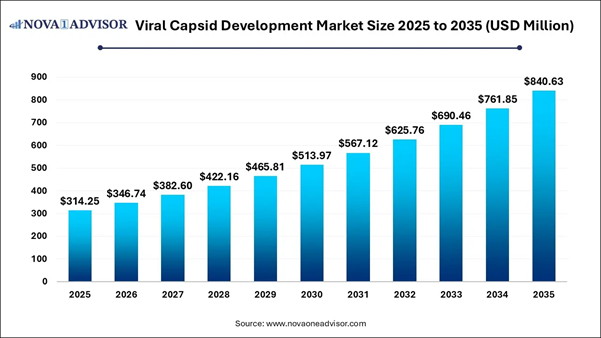
According to Nova One Advisor, the global viral capsid development market size is estimated to be valued at USD 314.25 million in 2025 and is expected to hit around USD 840.63 million by 2035, exhibiting a compound annual growth rate (CAGR) of 10.34% from 2026 to 2035.
From benchtop discovery to targeted therapeutic delivery, the viral capsid development market is rapidly evolving. This evolution is shifting from traditional serotype-based approaches to AI-driven, engineered vector systems that enable precise gene delivery. Powered by advanced directed evolution, synthetic biology, and machine learning guided structural design, this transformation reduces the time required for discovering novel capsids from years to months. It is turning the promise of gene therapy into a safe, reliable, and highly efficient clinical standard for treating genetic disorders, neurodegenerative diseases, and cancers.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/9282
Key Takeaways
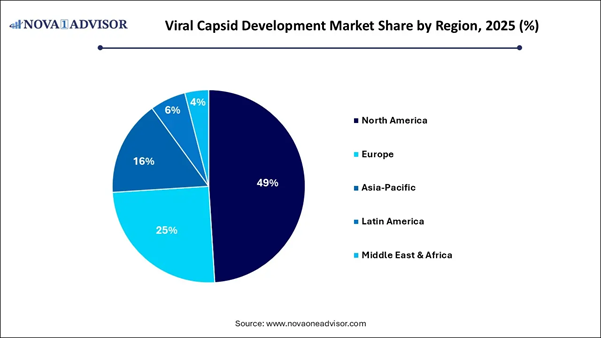
⬥︎ North America dominated with the largest market share in 2025.
⬥︎ Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period.
⬥︎ By type, the adeno-associated virus (AAV) capsid segment accounted for the largest market share in 2025.
⬥︎ By type, the lentiviral segment is projected to grow at a solid CAGR between 2026 and 2035.
⬥︎ By application, the gene therapy segment held a major market share in 2025.
⬥︎ By application, the vaccine development segment is expected to expand at a notable CAGR from 2026 to 2035.
⬥︎ By end-use, the pharmaceutical and biotechnology companies segment contributed the highest market share in 2025.
⬥︎ By end-use, the academic and research institute segment is expected to expand at a notable CAGR from 2026 to 2035.
Market Overview: The New Era of Viral Vector Engineering
The viral capsid development market focuses on engineering the protective protein shell of viruses to enhance gene delivery in gene therapies and vaccines. AAV-based capsids dominate this sector, utilizing AI-driven design, directed evolution, and rational protein engineering to create custom vehicles that improve tissue targeting, reduce immune responses, and allow for lower, safer doses. As gene therapy pipelines expand for rare diseases and oncology, this advanced protein engineering is moving from research and development to essential commercial manufacturing.
AI-Driven Rational Capsid Design: Major Potential
AI and next-generation sequencing are revolutionizing viral vector engineering, replacing slow, traditional methods with AI-driven design that explores billions of synthetic AAV capsid variants. By utilizing machine learning, researchers can now rationally optimize capsids for precise tissue targeting. These AI-engineered variants, currently being developed for central nervous system (CNS) diseases and Duchenne muscular dystrophy, allow for significantly lower therapeutic doses, overcoming the high-dose safety risks and exorbitant costs associated with natural serotypes.
The High Cost of Pure Viral Vectors: Major Limitation
Despite these advancements, the production of gene therapies is hampered by the empty capsid crisis. Inefficient purification processes struggle to separate therapeutic carriers from non-functional, immunogenic empty shells. Regulatory bodies such as the FDA enforce strict purity standards to ensure safety and efficacy, leading to substantial yield losses and extraordinarily high production costs, which pose a significant barrier to scaling up life-saving engineered viral vector therapies.
