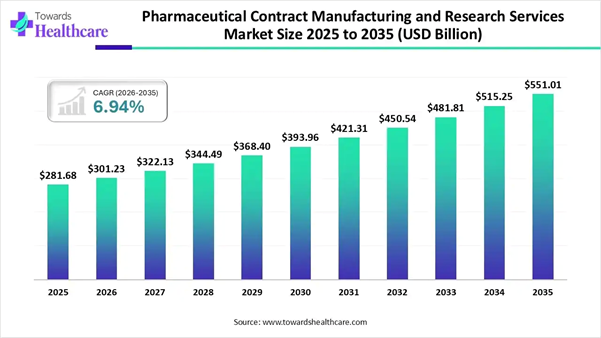
The global pharmaceutical contract manufacturing and research services market size was estimated at USD 281.68 billion in 2025 and is predicted to increase from USD 301.23 billion in 2026 to approximately USD 551.01 billion by 2035, expanding at a CAGR of 6.94% from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6697
The contract manufacturing and research services offered by the leading players, like Sun Pharmaceuticals, include high-end chemical process development, process engineering, impurity profiling, process validation and commercial manufacturing, and many others. The major players are committed to state-of-the-art research, regulatory and intellectual property expertise, and global manufacturing capabilities. The pharmaceutical companies rely on a third party for the production of drugs, which raises the trend of pharmaceutical contract manufacturing. It allows flexibility and cost savings, and provides services such as manufacturing, drug development, and compliance with regulatory requirements.
Market Highlights
• The pharmaceutical contract manufacturing and research services market will likely exceed USD 301.23 billion by 2026.
• Valuation is projected to hit USD 551.01 billion by 2035.
• Estimated to grow at a CAGR of 6.94% starting from 2026 to 2035.
• Asia Pacific dominated the global pharmaceutical contract manufacturing and research services market by 45% share in 2025.
• North America is expected to be the fastest-growing region between 2026 and 2035.
• By service type, the manufacturing segment held the largest revenue share of the market in 2025.
• By service type, the research segment is expected to witness the fastest growth during the forecast period.
• By end-user, the big pharma segment dominated the market in 2025.
• By end-user, the contract research organizations segment is expected to be the fastest growing during the forecast period.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Market Overview
The Expanded Development of High-Quality Products
The pharmaceutical contract manufacturing and research services market offers benefits in terms of increased production capacity, cost reduction, and the potential to enhance core activities like drug discovery and marketing. The contract development and manufacturing organizations (CDMOs) and contract manufacturing organizations (CMOs) strictly adhere to good manufacturing practices (GMP) of the U.S. FDA and meet compliance with regulatory requirements. The pharmaceutical contract manufacturing process of any drug undergoes blending, coating, serialization, and shipment stages, where contract manufacturers strictly adhere to the U.S. FDA guidelines. They ensure safe, timely, and efficient delivery of pharmaceutical products.
Major Challenges in Third-Party Pharma Manufacturing
The potential issues arising in the pharmaceutical contract manufacturing and research services market are associated with quality control, regulatory compliance, supply chain disruptions, intellectual property risks, cost overruns, lack of flexibility and scalability, and communication barriers. A poor quality of products lowers the manufacturing status of companies and greatly impacts the company’s revenue success. It results in product recalls, unsatisfied customer responses, and regulatory fines. Strong monitoring and great communication are the key solutions to resolving these issues.
Opportunities in Third-Party Pharma Manufacturing
The pharmaceutical contract manufacturing and research services market holds many fruitful opportunities to mark its global footprint. The quality control undergoes assurance through quality checks, regular audits, and compliance assessments to ensure the entry of final products into the market with set standards. There are exclusive platforms available for companies to strongly connect with third-party pharma manufacturing companies. Companies need to ensure the regulatory alignment of third-party pharma manufacturing companies with good manufacturing practices (GMP), the U.S. Food and Drug Administration (FDA), and other country-specific laws. The changes in legislation and compliance assurance are achieved through regular regulatory training and communication with third-party medicine manufacturers.
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
Resilience in Supply Chain and Strong Legal Agreements
The establishment of resilience in the supply chain reduces the threat of supply chain disruptions, which can result from increased production of several raw materials from multiple suppliers. A continuous supply chain is enabled by a good network of logistics partners and an effective supply network.
