BD (Becton, Dickinson and Company), a leading global medical technology company, is voluntarily recalling specified lots of the ChloraPrep™ Hi-Lite Orange™ 26 mL Applicator (2% w/w chlorhexidine gluconate (CHG) and 70% v/v isopropyl alcohol (IPA)) to the user level due to a defective applicator.
|
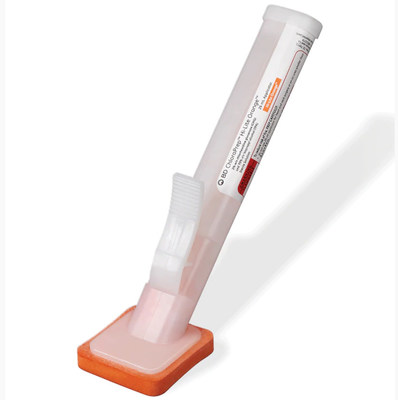
FRANKLIN LAKES, N.J., April 20, 2021 /PRNewswire/ -- BD (Becton, Dickinson and Company), a leading global medical technology company, is voluntarily recalling specified lots of the ChloraPrep™ Hi-Lite Orange™ 26 mL Applicator (2% w/w chlorhexidine gluconate (CHG) and 70% v/v isopropyl alcohol (IPA)) to the user level due to a defective applicator. The product is used as an antiseptic for the preparation of the patient's skin prior to surgery to help reduce bacteria that potentially can cause skin infection1. The ChloraPrep™ Hi-Lite Orange™ 26 mL Applicator contains glass ampules that house the sterile ChloraPrep™ solution. In normal circumstances, the product is activated by squeezing the wing on the applicator to break the ampule, which releases the solution to the sponge head for application to the patient's skin. In certain lots, the applicator end cap was improperly secured due to a manufacturing error. This can result in broken glass and solution dropping out of the applicator once activated. In some cases, the glass ampules can drop out before activation and shatter if striking a hard surface, resulting in solution and glass fragments scattering in the procedure area and potentially causing injury to patients and health care professionals. These products were distributed in the United States and Puerto Rico. BD Risk Assessment As part of the voluntary recall to the user level, the company is notifying customers and distributors affected by the recall. BD's Customer Recall Notification provides instructions to customers and distributors for disposal and replacement of the impacted ChloraPrep™ Hi-Lite Orange™ 26 mL Applicator (see list of affected lot numbers below).
Customer inquiries related to this recall, as well as adverse reaction/events experienced with the product should be addressed to BD Customer Support at 1-844-8BD- LIFE (1-844-823-5433); When calling Monday- Friday between the hours of 9 a.m. to 6 p.m. ET, say "Recall" when prompted. For additional information, customers can visit www.bd-chloraprep-action.com. Consumers should contact their physician or healthcare provider if they have experienced any problems that may be related to using this drug product. The U.S. Food and Drug Administration (FDA) has been notified of this recall. FDA MedWatch Reporting
About BD
____________________
SOURCE BD (Becton, Dickinson and Company) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
Company Codes: NYSE:BDX |
||||||||||||||||||||||||||||||||||||||||||||||||||||||