Immediate Delivery is Available | Get Full Report Access@ https://www.novaoneadvisor.com/report/checkout/9282
Viral Capsid Development Market Report Scope
|
Report Attribute |
Details |
|
Market size value in 2026 |
USD 346.74 Million |
|
Revenue forecast in 2035 |
USD 840.63 Million |
|
Growth Rate |
CAGR of 10.34% from 2026 to 2035 |
|
Base year for Estimation |
2025 |
|
Forecast period |
2026 - 2033 |
|
Quantitative units |
Revenue in USD million/billion, and CAGR from 2026 to 2035 |
|
Segments covered |
Type, Application, End-use, Region |
|
Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
|
Key companies profiled |
PackGene Biotech Inc.; Sartorius AG; Revvity; VectorBuilder; AAVnerGene; AskBio Inc.; Voyager Therapeutics; uniQure N.V.; Vigene Biosciences; Capsigen. |
You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com
| +1 804 441 9344 Viral Capsid Development Market: Segment
Analysis By Type Analysis The adeno-associated virus (AAV) capsid
segment led the market in 2025. This dominance is primarily due to its
exceptional efficiency in gene delivery, low immunogenicity, and extensive use
in gene therapy. AAV vectors are preferred for gene therapies, especially for
treating genetic and rare diseases. Recent developments in customized, engineered
capsids have improved their ability to cross the blood-brain barrier, reduced
immune responses, and enhanced cell receptor binding, allowing for the
large-scale production needed to meet clinical demand. The lentiviral segment is expected to
experience the fastest growth during the forecast period. This is driven by
lentiviruses' unique ability to transduce both dividing and non-dividing cells
for long-term transgene expression. Lentiviral vectors are highly favored in
CAR-T cell therapies for cancers, a rapidly expanding field. Their capability
to stably integrate genetic material into host genomes makes them crucial for
achieving durable therapeutic effects. Additionally, lentiviruses offer lower
immunogenicity and a reduced risk of inflammation compared to other vectors,
further increasing their clinical adoption. By Application Analysis The gene therapy segment was the market
leader in 2025, primarily driven by the high demand for engineered capsids for
addressing genetic and rare diseases. Techniques such as directed evolution and
machine learning are utilized to optimize capsids for specific tissue
targeting, thus improving clinical efficacy. A surge in regulatory approvals
and late-stage clinical trials, particularly for conditions like hemophilia A,
neuromuscular disorders, and ocular diseases, is driving robust capsid
development. Significant investments by pharmaceutical
companies in gene-based treatments for rare diseases. The vaccine development segment is
predicted to grow the fastest during the forecast period, mainly due to the
increasing use of viral vectors in next-generation vaccines, growing research
and development investments, and the demand for rapid, scalable production
platforms. Viral vectors are now being leveraged to create adaptable platforms
that enable quick development, production, and regulatory approval in response
to emerging viral outbreaks. The application of AI-driven design, directed
evolution, and rational protein engineering facilitates the creation of custom
capsids. By End-use Analysis The pharmaceutical and biotechnology
companies dominated the market in 2025, driven by extensive investments in gene
therapy pipelines, the expansion of clinical trials, and vital partnerships
with CDMOs for complex, high-quality viral vector production. These companies
possess the capital and infrastructure necessary to advance gene therapy from
concept to approval, with a specific focus on viral vector engineering for
high-impact applications. Furthermore, companies are increasingly employing AI
and machine learning to accelerate capsid discovery, optimize payload design,
and enhance targeted delivery. The academic and research institute segment
is projected to experience the fastest growth during the forecast period. This
growth is fueled by rising funding for preclinical gene and cell therapy
research, early-stage vector innovation, and collaborations with biotech firms.
There is an increased reliance on viral vectors for exploratory, non-clinical
research related to rare genetic diseases, cancer, and neurological disorders.
Enhanced investment is being directed toward closing the gap between basic
genetic studies and practical, functional, therapeutic applications targeting
specific tissue types. Viral Capsid Development Market:
Regional Analysis North America dominated the market in 2025,
driven by a concentration of advanced biotech hubs, extensive research and
development investments, and a robust regulatory environment that accelerates clinical
trials for gene therapies. There is a high concentration of
biopharmaceutical firms, academic research centers, and CDMOs in areas like
Massachusetts, California, and North Carolina. These regions benefit from
abundant venture capital, public funding, and high R&D spending, which
support the rapid transition from discovery to commercialization. The U.S. leads the market thanks to
significant investments in customized, AI-driven AAV engineering and serves as
the global epicenter for viral capsid innovation. By focusing on advanced CNS
and muscular therapies, U.S. firms are setting the standard for designing
safer, highly efficient vectors that enhance receptor binding and minimize
dosing in gene therapy. 🔹 In April 2026, Aligos achieved a significant milestone with the FDA
granting Fast Track Designation to pevifoscorvir sodium (formerly ALG-000184),
their potential best-in-class CAM-E drug for chronic hepatitis B. Canada is emerging as a powerhouse in viral
capsid development, driving innovation through specialized viral sensitizers.
