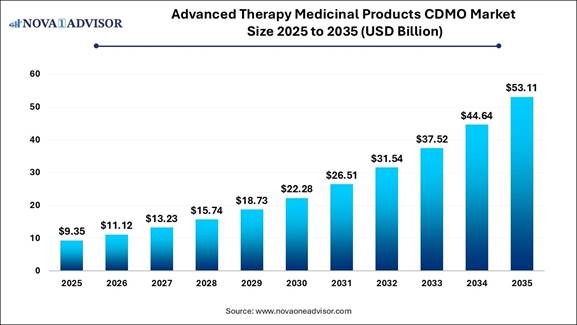
According to Nova One Advisor, the global Advanced Therapy Medicinal Products (ATMP) CDMO market was valued at USD 9.35 billion in 2025 and is anticipated to reach approximately USD 53.11 billion by 2035, growing at a CAGR of 18.97% during the forecast period from 2026 to 2035.
Key Insights on the Advanced Therapy Medicinal Products CDMO Market:
· By Product: Gene therapy led the market in 2025, while cell therapy is the fastest-growing segment.
· By Phase: The Phase II segment held the largest market share in 2025. However, the Phase III segment is expected to experience the fastest growth moving forward.
· By Indication: Oncology was the dominant indication in 2025, but neurological and genetic disorders are projected to see the highest growth rate.
· By Region: North America held the largest market share in 2025. Asia-Pacific is expected to have the fastest growth throughout the forecast period.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/8439
Market Overview: Integrated CDMO Platforms and Advanced Analytics
The advanced therapy medicinal products CDMO market includes contract development and manufacturing organizations providing specialized services such as process development, cGMP manufacturing, and testing for gene therapies, somatic cell therapies, and tissue-engineered products. ATMP CDMOs serve as vital partners for biotech and pharmaceutical companies by offering end-to-end development and GMP manufacturing services for complex, personalized living medicines. This growth is driven by increasing clinical trials for rare diseases and cancer, with high demand for outsourcing due to complex manufacturing needs.
Emergence of New Transcriptomics: Major Potential
The advent of advanced spatial and single-cell transcriptomics, including Xenium and MERSCOPE, presents ATMP CDMOs with a transformative opportunity to expand their service capabilities through improved cellular characterization, high-resolution quality control, and optimized manufacturing processes. These technologies help ensure better reproducibility of complex therapies. By analyzing cellular heterogeneity and tissue interactions, CDMOs can speed up process development and enhance patient-specific modeling. Combining AI with transcriptomics technologies offers a competitive advantage by automating data analysis and predictive modeling, boosting therapy effectiveness, and reducing time to market.
Barriers in Regulation and Standardization: Major Obstacles
The rapid adoption and commercialization of new ATMP technologies are significantly hampered by a fragmented global regulatory landscape, a lack of harmonized guidelines, and scarce standardized reference materials. These challenges increase compliance costs and hinder reproducibility. Inaccurate data interpretation, high capital investments for advanced infrastructure, and a shortage of skilled personnel make scaling up these personalized therapies difficult, especially for smaller CDMOs.
Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/8439
Advanced Therapy Medicinal Products CDMO Market Report Scope
|
Report Attribute |
Details |
|
Market Size in 2026 |
USD 11.12 Billion |
|
Market Size by 2035 |
USD 53.11 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 18.97% |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2035 |
|
Segments Covered |
By Product, By Phase, By Indication |
|
Market Analysis (Terms Used) |
Value (USD Million/Billion) or (Volume/Units) |
|
Report Coverage |
Revenue Forecast, Company Ranking, Competitive Landscape, Growth Factors, and Trends |
|
Key Companies Profiled |
Celonic; Bio Elpida; CGT Catapult; Rentschler Biopharma SE; AGC Biologics; Catalent; Lonza; WuXi Advanced Therapies; BlueReg; Minaris Regenerative Medicine; Patheon |
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344
Advanced Therapy Medicinal Products CDMO Market: Segmentation Analysis
By Product Analysis
In 2025, the gene therapy segment dominated the market, driven by a large and rapidly growing pipeline of clinical trials, increasing regulatory approvals for rare diseases, and the urgent need for complex viral vector manufacturing. Innovations in gene editing, such as CRISPR-Cas9, along with improved non-viral delivery systems, have broadened potential therapeutic applications, leading to an increase in the number of projects managed by CDMOs. Gene therapies depend on complex viral vector manufacturing, which necessitates high-level technical expertise that biotechnology companies often outsource to CDMOs.
