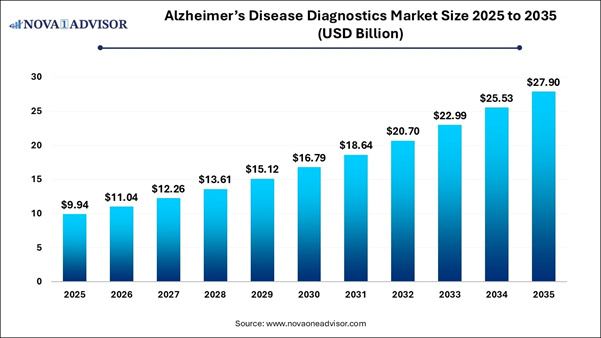
According to latest report, the global Alzheimer’s disease diagnostics market size was USD 9.94 billion in 2025, calculated at USD 11.04 billion in 2026 and is expected to reach around USD 27.90 billion by 2035, expanding at a CAGR of 10.87% from 2026 to 2035.
The global shift from symptom-led approaches to biology-first pathways is driven by the need for early detection following the introduction of new disease-modifying therapies (DMTs). The industry is moving away from costly and invasive cerebrospinal fluid (CSF) tests toward automated, blood-based biomarkers and advanced imaging techniques that are suitable for primary care settings. This transformation is further accelerated by the integration of AI-driven neuroimaging and digital twins, which enhance diagnostic precision, reduce time to diagnosis, and support personalized monitoring for early-stage patients.
Key Takeaways
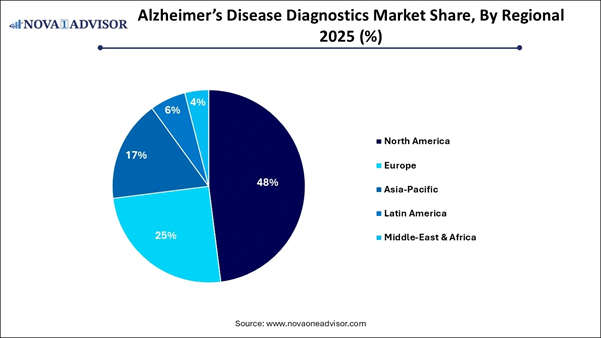
⬥︎North America dominated with the largest market share in 2025.
⬥︎Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period
⬥︎By diagnostics technique, the imaging techniques segment captured the highest market share in 2025.
⬥︎By diagnostics technique, the biomarkers segment is expected to expand at a notable CAGR from 2026 to 2035.
⬥︎By type, the diagnosis segment contributed the highest market share in 2025.
⬥︎By type, the screening segment is poised to grow at a healthy CAGR between 2026 and 2035.
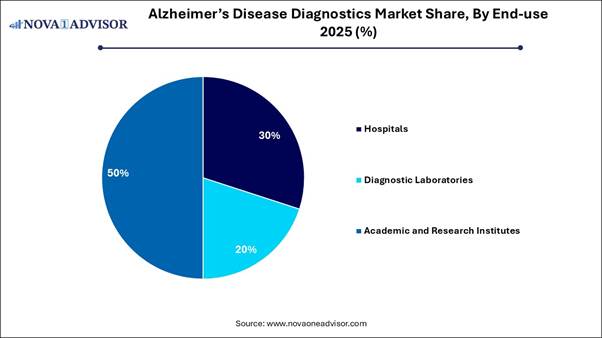
⬥︎By end-use, the academic and research institutes segment accounted for the largest market share in 2025.
⬥︎By end-use, the hospitals segment is projected to grow at a solid CAGR between 2026 and 2035.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/9158
Market Overview: The New Era of Alzheimer’s Detection
The Alzheimer's disease diagnostics market encompasses tools and techniques, such as neuroimaging, biomarkers (blood/CSF), and cognitive tests, to detect and monitor dementia. This market is rapidly evolving from clinical observation to high-tech, biology-first screening, driven by a rapidly aging global population and the urgent need for early and accurate detection. The increased adoption of these diagnostic tools is supported by regulatory approvals and a focus on precision, aiming to identify pathological brain changes before symptoms appear. This improvement in patient management also facilitates the rollout of novel therapeutics.