Supported by investments from the Strategic Innovation Fund (SIF), the country
is scaling up its domestic production capacity for viral vectors. Canada also
offers a high-skilled ecosystem that facilitates rapid clinical trials and
advanced biological medicine development. Asia Pacific region is expected to
experience the fastest growth during the forecast period, primarily due to
substantial investments in gene therapy, a high prevalence of genetic diseases,
and rapidly advancing manufacturing infrastructure. The rapid expansion of
local CDMO, such as WuXi AppTec and GenScript ProBio, is enabling production to
meet international GMP standards. An aging population and a high prevalence of
chronic and rare genetic diseases create a vast, untapped market for advanced
therapies, driving the demand for local, efficient capsid production. India is solidifying its status as a global
pharmaceutical leader by producing most of the the world's vaccines and
expanding into complex biologics, viral vector technology, and capsid-based
therapies. By leveraging its extensive regulatory expertise and increasing
research and development investments, India is transitioning from a
cost-effective manufacturing hub to a premier center for indigenous innovation.
🔹 In November 2025, India introduced
BIRSA 101, its first indigenous and affordable CRISPR-based gene therapy aimed
at curing sickle cell disease. This initiative is part of the country's goal to
achieve a Sickle Cell-Free Nation by 2047 developed by CSIR-IGIB, BIRSA 101 to
correct the genetic mutation responsible for the disease. China is quickly emerging as a global
biotech powerhouse, channeling substantial state investment into a growing
pipeline of gene therapies and viral vector manufacturing. By shifting away
from dependence on imports, China is aggressively fostering high-tech innovation
and biosimilars to enhance its technological self-reliance to accelerate the
commercialization of novel, domestically developed vaccines and therapies. Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/9282
Viral Capsid Development Market
Companies ⬥︎ Revvity Revvity, in collaboration with SIRION
Biotech, enhances the viral capsid market by offering advanced AAV capsid
engineering and directed evolution services. Their expertise focuses on
improving tissue tropism through customized, high-performance capsid libraries,
alongside providing comprehensive analytical, QC, and manufacturing services
that expedite gene therapy development. ⬥︎ VectorBuilder VectorBuilder offers an integrated platform
that combines AI-driven in silico design with high-throughput in vivo screening
in non-human primates to develop, optimize, and validate novel AAV capsids. The
company specializes in engineering capsids with enhanced tissue specificity,
such as those capable of penetrating the blood-brain barrier. Additionally,
VectorBuilder provides end-to-end CDMO services for the large-scale production
of these tailored vectors. ⬥︎ AAVnerGene AAVnerGene focuses on the development of
customized AAV capsids using their proprietary ATHENA platforms (I, II, and
III). These platforms utilize directed evolution, random peptide insertion, and
barcoding technology to improve targeting and efficiency, offering tailored
solutions for gene therapy. ⬥︎ PackGene Biotech Inc. PackGene Biotech operates as a leading
CRO/CDMO, providing end-to-end AAV vector services. Their proprietary Alpha 293
high-yield platform significantly boosts production efficiency while reducing
costs, making them a key player in advancing gene therapies. ⬥︎ Sartorius AG Sartorius AG offers a comprehensive
technology portfolio supporting the entire viral capsid production process.
From upstream cell culture in bioreactors to downstream purification using
advanced membrane chromatography, Sartorius provides a full range of solutions
for viral capsid production Key Emerging Innovations in the Viral
Capsid Development Market Emerging Innovation Example Technology Focus Area AI-Driven Capsid Design Dyno bCap1™ (Apertura/Dyno Therapeutics) Enhanced BBB crossing and neural targeting Engineered AAVs (In Vivo) AAV7m8 (Retinal variants) Enhanced intravitreal delivery for blindness Synthetic/Evasive Capsids Immune-evasive AAV (Rational Design) Repeat dosing & reducing neutralising antibodies Hybrid Rep Systems Dual/Single-plasmid design (AAVone) Increasing full-to-empty capsid ratio (Manufacturing) Engineered VLPs (eVLPs) Nanoblades / Base editor-eVLPs Efficient genome editing with high safety profile
Major Shifts in the Viral Capsid
Development Market 🔹 In March 2026, Dyno Therapeutics
launched Dyno Psi-Phi, an AI-powered protein design suite developed in