The cell therapy segment is expected to experience the fastest growth during the forecast period, primarily due to the rapid expansion of CAR-T cell therapies for cancer. There are a surging pipeline of clinical trials and a notable shift toward allogeneic off-the-shelf manufacturing. The number of clinical trials for cell therapies targeting oncology, rare diseases, and autoimmune disorders is growing quickly, necessitating advanced infrastructure for development. The high demand for complex, individualized, and sometimes regenerative therapies is pushing biotech firms to outsource production to CDMOs.
Advanced Therapy Medicinal Products CDMO Market by Product, 2025-2035 (USD Billion)
|
Year |
2025 |
2026 |
2027 |
2028 |
2029 |
2030 |
2031 |
2032 |
2033 |
2034 |
2035 |
|
Gene Therapy |
3.35 |
3.97 |
4.67 |
5.53 |
6.52 |
7.71 |
9.11 |
10.76 |
12.73 |
15.02 |
17.75 |
|
Cell Therapy |
2.98 |
3.55 |
4.24 |
5.05 |
6.03 |
7.19 |
8.57 |
10.22 |
12.18 |
14.53 |
17.32 |
|
Tissue Engineered |
0.75 |
0.9 |
1.1 |
1.33 |
1.61 |
1.95 |
2.36 |
2.86 |
3.46 |
4.19 |
5.07 |
|
Others |
0.37 |
0.44 |
0.53 |
0.63 |
0.75 |
0.89 |
1.06 |
1.26 |
1.49 |
1.78 |
2.11 |
By Phase Analysis
In 2025, the Phase II segment led the market, primarily because it handles the highest volume of ongoing trials and requires critical, complex Good Manufacturing Practice (GMP) production for intermediate efficacy trials, which demands the expertise of specialized CDMOs. The growing popularity of cell and gene therapies, coupled with high success rates from Phase I trials, is driving significant funding into this segment. CDMOs in this phase specialize in enhancing product consistency, scalability, and in preparing documentation for potential commercialization.
The Phase III segment is predicted to expand at a notable CAGR. This growth is mainly driven by an increase in late-stage clinical trials for gene and cell therapies transitioning to commercialization, which demands massive process validation, large-scale viral vector production, and stringent quality assurance. As more advanced therapy medicinal products (ATMPs) move into Phase III, CDMOs will be tasked with managing pivotal, large-scale studies and preparing for product launches.
Advanced Therapy Medicinal Products CDMO Market by Phase, 2025-2035 (USD Billion)
|
Year |
2025 |
2026 |
2027 |
2028 |
2029 |
2030 |
2031 |
2032 |
2033 |
2034 |
2035 |
|
Phase I |
1.64 |
1.89 |
2.17 |
2.5 |
2.86 |
3.28 |
3.76 |
4.29 |
4.9 |
5.58 |
6.34 |
|
Phase II |
2.82 |
3.32 |
3.9 |
4.57 |
5.37 |
6.29 |
7.38 |
8.66 |
10.15 |
11.9 |
13.94 |
|
Phase III |
2.24 |
2.7 |
3.27 |
3.95 |
4.77 |
5.77 |
6.96 |
8.41 |
10.15 |
12.25 |
14.79 |
|
Phase IV |
0.75 |
0.95 |
1.2 |
1.52 |
1.91 |
2.4 |
3 |
3.74 |
4.66 |
5.79 |
7.18 |
By Indication Analysis
The oncology segment dominated the market in 2025, primarily due to the high demand for CAR-T cell therapies, gene-modified cell therapies, and oncolytic viruses. This dominance is driven by increased investment in cancer research and development, personalized medicine initiatives, and the need for specialized manufacturing capacity. To meet the growing global demand for targeted cancer treatments, CDMOs are increasingly relied upon for their specialized facilities and technical expertise, offering faster development timelines and high-precision outsourced production.
The segment focused on neurological and genetic disorders is expected to grow at the fastest CAGR during the forecast period. This growth is attributed to a rapid increase in clinical trials for gene therapies, the demand for specialized vector design, and significant unmet medical needs for rare. Due to the complexity and high costs associated with developing in-house infrastructure for these advanced and often curative therapies, pharmaceutical companies are heavily partnering with specialized ATMP CDMOs with rapid apid advancements in gene editing technologies and viral vector development.