What happens if a doctor thinks it's Alzheimer's disease?
If a doctor suspects Alzheimer’s disease, the next step is usually a referral to a specialist for a more detailed evaluation and confirmation. These specialists have specific expertise in diagnosing memory and cognitive disorders and may conduct advanced assessments.
The patient may be referred to:
🔹 Geriatricians, who focus on healthcare for older adults and understand how aging affects the body. They can determine whether symptoms are part of normal aging or a more serious condition.
🔹 Geriatric psychiatrists, who specialize in mental and emotional health in older adults and evaluate memory, behavior, and thinking issues.
🔹 Neurologists, who deal with disorders of the brain and nervous system and can order or interpret brain imaging tests.
🔹 Neuropsychologists, who perform detailed tests to assess memory, reasoning, and other cognitive abilities.
In many cases, patients may also be directed to memory clinics or specialized centers, such as Alzheimer’s research centers. These facilities bring together a team of experts who collaborate to diagnose the condition and often have access to advanced tools like brain scans and comprehensive testing methods.
Emergence of Novel Blood-Based Biomarkers: Major Potential
The shift from expensive, invasive diagnostics to highly sensitive blood-based biomarkers (BBBs) represent the primary growth driver in Alzheimer's care, enabling early screening at the primary care level. Authorized assays, like C2N’s PrecivityAD2 and Fujirebio’s Lumipulse G, accurately measure p-tau217/ß-amyloid ratios. These tests provide a scalable alternative to PET scans and assist in determining patient eligibility for new anti-amyloid therapies.
The High Cost of Regulatory Fragmentation: Major Limitation
Significant barriers to the adoption of blood-based Alzheimer's tests stem from a lack of regulatory standardization and limited infrastructure, which hinder rapid global implementation. The diverse and uneven regulatory landscapes across regions complicate the commercialization of new technologies. Additionally, a lack of consensus regarding the interpretation of p-tau217 levels restricts consistent clinical application, slowing the rollout of high-accuracy diagnostic tools.
Immediate Delivery is Available | Get Full Report Access@ https://www.novaoneadvisor.com/report/checkout/9158
Report Scope of Alzheimer’s Disease Diagnostics Market
|
Report Coverage |
Details |
|
Market Size in 2026 |
USD 11.04 Billion |
|
Market Size by 2035 |
USD 27.90 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 10.87% |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2035 |
|
Segments Covered |
Diagnostics Technique, Type, End-use, Region |
|
Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
|
Regional scope |
North America; Europe; Asia Pacific; Latin America; MEA |
|
Key Companies Profiled |
C2N diagnostics, Fujirebio, F. Hoffmann-La Roche AG, Labcorb, Lantheus, Quanterix, Quest Diagnostics, Sysmex, Siemens Healthineers AG |
You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com
| +1 804 441 9344 Alzheimer’s Disease Diagnostics Market:
Segmental Analysis By Diagnostics Technique Analysis In 2025, the imaging techniques segment
dominated the market, primarily due to its established role in visualizing
brain structure and function, ruling out other potential causes of dementia,
and providing critical confirmation for early-stage diagnoses. Magnetic
Resonance Imaging (MRI) is commonly used to identify structural brain
changes, especially atrophy in the hippocampus and entorhinal cortex, and is
frequently employed to monitor disease progression. There is a significant
demand for amyloid PET imaging, as FDA-approved anti-amyloid treatments require
confirmation of amyloid pathology. The biomarkers
segment is projected to experience the fastest growth during the forecast
period. This increase is largely driven by the rapid transition towards early,
non-invasive detection methods. Biomarkers like amyloid-beta, tau, and
neurofilament light chain (NfL) can be detected in peripheral blood, allowing
clinicians to identify Alzheimer’s pathology in its preclinical or early
stages, which is essential for the success of new disease-modifying therapies. By Type Analysis The diagnosis segment led the market in
2025 due to intense demand for confirming the presence of amyloid plaques and
tau tangles, enabling timely and accurate treatment planning as well as
participation in clinical
trials. The biomarker segment, particularly cerebrospinal fluid and blood-based
tests, is witnessing rapid growth as it provides highly accurate and objective
