidney function loss. Atrasentan significantly reversed the pathogenic gene expression changes in FR PTECs that were induced in the IgAN model. However, ACEi had minimal impact on IgAN-induced gene expression changes in FR PTECs and rather primarily induced new gene expression changes, unrelated to disease pathogenesis, most prominently in vascular smooth muscle cells. The atrasentan transcriptional response signature identified in this study is currently being evaluated in IgAN patient kidney biopsies, and matched urine and serum samples are being screened for non-invasive surrogate biomarkers.
INFO27 – A Phase 3, Randomized, Double-Blind, Placebo-Controlled Study of Atrasentan in Patients with IgA Nephropathy – The ALIGN Study
A trial-in-progress overview for the global, phase 3, randomized, double-blind, placebo-controlled ALIGN study of atrasentan was presented as an informational poster.
INFO31 – Atrasentan in Patients with Proteinuric Glomerular Diseases – The AFFINITY Study
A trial-in-progress overview for the ongoing, global, phase 2, open-label AFFINITY basket study of atrasentan was presented as an informational poster.
INFO28 – A Phase 1/2 Multicenter Study to Investigate the Safety, Tolerability, Pharmacokinetics and Pharmacodynamics of BION-1301 in Healthy Volunteers and Adults with IgA Nephropathy
A trial-in-progress overview for the ongoing phase 1/2 study of BION-1301 was presented as an informational poster.
All five presentations can be found in the Scientific Publications section of Chinook’s website.
Live Conference Call and Webcast
Chinook will host a live conference call, webcast and in-person event on Friday, November 4th at 6:30 pm EDT to discuss the presentations at ASN Kidney Week 2022 and provide program updates. Members of the Chinook executive team will be joined by Dr. Sreedhar A. Mandayam, Professor of Nephrology at the University of Texas MD Anderson Cancer Center and Baylor College of Medicine, and Laura Kooienga, MD, practicing nephrologist and director of research at Colorado Kidney Care.
Conference Call and Webcast Details
To access the call, please dial (800) 715-9871 (domestic) or (646) 307-1963 (international) and provide the Conference ID 7505851 to the operator.
To access the live webcast and subsequent archived recording of this and other company presentations, please visit the Investors section of Chinook’s website. The archived webcast will remain available for replay on Chinook’s website for 90 days.
About Chinook Therapeutics, Inc.
Chinook Therapeutics, Inc. is a clinical-stage biopharmaceutical company developing precision medicines for kidney diseases. Chinook’s product candidates are being investigated in rare, severe chronic kidney disorders with opportunities for well-defined clinical pathways. Chinook’s lead program is atrasentan, a phase 3 endothelin receptor antagonist for the treatment of IgA nephropathy and other proteinuric glomerular diseases. BION-1301, an anti-APRIL monoclonal antibody, is being evaluated in a phase 1/2 trial for IgA nephropathy. CHK-336, an oral small molecule LDHA inhibitor for the treatment of hyperoxalurias, is being evaluated in a phase 1 healthy volunteer trial. In addition, Chinook is advancing research programs for other rare, severe chronic kidney diseases. Chinook is building its pipeline by leveraging insights in kidney single cell RNA sequencing, human-derived organoids and new translational models, to discover and develop therapeutics with differentiating mechanisms of action against key kidney disease pathways. To learn more, visit www.chinooktx.com.
Cautionary Note on Forward-Looking Statements
Certain of the statements made in this press release are forward looking, including those relating to Chinook’s business, future operations, advancement of its product candidates and product pipeline, clinical development of its product candidates, including expectations regarding cash forecasts and timing of initiation and results of clinical trials, and regulatory submissions, including the timing of the results of our phase 3 ALIGN trial and phase 2 AFFINITY trial of atrasentan and submission for potential accelerated approval. In some cases, you can identify these statements by forward-looking words such as “may,” “will,” “continue,” “anticipate,” “intend,” “could,” “project,” “expect” or the negative or plural of these words or similar expressions. Forward-looking statements are not guarantees of future performance and are subject to risks and uncertainties that could cause actual results and events to differ materially from those anticipated, including, but not limited to, our ability to develop and commercialize our product candidates, including initiation of clinical trials of our existing product candidates or those developed as part of the Evotec collaboration, whether results of early clinical trials or preclinical studies will be indicative of the results of future trials, our ability to obtain and maintain regulatory approval of our product candidates, our ability to operate in a competitive industry and compete successfully against competitors that may be more advanced or have greater resources than we do, our ability to obtain and adequately protect intellectual property rights for our product candidates and the effects of COVID-19 on our clinical programs and business operations. Many of these risks are described in greater detail in our filings with the SEC. Any forward-looking statements in this press release speak only as of the date of this press release. Chinook assumes no obligation to update forward-looking statements whether as a result of new information, future events or otherwise, after the date of this press release.
A photo accompanying this announcement is available at https://www.globenewswire.com/NewsRoom/AttachmentNg/06c41af8-ee4d-4189-ae29-ac0dcac33bb2

Contact: Noopur Liffick Vice President, Investor Relations & Corporate Communications investors@chinooktx.com media@chinooktx.com