The pharmaceutical companies need to keep the safety of their intellectual properties by providing third-party pharma manufacturing partners with robust legal agreements. The examples of such agreements include patents and detailed contracts that protect confidential information from being stolen or misused by third-party manufacturers. Additionally, regular auditing ensures that third-party manufacturers strictly adhere to these agreements and intellectual property rules.
Transparent Pricing, Clear Contracts, and Strong Communication
The pharmaceutical companies need to enter into clear and detailed contracts to ensure transparent pricing of every aspect of manufacturing. The key aspects related to the cost of labor, raw materials, packaging, and logistics must be specified in contracts to avoid unexpected increases in prices. A periodic review of the costs and informed decision-making with third-party manufacturers is essential to keep the project within budget.
Regional Analysis
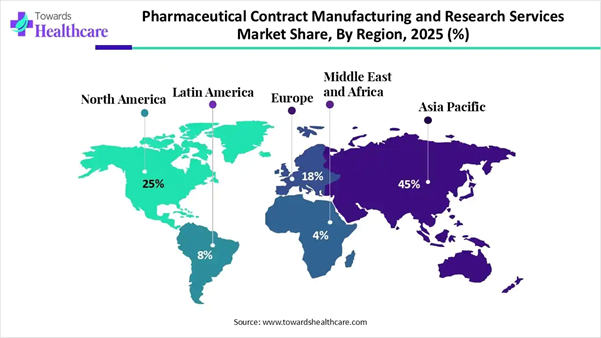
Asia Pacific dominated the pharmaceutical contract manufacturing and research services market by 45% share in 2025, owing to cost and operational efficiency, investments in capacity and infrastructure, and strategic government support. The regional industries are transitioning from low-cost generic manufacturing to high-value research and innovation. The industries are adopting smart and advanced technologies and services offered by CMOs and CDMOs to expand their global footprint in R&D, supply chain, commercialization, and regulatory framework.
India Market Analysis
The pharmaceutical contract manufacturing and research services market in India is witnessing a surge in government incentives and investments in high-value products like APIs, complex generics, and biosimilars in the coming years. The government mandated a new GMP schedule to align Indian facilities with global standards, which makes them more competitive for international contract manufacturing.
North America is expected to grow at the fastest rate in the pharmaceutical contract manufacturing and research services market during the forecast period due to regulatory support for domestic manufacturing that boosts local production capacity. There is an increased focus on mRNA therapies, biologics, and the integration of AI for real-time quality control and predictive maintenance. The government programs aim to streamline the regulatory review process for new domestic manufacturing facilities.
U.S. Market Trends
The pharmaceutical contract manufacturing and research services market in the U.S. is experiencing a shift from traditional batch processing to continuous manufacturing, which improves efficiency and reduces the long-term cost of goods. The U.S. government launched several initiatives to speed up the construction of domestic facilities and reduce dependency on foreign active pharmaceutical ingredients.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
Segmentation Analysis
Service Type Insights
The manufacturing segment dominated the pharmaceutical contract manufacturing and research services market in 2025, owing to the key stages such as research and development, formulation, production, quality control, packaging, and distribution. The R&D stage allows scientists to conduct tests, perform clinical trials, and develop new drugs. The pharmaceutical companies make efforts for the manufacturing of drugs in bulk by ensuring consistency and quality.
The research segment is expected to witness the fastest growth in the pharmaceutical contract manufacturing and research services market during the forecast period due to the importance of various stages, including the identification of a potential target, assay development, screening methods, and lead optimization. R&D services offered by pharmaceutical companies generate income for them and save lives. The technical and manufacturing procedures, quality control measures, and production processes drive the expansion of reliable R&D services.
End User Insights
The big pharma segment dominated the pharmaceutical contract manufacturing and research services market in 2025, owing to the major role of big pharma in creating a safe and affordable treatment. The big pharmaceutical companies specialize in the development, production, and sale of medical products and pharmaceutical drugs. They conduct a lot of scientific research to produce a drug or a medicinal product, and are involved in the complete lifecycle of a drug.
The contract research organizations segment is expected to be the fastest-growing in the pharmaceutical contract manufacturing and research services market during the forecast period due to their assistance to the pharmaceutical, biotechnology, and medical devices industry in research and development. The CROs provide them with research services in an outsourced form on a contract basis, which include clinical development, biopharmaceutical development, pharmacovigilance, and clinical trial management. They contribute to streamlining the drug development process by simplifying the entry to the drug market and providing specialized expertise and resources.