measures of Alzheimer's pathology. Increased public awareness and
government-supported initiatives are further driving the shift towards early
detection using PET scans and liquid biomarkers, regarded as the clinical gold
standard. The screening segment is expected to grow
the fastest during the forecast period, largely due to the urgent need for
early detection, the wide accessibility of assessment tools, and greater awareness
of dementia. The integration of AI and digital platforms for tracking cognitive
decline is boosting the development and use of innovative, remote, and
efficient screening tools. As the global elderly population expands, the demand
for quick diagnostic screening tools will increase, particularly for
early-onset cases. By End-use Analysis In 2025, the academic and research
institutes segment led the market, driven by high volume, continuous, and
early-phase clinical research that leverages advanced imaging and biomarker
technologies. The surge in neurological studies over the last decade has
heightened the demand for tools to study disease progression, validate
biomarkers, and assess treatment efficacy. Academic centers often lead
multi-center clinical trials, thereby driving the adoption and acquisition of
advanced imaging and biomarker kits for participant screening, setting
standards for future diagnostic techniques. The hospitals segment is anticipated to
exhibit the fastest growth during the forecast period, fueled by the increasing
need for advanced diagnostic imaging, the adoption of new high-sensitivity
blood-based biomarker tests, and improved access to comprehensive memory
clinics. Hospitals and specialty neurology clinics are the primary settings for
high-value diagnostic procedures, such as neuroimaging and biomarker testing.
These facilities are better equipped to manage complex diagnostic protocols and
navigate reimbursement pathways for newly approved diagnostic techniques. Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/9158
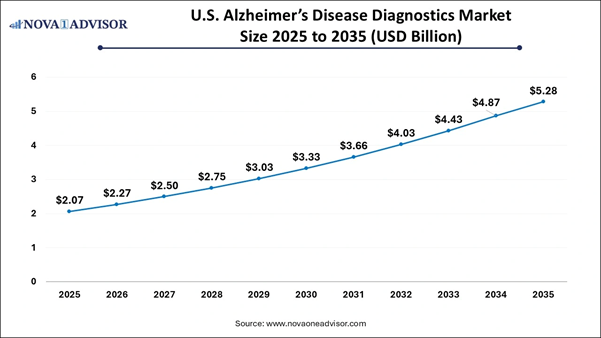
U.S. Alzheimer's Disease Diagnostics
Market Size and Growth 2026 to 2035 The U.S. Alzheimer's Disease Diagnostics
Market size is calculated at USD 2.07 billion in 2025 and is expected to reach
nearly USD 5.28 billion in 2035, accelerating at a strong CAGR of 9.82% between
2026 to 2035. Alzheimer’s Disease Diagnostics Market:
Regional Analysis North America dominated the market in 2025
due to a rapidly aging population, high prevalence rates, advanced healthcare
infrastructure, substantial healthcare spending, and significant research and
development investments. Both government bodies and private foundations provide
substantial funding that encourages innovation and clinical trials. The region
quickly adopts cutting-edge technologies, including advanced neuroimaging and
highly sensitive blood-based biomarkers. Major companies leading diagnostic
innovation, such as Quest Diagnostics, Labcorp, C2N Diagnostics, and Quanterix,
are concentrated in this area. The U.S. acts as the engine of the global
Alzheimer’s diagnostics market, driving the transition from research to
real-world application by leveraging unmatched NIH and private funding to
accelerate the development of groundbreaking, FDA-cleared plasma tests and AI
imaging. With industry leaders like C2N and Quanterix headquartered there, the
U.S. ecosystem effectively connects cutting-edge science with clinical
application, establishing itself as a high-speed innovator in non-invasive
early detection. 🔹 In May 2025, the FDA granted 510(k) clearance to Fujirebio's
Lumipulse G pTau 217 blood tests. This marked the launch of the first
non-invasive in-vitro diagnostic tool designed to accelerate the detection of
Alzheimer's pathology. This breakthrough allows for quick and accessible
amyloid plaque detection via a simple blood draw for adults over 50. Canada is pioneering Alzheimer’s
diagnostics by promoting early detection through a powerful combination of
world-class research via the Canadian Consortium on Neurodegeneration in Aging
(CCNA) and the rapid clinical implementation of innovative tools, such as blood
tests and AI-driven retinal scanning. By transitioning from expensive PET scans
to accessible, non-invasive screenings, Canada is changing the focus of care
from late-stage reactions to early-stage interventions. Asia-Pacific region is expected to
experience the fastest growth during the forecast period. This growth will
primarily be driven by a rapidly aging population, increasing prevalence of
dementia, and significant investments in healthcare infrastructure.
