FDA
With CBER director Vinay Prasad set to depart the agency at the end of the month, a coalition of patient groups and biotech executives penned a letter imploring the Trump administration to “restore regulatory clarity” for rare disease therapies. Experts on a BioSpace panel last week also acknowledged the challenges faced by a more stringent FDA.
FEATURED STORIES
Draft guidance, issued by the FDA last week, could remove ambiguity and uncertainty that may have so far limited uptake of new approach methodologies, experts told BioSpace, particularly emphasizing the agency’s recommendations around defining NAMs’ regulatory purpose.
The FDA’s Commissioner’s National Priority Voucher program, unveiled in June 2025, is “shrouded in secrecy,” Democratic representative Jake Auchincloss said last month, as regulatory and biopharma leaders try to decode the criteria for investigational or approved drugs to receive a voucher.
Although FDA Commissioner Marty Makary promised “an exciting treatment” for autism, what the agency delivered was a label expansion for leucovorin to treat the ultrarare cerebral folate deficiency. The regulatory process, which relied on a literature review rather than new evidence, stands in contrast to recent rare disease rejections in which the FDA cited a need for more rigorous evidence.
Subscribe to ClinicaSpace
Clinical trial results, research news, the latest in cancer and cell and gene therapy, in your inbox every Monday
THE LATEST
The antibody-drug conjugate, withdrawn from the market by GSK in 2022 after failing a confirmatory study, was approved Thursday by the FDA for previously treated patients with multiple myeloma. This is despite a negative advisory committee vote in July.
The company’s technology, a modified herpes simplex virus used to deliver gene therapies, was given the FDA’s new designation based on its approved topical skin cream. What this will mean for Krystal’s still-in-development eye drop is unclear.
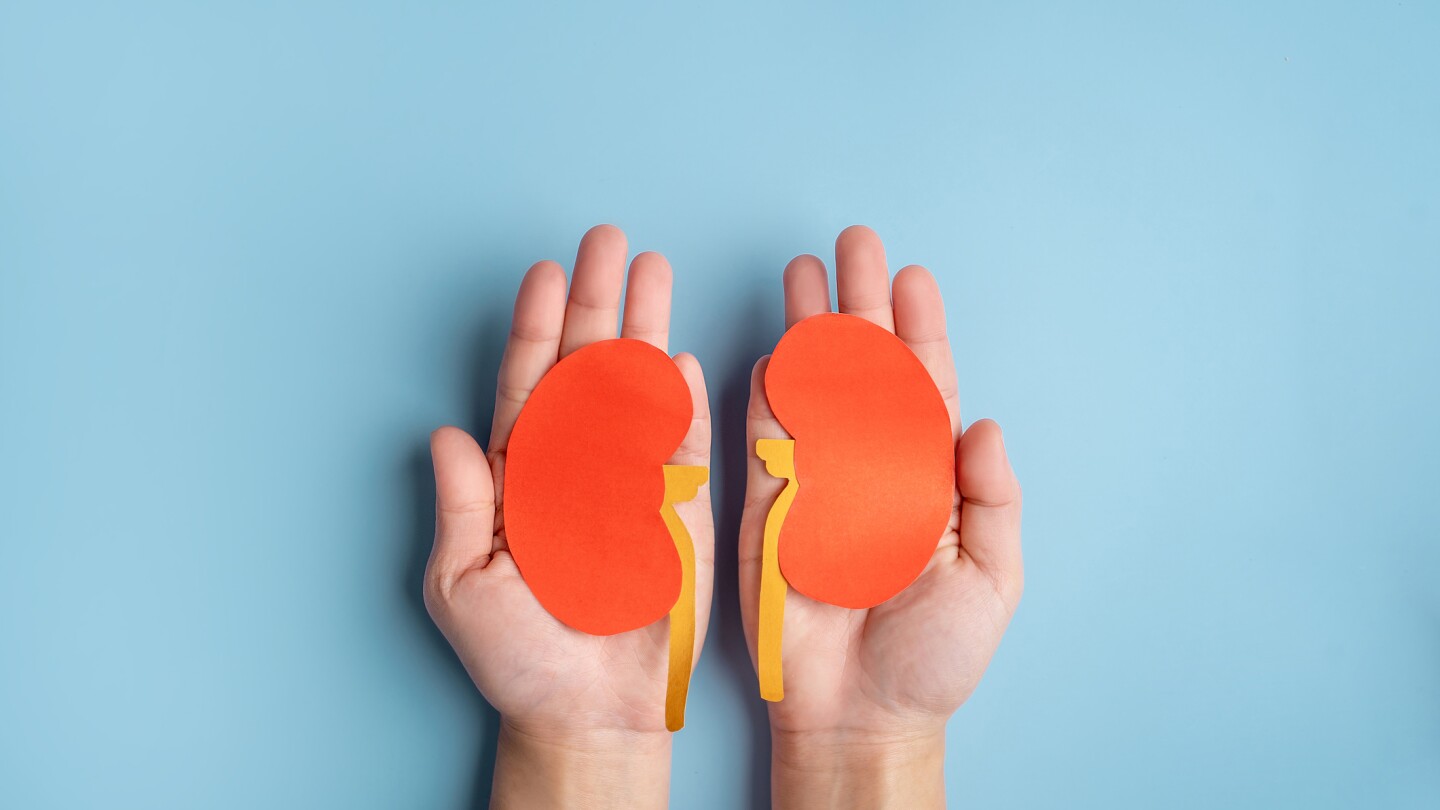
The anti-CD20 antibody, developed by Roche’s Genentech along with Biogen and already approved for multiple oncology indications, is branching into autoimmune disease. Its B cell–targeting mechanism of action gives it activity in improving kidney conditions in patients with lupus nephritis.
Last month, investors’ hopes were dashed as Replimune suggested alignment had not yet been reached with the FDA on RP1’s future. Now, the regulator has accepted a refiled application after all.
Rybelsus can now be used as a primary or secondary prevention pill to lower the risk of major adverse cardiovascular events in at-risk patients with type 2 diabetes.
Glaukos’ Epioxa allows cross-linking therapy for keratoconus without requiring an incision. The biotech expects to launch the drug early next year.
After a chaotic year that has seen the attrition of over half the FDA’s senior leadership, many of these individuals have landed new roles—at Eli Lilly, Pfizer, Iovance and more. The FDA’s loss, it seems, is largely the pharmaceutical industry’s gain.
The Commissioner’s National Priority Voucher awards companies that align with specific national priorities—such as boosting domestic manufacturing and lowering drug prices—with faster reviews and more frequent interactions with the FDA.
During this webinar BioSpace sits down with former FDA Chief Information Officer Vid Desai to discuss the potential advantages and pitfalls of deregulation stemming from one of President Donald Trump’s first executive orders. Together, they uncover what this shift means for drugmakers, compliance and patient safety.
After the FDA revoked Sarepta Therapeutics’ platform designation in July, Krystal Biotech is “poised to be the first major beneficiary” of this program, according to analysts at William Blair.