Advanced Therapy Medicinal Products CDMO Market by Indication, 2025-2035 (USD Billion)
|
Year |
2025 |
2026 |
2027 |
2028 |
2029 |
2030 |
2031 |
2032 |
2033 |
2034 |
2035 |
|
Oncology |
2.61 |
3.08 |
3.63 |
4.28 |
5.05 |
5.95 |
7 |
8.25 |
9.74 |
11.45 |
13.53 |
|
Cardiology |
0.52 |
0.62 |
0.74 |
0.88 |
1.04 |
1.24 |
1.48 |
1.76 |
2.09 |
2.49 |
2.96 |
|
Central nervous system |
0.67 |
0.8 |
0.95 |
1.13 |
1.34 |
1.6 |
1.9 |
2.26 |
2.69 |
3.2 |
3.8 |
|
Musculoskeletal |
0.45 |
0.53 |
0.63 |
0.75 |
0.89 |
1.06 |
1.27 |
1.51 |
1.79 |
2.13 |
2.53 |
|
Infectious disease |
0.6 |
0.7 |
0.82 |
0.97 |
1.13 |
1.33 |
1.56 |
1.83 |
2.15 |
2.52 |
2.96 |
|
Dermatology |
0.3 |
0.35 |
0.42 |
0.5 |
0.6 |
0.71 |
0.84 |
1 |
1.19 |
1.42 |
1.69 |
|
Endocrine, metabolic, genetic |
0.52 |
0.64 |
0.78 |
0.95 |
1.16 |
1.42 |
1.73 |
2.11 |
2.57 |
3.13 |
3.8 |
|
Immunology & inflammation |
0.52 |
0.64 |
0.78 |
0.95 |
1.16 |
1.42 |
1.73 |
2.11 |
2.57 |
3.13 |
3.8 |
|
Ophthalmology |
0.45 |
0.54 |
0.65 |
0.79 |
0.95 |
1.15 |
1.39 |
1.68 |
2.03 |
2.45 |
2.96 |
|
Hematology |
0.37 |
0.44 |
0.53 |
0.63 |
0.75 |
0.89 |
1.06 |
1.26 |
1.49 |
1.78 |
2.11 |
|
Gastroenterology |
0.22 |
0.27 |
0.34 |
0.41 |
0.51 |
0.62 |
0.76 |
0.93 |
1.13 |
1.39 |
1.69 |
|
Others |
0.22 |
0.25 |
0.27 |
0.3 |
0.33 |
0.35 |
0.38 |
0.4 |
0.42 |
0.43 |
0.42 |
By Regional Insights
North America led the market in 2025. This leadership is mainly due to high investment in research and development, strong biopharma infrastructure, and a boom in clinical trials for cell and gene therapies. A notable rise in Investigational New Drug (IND) applications for therapies in oncology and rare diseases has created high demand for specialized, scalable, and compliant manufacturing facilities. Strict but clear FDA guidelines foster a stable environment for investment and commercialization, and high outsourcing rates from biotech startups further strengthen this regional leadership.
With a dense biotech network and faster FDA approvals for therapies like CAR-T, the U.S. maintains a dominant position globally. Heavy investment in automated, large-capacity viral vector manufacturing solidifies the U.S. as the top hub for translating advanced, complex cell and gene therapies from lab to market.
· In March 2026, Agilent Technologies launched Agilent Advanced Therapeutics, a unified CDMO solution integrating BIOVECTRA and Nucleic Acid Solutions across North America to offer comprehensive, end-to-end manufacturing for life-changing therapies.
Canada is quickly establishing itself as an affordable extension of the U.S. market, combining specialized expertise in high-growth cell and gene therapies with strict FDA cGMP standards. Canadian CDMOs provide seamless, high-quality analytical and manufacturing solutions for U.S. biotech sponsors, supporting faster, compliant innovation within North America.
· In March 2025, Merck began construction on a $1 billion, 470,000-square-foot biologics center of excellence in Wilmington, Delaware, aimed at boosting U.S. manufacturing of next-generation therapies, including antibody-drug conjugates (ADCs) and Keytruda, and creating thousands of high-paying jobs.
Asia Pacific is expected to experience the fastest growth during the forecast period. This rapid expansion results from increasing demand for personalized medicine, rising research and development investments, and a strategic move toward regionalizing supply chains. Governments in China, Japan, and South Korea are actively supporting the sector through incentives, subsidies, and faster regulatory pathways for advanced therapies. The high number of biotech firms in China and emerging hubs in Singapore and India focusing on orphan diseases and cancer fuels demand for external manufacturing capacity.
India is becoming a key hub, offering cost-effective, FDA-compliant services to Western companies looking to diversify supply chains through the China+1 strategy. Building on its vaccine manufacturing expertise, India's market is poised for significant growth, shifting toward a complex biologics and advanced cell and gene therapies landscape.
· In December 2025, India’s Bulk Drug Parks are game-changers, transforming the nation from an import-dependent pharmacy to a self-reliant powerhouse by creating world-class, cost-effective API manufacturing hubs that fuel local CDMO growth.