Governments, especially in China and Japan, are heavily funding dementia care
infrastructure and conducting public awareness campaigns. China has mandated
that amyloid PET and blood biomarker testing be available at every
prefecture-level hospital. The growing economic stability in emerging economies
has led to higher healthcare expenditures and improved accessibility to
specialized neuroimaging and diagnostic laboratories. India is rapidly emerging as a key player
in the healthcare sector, driven by advancements in healthcare infrastructure
and a growing demand for early detection methods. The country has also become a
global hub for clinical research, utilizing its vast and genetically diverse population
to generate critical epidemiological insights through innovative studies. This
combination of local diagnostic manufacturing capabilities positions India as a
significant contributor in the battle against dementia. 🔹 In January 2026, Mahajan Imaging and
Labs launched India’s first AI-integrated blood biomarker test for Alzheimer's.
This assay, approved by USFDA and CDSCO, employs a biology-first approach to
detect neurodegenerative changes before symptoms appear. By integrating AI
analytics with PET/MRI scans, it transforms dementia diagnosis from a reactive
to a proactive approach, ultimately improving patient care. China has also emerged as a vital force in
dementia detection, particularly through advanced PET imaging. The market is
increasingly focusing on early detection, bolstered by government-mandated
lifelong management guidelines and the incorporation of cutting-edge AI-driven
diagnostic tools and blood-based biomarkers in public hospitals to tackle the
rising dementia crisis. Alzheimer’s Disease Diagnostics Market
Companies ⬥︎Fujirebio ⬥︎F. Hoffmann-La Roche AG ⬥︎Labcorb ⬥︎Lantheus ⬥︎Quest Diagnostics ⬥︎Sysmex ⬥︎Siemens Healthineers AG Key Initiatives and Programmes
Supporting the Alzheimer’s Disease Diagnostics Market Key Initiatives and Programmes Description Recent Updates Blood-Based Biomarker Commercialization Developing highly sensitive plasma tests (p-tau217, p-tau181) to
detect amyloid pathology without spinal taps or PET scans. FDA approved the first blood-based in vitro diagnostic (Lumipulse
G) for detecting amyloid plaques in adults 55+. ADDF Diagnostics Accelerator (DxA) A $100M+ initiative funding academia/startups to accelerate
biomarker, blood test, and digital tool development. DxA 3.0 launched with added support from Biogen/Eisai; invested in
Alamar's NULISA platform for ultra-sensitive blood testing. AI-Powered Imaging and Diagnostics Integrating AI to improve MRI/PET accuracy in detecting amyloid,
tau, and brain atrophy early. AI-driven platforms like Cortechs.ai are increasingly integrated
with ALZ-NET (Alzheimer's Network) for real-world data tracking. Davos Alzheimer's Collaborative (DAC) Global initiative to build health systems for early diagnosis,
focusing on bringing advanced diagnostics to primary care. Expanding the Global Clinical Trial Network to speed recruitment
and improve diversity in diagnostic testing. Blood Biomarker Clinical Guidelines (AAIC) Efforts by the Alzheimer's Association to establish standardized,
evidence-based use of blood tests in routine clinical practice. Released the first clinical practice guidelines for using blood
biomarkers, providing a framework for primary care adoption.