Access our comprehensive healthcare dashboard for detailed market insights, segment breakdowns, regional performance, and company profiles: https://www.towardshealthcare.com/access-dashboard
Top Companies in the Market

• Lonza Group
• Thermo Fisher Scientific
• Catalent
• WuXi AppTec
• IQVIA
• Siegfried Holding
• Boehringer Ingelheim
• Recipharm
• Samsung Biologics
• Eurofins Scientific
Segments Covered in the Report
By Service
• Manufacturing
• API/Bulk Drugs
• Advanced Drug Delivery Formulations Packaging
• Packaging
• Finished Dose Formulations
• Solid Formulations
• Liquid Formulations
• Semi-solid Formulations
• Research
• Oncology
• Vaccines
• Inflammation & Immunology
• Cardiology
• Neuroscience
• Others
By End-User
• Big Pharma
• Small Pharma
• Generic Pharma
• Contract Research Organizations
By Region
• North America
• South America
• Europe
• Asia Pacific
• MEA
Immediate Delivery Available | Buy This Research Report Now @ https://www.towardshealthcare.com/checkout/6697
About Us
Towards Healthcare Research & Consulting is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region: +44 778 256 0738
North America Region: +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Our Trusted Data Partners
Precedence Research | Towards Packaging | Towards Food and Beverages | Towards Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Browse More Insights of Towards Healthcare:
➡️ The global IVD contract manufacturing market stands at US$ 23.34 billion in 2025 and is projected to reach US$ 62.99 billion by 2035, expanding at a CAGR of 10.44% (2026–2035).
➡️ The global biosimilar contract manufacturing market is estimated at US$ 9.22 billion in 2025 and is expected to climb to US$ 52.23 billion by 2035, registering a CAGR of 18.94% (2026–2035).
➡️ The global oral solid dosage contract manufacturing market was valued at USD 41.08 billion in 2025 and is forecast to grow to USD 75.26 billion by 2035, advancing at a CAGR of 6.24% (2026–2035).
➡️ The global generic pharmaceuticals contract manufacturing market accounts for US$ 81.24 billion in 2025 and is anticipated to reach US$ 143.22 billion by 2035, progressing at a CAGR of 5.85% (2026–2035).
➡️ The global medical device contract manufacturing market reached USD 87.15 billion in 2025 and is predicted to surge to USD 244.34 billion by 2035, at a CAGR of 10.86% (2026–2035).
➡️ The global biopharmaceutical contract manufacturing market totaled US$ 44.61 billion in 2025 and is set to hit US$ 115.65 billion by 2034, rising at a CAGR of 11.14% (2025–2034).
➡️ The global contract pharmaceutical manufacturing market generated USD 232.32 billion in 2025 and is likely to grow to USD 599.3 billion by 2035, reflecting a CAGR of 9.94% (2026–2035).
➡️ The contract GMP manufacturing market is witnessing robust expansion, with revenues projected to reach several hundred million dollars by 2035.
➡️ The global biologics CRO market recorded USD 31.15 billion in 2024 and is projected to rise to USD 106.28 billion by 2034, at a CAGR of 13.04% (2025–2034).
➡️ The global healthcare CRO market held USD 53.87 billion in 2024 and is forecast to increase to USD 106.25 billion by 2034, growing at a CAGR of 7.04% (2025–2034).
➡️ The AAV manufacturing services market reached US$ 1.44 billion in 2025 and is expected to expand to US$ 6.25 billion by 2035, with a CAGR of 15.84% (2026–2035).
➡️ The global viral vector manufacturing market is pegged at USD 1.82 billion in 2025 and is projected to escalate to USD 12.91 billion by 2035, posting a CAGR of 21.64%.
➡️ The APAC viral vector and plasmid DNA manufacturing market is valued at USD 1.68 billion in 2025 and is forecast to grow to USD 12.22 billion by 2035, at a CAGR of 21.93% (2026–2035).
➡️ The global cell therapy manufacturing market is calculated at USD 6.12 billion in 2025 and is likely to reach USD 23.27 billion by 2034, growing at a CAGR of 16% (2025–2034).
➡️ The global cell and gene therapy manufacturing isolators market is estimated at USD 140.16 million in 2025 and is projected to reach USD 321.3 million by 2035, expanding at a CAGR of 8.65% (2026–2035).