China has become the leading low-cost, high-speed partner for global biotech, largely due to rapid regulatory reforms and significant innovation. By transitioning from a follower to an innovator with greater diversity in research targets and improved regulatory capacity at the CDE, China is establishing itself as an essential, integrated ecosystem for advanced therapy medicinal products.
· In October 2025, China’s NMPA is fast-tracking the future of medicine by slashing red tape and accelerating clinical trials for advanced therapies. These sweeping regulatory reforms foster homegrown innovation, align with global standards, and promise rapid patient access to life-saving treatments.
Advanced Therapy Medicinal Products CDMO Market by Region, 2025-2035 (USD Billion)
|
Year |
2025 |
2026 |
2027 |
2028 |
2029 |
2030 |
2031 |
2032 |
2033 |
2034 |
2035 |
|
North America |
2.84 |
3.33 |
3.91 |
4.61 |
5.42 |
6.39 |
7.51 |
8.83 |
10.39 |
12.21 |
14.37 |
|
Europe |
2.38 |
2.81 |
3.31 |
3.9 |
4.59 |
5.41 |
6.37 |
7.51 |
8.84 |
10.41 |
12.25 |
|
Asia-Pacific |
1.64 |
2.02 |
2.49 |
3.06 |
3.76 |
4.61 |
5.66 |
6.93 |
8.48 |
10.37 |
12.67 |
|
Latin America |
0.37 |
0.43 |
0.51 |
0.59 |
0.69 |
0.8 |
0.93 |
1.08 |
1.25 |
1.46 |
1.69 |
|
Middle East & Africa (MEA) |
0.22 |
0.27 |
0.32 |
0.38 |
0.45 |
0.53 |
0.63 |
0.75 |
0.9 |
1.07 |
1.27 |
Advanced Therapy Medicinal Products CDMO Market Companies
· Celonic GmbH
· Bio Elpida
· Cell and Gene Therapy Catapult (CGT Catapult)
· Rentschler Biopharma SE
· AGC Biologics
· Catalent, Inc.
· Lonza Group Ltd.
· WuXi Advanced Therapies (WuXi AppTec Group)
· BlueReg Group
· Minaris Regenerative Medicine
Pervasive Adoption of Cell and Gene Therapies in Modern Medicine
|
Company |
Latest Updates |
|
Sumitomo Chemical (August 2025) |
Announced expansion of cell therapy manufacturing capacity in Japan, including a fourth regenerative medicine facility, supported by the 2024 supplementary budget from METI. |
|
Vector BioMed (June 2025) |
Partnered with Kailash Cancer Hospital and Research Center in India to bring CAR T-Cell therapy to rural areas, expanding access and local production. |
|
Lonza (Feb 2025) |
Expanded cell and gene therapy manufacturing capacity in the U.S. to meet growing demand for late-stage and commercial CGT programs. |
|
Catalent (Jan 2025) |
Partnered with Galapagos NV to support the decentralized manufacturing of CAR-T therapy GLPG5101, designed to enhance patient access to treatment. |
Recent Developments
· In May 2025, Altaris combined Minaris Regenerative Medicine and WuXi Advanced Therapies’ U.S. and U.K. operations to create Minaris Advanced Therapies™, a global cell therapy CDMO headquartered in Philadelphia. The company manufactures two commercial cell therapies and tests over 27 products. With more than 1,400 experts, it offers advanced platforms for cell therapies and viral vectors and aims to streamline the commercialization of these therapies.
· In January 2025, Immuneel Therapeutics launched Qartemi, a CAR T-cell therapy for adult B-cell Non-Hodgkin Lymphoma (B-NHL) in India. This personalized therapy addresses a crucial need as the country faces over 120,000 new blood cancer cases yearly. Co-founder Kiran Mazumdar-Shaw emphasized their goal of providing accessible, innovative cancer treatments.
· In March 2026, Indena announced its expanded capabilities in Highly Potent Active Pharmaceutical Ingredients (HPAPIs) and Antibody-Drug Conjugates (ADCs) at DCAT Week 2026. They produce multiple commercial HPAPIs and integrate advanced technologies to meet the rising demand in oncology. Global CDMO Director Bernard Vianes highlighted Indena's commitment to innovation and safety, achieving low occupational exposure levels in HPAPI manufacturing.
Related Report –
⬥︎ Active Pharmaceutical Ingredients CDMO Market - The global active pharmaceutical ingredients CDMO market size was valued at USD 139.33 billion in 2025 and is anticipated to reach around USD 292.79 billion by 2035, growing at a CAGR of 7.71% from 2026 to 2035.