collaboration with NVIDIA, aimed at accelerating therapeutic discovery by
improving the design of sequence-based medicines. Sam Sinai, Ph.D., noted that
Psi-Phi addresses challenges in connecting computational benchmarks to
real-world outcomes and enhances the diversity of protein design. 🔹 In March 2026, Apertura Gene Therapy announced a manufacturing
collaboration using its TfR1 CapX, an AAV capsid designed to cross the
blood-brain barrier. This partnership with Viralgen aims to streamline the
transition from research to clinical development, supporting the advancement of
gene therapies targeting the CNS. 🔹 In January 2025, Bio-Rad Labs expanded its Vericheck ddPCR
Empty-Full Capsid Kit for AAV serotypes 2 and 8, complementing existing kits
for serotypes 5 and 9. The new kits enable precise measurement of capsid and
genome titers, enhancing compliance with regulatory standards and advancing
gene therapy development. Related Report – ➡️ Viral Capsid Development Market: Explore
advancements in viral capsid engineering for gene therapy applications Viral Capsid Development Market
Segmentation By Type ·
Adeno-associated Virus (AAV) Capsids ·
Adenoviral ·
Lentiviral ·
Others By Application ·
Gene Therapy ·
Vaccine Development ·
Cancer Therapy ·
Others By End-use ·
Pharmaceutical and Biotechnology Companies ·
Academic and Research Institute ·
CROs & CMOs By Region ·
North America o
U.S. o
Canada ·
Asia Pacific o
China o
Japan o
India o
South Korea o
Thailand ·
Europe o
Germany o
UK o
France o
Italy o
Spain o
Sweden o
Denmark o
Norway ·
Latin America o
Brazil o
Mexico o
Argentina ·
Middle East and Africa (MEA) o
South Africa o
UAE o
Saudi Arabia o
Kuwait Immediate Delivery
Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/9282 About-Us Nova One Advisor is a global leader
in market intelligence and strategic consulting, committed to delivering deep, data-driven
insights that power innovation and transformation across industries. With a
sharp focus on the evolving landscape of life sciences, we specialize in
navigating the complexities of cell and gene therapy, drug development, and the
oncology market, enabling our clients to lead in some of the most revolutionary
and high-impact areas of healthcare. Our expertise spans the entire
biotech and pharmaceutical value chain, empowering startups, global
enterprises, investors, and research institutions that are pioneering the next
generation of therapies in regenerative medicine, oncology, and precision
medicine. Web: https://www.novaoneadvisor.com/ Our Trusted Data Partners: Nova One Advisor - Market
Research Reports & Consulting Firm Nova One Advisor offers
comprehensive market research reports with in-depth industry analysis and
market data. Call us: +1 804 420 9370 Email: sales@novaoneadvisor.com Web: https://www.novaoneadvisor.com/ You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344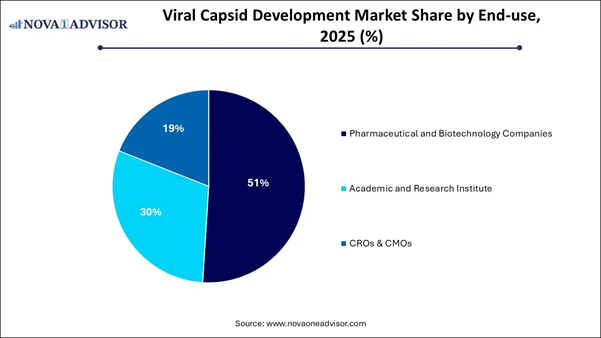
➡️ Cell Therapy Human Raw Materials Market:
Understanding the critical role of human raw materials in advancing cell
therapy
➡️ Scaffold-free 3D Cell Culture Market: Investigating
the rise of scaffold-free 3D cell culture for regenerative medicine
➡️ Effective Microorganisms Market: The growing demand
for effective microorganisms in agriculture and industrial applications
➡️ Automated Liquid Handling Technologies Market:
Innovations driving the growth of automated liquid handling systems in
laboratories
➡️ Platelet Rich Plasma Market: Market trends in
platelet-rich plasma for regenerative medicine and aesthetic applications
➡️ Synthetic Gene Circuits Market: The future of
synthetic biology through innovative gene circuits
➡️ Lyophilization Equipment And Services Market:
Insights into lyophilization processes and equipment in biopharmaceuticals
➡️ Cell Culture Media & Cell Lines Market: Growth
drivers and applications in the cell culture media and cell lines market
➡️ Automated Cell Counting Market: The demand for
automation in cell counting technologies for clinical and research use
➡️ U.S. Fluorescence Microscopy Market: The impact of
fluorescence microscopy advancements on research in the U.S.
➡️ U.S.
Wound Wash Market: Trends and innovations in wound wash solutions
across the U.S. healthcare market
➡️ U.S. Breast Biopsy Devices Market: Analyzing the
growth and technological advancements in breast biopsy devices in the U.S.
➡️ U.S. Senior Living Market: Understanding market
dynamics and trends in the U.S. senior living industry
➡️ U.S. Chemical Indicators Market: The importance of
chemical indicators in U.S. healthcare and sterilization procedures
➡️ Asia Pacific Bioregenerative Aesthetic Injectable Market:
Exploring the market for bioregenerative aesthetic injectables in Asia Pacific