Major Shifts in the Alzheimer’s Disease
Diagnostics Market ·
In January 2026, BGI Genomics and Roche
Diagnostics launched Alzheimer’s disease tests in China to improve diagnosis
and monitoring options. BGI’s blood test is being used in several cities as an
auxiliary tool to assess disease progression, while Roche is working with local
authorities to introduce its pTau181 test in Boao, Hainan province. These blood
tests could accelerate diagnosis and facilitate access to treatments. ·
In August 2025, C2N Diagnostics introduced the
PrecivityAD2 blood test, which compares favorably with existing PET scans and
cerebrospinal fluid tests. It quantifies specific plasma amyloid beta and tau
levels to help healthcare providers evaluate Alzheimer’s disease in patients
with cognitive decline. ·
In October 2025, NeuroClin partnered with
Advance Tests to offer Scotland’s first commercial blood biomarker test for early
Alzheimer’s detection, developed by Lucent Diagnostics. This test aids in
identifying potential Alzheimer’s in individuals with mild cognitive
impairment, promoting earlier access to treatment options. Related Report – ➡️ Viral Capsid Development Market: Explore
advancements in gene therapy delivery systems and viral vector engineering ➡️ Cell Therapy Human Raw Materials Market: Understand
the growing demand for high-quality human-derived inputs in regenerative
medicine ➡️ Scaffold-free 3D Cell Culture Market: Discover
innovations in physiologically relevant cell modeling without structural
scaffolds ➡️ Effective Microorganisms Market: Analyze the rise
of sustainable agriculture and eco-friendly microbial solutions ➡️ Automated Liquid Handling Technologies Market:
Track automation trends improving precision and throughput in laboratories ➡️ Platelet Rich Plasma Market: Examine expanding
applications in orthopedics, dermatology, and regenerative therapies ➡️ Synthetic Gene Circuits Market: Gain insights into
programmable biology and engineered cellular functions ➡️ Lyophilization Equipment And Services Market:
Explore growth in freeze-drying technologies for pharmaceuticals and biologics ➡️ Cell Culture Media & Cell Lines Market:
Understand the backbone of biopharma research and production workflows ➡️ Automated Cell Counting Market: Discover efficiency
gains in cell analysis through automation and AI integration ➡️ U.S. Fluorescence Microscopy Market: Analyze
cutting-edge imaging technologies enabling advanced biological research ➡️ U.S.
Wound Wash Market: Track demand for infection prevention and
improved wound care solutions ➡️ U.S. Breast Biopsy Devices Market: Examine
advancements in early cancer detection and minimally invasive diagnostics ➡️ U.S. Senior Living Market: Understand evolving care
models driven by an aging population ➡️ U.S. Chemical Indicators Market: Explore quality
assurance tools in sterilization and healthcare safety ➡️ Asia Pacific Bioregenerative Aesthetic Injectable Market:
Discover rising demand for minimally invasive aesthetic treatments ➡️ U.S. Latent Tuberculosis Infection Detection Market:
Analyze advancements in early detection and public health screening ➡️ U.S.
Exoskeleton Market: Track innovation in mobility assistance and
industrial wearable robotics ➡️ In-Vitro Diagnostics (IVD) Market: Explore the
expanding role of diagnostic testing in precision medicine ➡️ RNA Extraction And Purification Market: Understand
critical workflows supporting molecular biology and genomics ➡️ Chronic Disease Management Market: Gain insights
into digital health solutions and long-term patient care strategies Segments Covered in the Report This report forecasts revenue growth at
country levels and provides an analysis of the latest industry trends in each
of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc.
has segmented the Alzheimer’s disease diagnostics market. By Diagnostics Technique By Type By End-use By Regional Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/9158 About-Us Nova One Advisor is a global leader
in market intelligence and strategic consulting, committed to delivering deep, data-driven
insights that power innovation and transformation across industries. With a
sharp focus on the evolving landscape of life sciences, we specialize in
navigating the complexities of cell and gene therapy, drug development, and the
oncology market, enabling our clients to lead in some of the most revolutionary
and high-impact areas of healthcare. Our expertise spans the entire
biotech and pharmaceutical value chain, empowering startups, global
enterprises, investors, and research institutions that are pioneering the next
generation of therapies in regenerative medicine, oncology, and precision
medicine. Web: https://www.novaoneadvisor.com/ Our Trusted Data Partners: Nova One Advisor - Market
Research Reports & Consulting Firm Nova One Advisor offers
comprehensive market research reports with in-depth industry analysis and
market data. Call us: +1 804 420 9370 Email: sales@novaoneadvisor.com Web: https://www.novaoneadvisor.com/ You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344