⬥︎ Biopharmaceutical CDMO Market- The global biopharmaceutical CDMO market was valued at USD 21.15 billion in 2024 and is projected to hit around USD 49.61 billion by 2034, expanding at a CAGR of 8.9% during the forecast period of 2025 to 2034.
⬥︎ Biologics CDMO Market- The global biologics CDMO market size is calculated at USD 22.45 billion in 2024, grows to USD 25.92 billion in 2025, and is projected to reach around USD 94.60 billion by 2034, growing at a solid CAGR of 15.47% from 2025 to 2034.
⬥︎ Oligonucleotide CDMO Market- The global oligonucleotide CDMO market size is calculated at USD 3.15 billion in 2024, grows to USD 3.84 billion in 2025, and is projected to reach around USD 22.73 billion by 2034, at a compound annual growth rate (CAGR) of 21.85% from 2024 to 2034.
⬥︎ Cell and Gene Therapy CDMO Market- The global cell and gene therapy CDMO market size was estimated at USD 10.35 billion in 2025 and is projected to hit around USD 125.09 billion by 2035, growing at a CAGR of 28.3% during the forecast period from 2026 to 2035.
⬥︎ U.S. Active Pharmaceutical Ingredients CDMO Market- The U.S. active pharmaceutical ingredients CDMO market size is calculated at USD 25.85 billion in 2024, grows to USD 27.22 billion in 2025, and is projected to reach around USD 43.33 billion by 2034, growing at a CAGR of 5.3% from 2025 to 2034.
⬥︎ North America Topical Drugs CDMO Market- The North America topical drugs CDMO market size was exhibited at USD 15.25 billion in 2024 and is projected to hit around USD 42.87 billion by 2034, growing at a CAGR of 10.89% during the forecast period 2025 to 2034.
⬥︎ Topical Drugs CDMO Market- The topical drugs CDMO market size was exhibited at USD 46.15 billion in 2025 and is projected to hit around USD 137.06 billion by 2035, growing at a CAGR of 11.31% during the forecast period 2026 to 2035.
⬥︎ U.S. Pharmaceutical CDMO Market- The U.S. pharmaceutical CDMO market size was exhibited at USD 43.7 billion in 2025 and is projected to hit around USD 89.45 billion by 2035, growing at a CAGR of 7.43% during the forecast period 2026 to 2035.
⬥︎ Veterinary CRO and CDMO Market- The Veterinary CRO and CDMO market size was exhibited at USD 7.15 billion in 2024 and is projected to hit around USD 17.56 billion by 2034, growing at a CAGR of 9.4% during the forecast period 2025 to 2034.
⬥︎ U.S. Small Molecule Innovator API CDMO Market - The U.S. small molecule innovator API CDMO market size was exhibited at USD 9.85 billion in 2025 and is projected to hit around USD 17.96 billion by 2035, growing at a CAGR of 6.19% during the forecast period 2026 to 2035.
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the Advanced Therapy Medicinal Products CDMO market.
By Product
· Gene Therapy
· Cell Therapy
· Tissue Engineered
· Others
By Phase
· Phase I
· Phase II
· Phase III
· Phase IV
By Indication
· Oncology
· Cardiology
· Central nervous system
· Musculoskeletal
· Infectious disease
· Dermatology
· Endocrine, metabolic, genetic
· Immunology & inflammation
· Ophthalmology
· Hematology
· Gastroenterology
· Others
By Region
· North America
· Europe
· Asia-Pacific
· Latin America
· Middle East & Africa (MEA)
Immediate Delivery Available | Buy This Premium Research: https://www.novaoneadvisor.com/report/checkout/8439
Nova One Advisor is a global leader in market intelligence and strategic consulting, committed to delivering deep, data-driven insights that power innovation and transformation across industries. With a sharp focus on the evolving landscape of life sciences, we specialize in navigating the complexities of cell and gene therapy, drug development, and the oncology market, enabling our clients to lead in some of the most revolutionary and high-impact areas of healthcare.
Our expertise spans the entire biotech and pharmaceutical value chain, empowering startups, global enterprises, investors, and research institutions that are pioneering the next generation of therapies in regenerative medicine, oncology, and precision medicine.
Web: https://www.novaoneadvisor.com/
Contact Us
USA: +1 804 420 9370
Email: sales@novaoneadvisor.com
For Latest Update Follow Us: LinkedIn
Towards Healthcare | Towards Packaging | Towards Automotive | Towards Chem and Materials | Towards FnB | Towards Consumer Goods | Statifacts | Towards EV Solutions | Towards Dental | Market Stats Insight | Nutraceuticals Func Foods | Onco Quant | Sustainability Quant | Specialty Chemicals Analytics


