Quick Facts
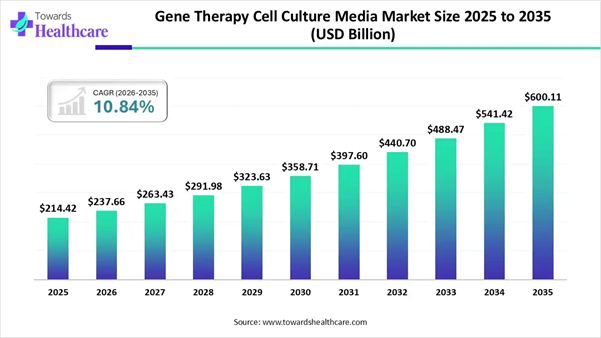
➔Gene therapy cell culture media industry poised to reach USD 237.66 billion by 2026.
➔Forecasted to grow to USD 600.11 billion by 2035.
➔Expected to maintain a CAGR of 10.84% from 2026 to 2035.
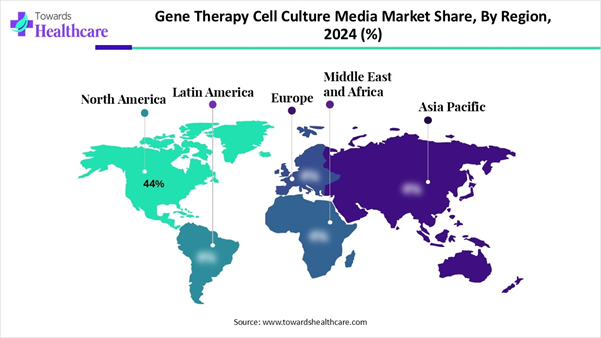
➔North America dominated the gene therapy cell culture media market revenue share of approximately 44% in 2025.
➔Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
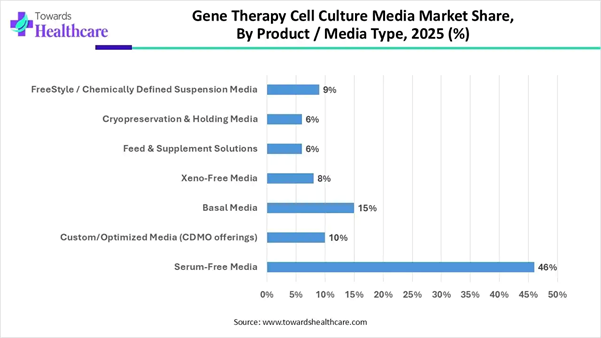
➔By product/media type, the serum-free media segment dominated the market with a revenue share of approximately 46%.
➔By product/media type, the custom/optimized media (CDMO offerings) segment is expected to grow at the fastest CAGR in the market during the forecast period.
➔By application/process stage, the upstream viral vector production segment led the gene therapy cell culture media market with the largest revenue share of approximately 44% in 2025.
➔By application/process stage, the process development & optimization segment is expected to grow at the fastest CAGR in the market during the forecast period.
➔By cell type/target cell line, the HEK293/HEK293T segment held the highest market share of approximately 38% in 2025.
➔By cell type/target cell line, the primary T cells segment is expected to grow at the fastest CAGR in the market during the forecast period.
➔By formulation/format, the liquid ready-to-use media segment held the highest revenue shares of approximately 62% in 2025.
➔By formulation/format, the custom blends segment is expected to grow at the fastest CAGR in the market during the forecast period.
➔By end user, the pharmaceutical & biotechnology companies segment dominated the gene therapy cell culture media market with a major revenue share of approximately 50% in the market in 2025.
➔By end user, the CDMOs/viral vector CMOs segment is expected to grow at the fastest CAGR in the market during the forecast period.
Gene Therapy Cell Culture Media Market Size and Trends for 2026
The global gene therapy cell culture media market size was estimated at USD 214.42 billion in 2025 and is predicted to increase from USD 237.66 billion in 2026 to approximately USD 600.11 billion by 2035, expanding at a CAGR of 10.84% from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6089
The cell culture media are indispensable for cell growth and function due to the presence of necessary cell culture components and an ideal cell culture environment. The formulations vary depending on the need of cell culture conditions, including pH, osmotic pressure, temperature, etc. The different kinds of media used for cell and gene therapy are benchstable media, human plasma-like medium, media with GlutaMAX supplement, cell culture media for bioprocessing, media 199, Ham’s F-12 nutrient mixture, Ham’s F-10 nutrient mixture, and Iscove’s Modified Dulbecco’s Medium (IMDM).
Comprehensive Media Solutions and Services Offered by Sartorius AG
|
Sr. No. |
Cell Therapy Media |
HEK Media |
Viral Vaccine Media |
CHO Media |
Classical Media |
|
|
Immune cell Media |
HEK293 Media |
MDCK Suspension Platform |
CHO Cell Culture Media and Feeds |
Classical Cell Culture Media |
|
|
Stem Cell Media |
Recombinant Human Albumin |
Vero Cell Media |
CHO CLD Services |
Cell Culture Lab Waters & Buffers |
|
|
Preclinical and GMP Growth Factors and Cytokines |
Spent Media Analysis
|
Buffers |
Spent Media Analysis |
Cell Culture Reagents & Supplements |
|
|
Research Use Only Growth Factors and Cytokines |
Custom Media and Buffers |
Custom Media & Buffers |
Custom Media & Buffers |
Ultroser |
|
|
Cell Biopreservation Solutions |
Transfection Reagents |
Microcarriers |
Buffers |
Research Use Only (RUO) Growth Factors & Cytokines |
|
|
Recombinant Human Albumin |
Plasmid Engineering & Sequencing Service |
- |
Transfection Reagents |
NexaGel 3D Cell Culture Matrices |
|
|
Linkit AX Aliquoting System |
Plasmid Manufacturing Service |
- |
Plasmid Engineering & Sequencing Service |
- |
|
|
Spent Media Analysis |
AAV Testing Services |
- |
Plasmid Manufacturing Service |
- |
|
|
Non-Viral Delivery Solutions |
Buffers |
- |
- |
- |
|
|
Microcarriers |
- |
- |
- |
- |
You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Market Overview: Expanding Scientific
Innovations and Deep Research Expertise The gene therapy cell culture media market
is driven by the leading companies, such as Thermo Fisher Scientific Inc.,
which provide media for stem cell culture, organoid, spheroid, and 3D
cell culture, neuronal cell culture, primary cell culture, cell therapy
systems, protein expression media, serum-free media, and media for cytogenetic
research. The products present in the cell culture media line contribute to the
growth and maintenance of a wide range of mammalian cells and cell lines. Become a valued research partner with us
- https://www.towardshealthcare.com/schedule-meeting Gene Therapy Cell Culture Media Market:
Regional Analysis North America led the market share by 44% in
2025, owing to biomanufacturing scale-up, government funding and incentives for
research and development, and expanding clinical pipelines. The government
programs boost clinical trials and harmonise global regulatory
standards. The U.S. FDA aims to advance the rare disease therapeutics program
through clinical trials. The FDA Rare Disease Innovation Hub is responsible for
providing cross-agency expertise to address policy and scientific issues in the
development of rare disease products. For instance, ➔ In
February 2026, Pfizer launched a cost savings program on TrumpRx to lower drug
costs for millions of Americans. This program offers cost savings on over 30
brands related to women’s health, arthritis, migraine, rare diseases, and more.
U.S. Market Analysis The gene therapy cell culture media market
is gaining momentum in the U.S. due to the significant role of the U.S.
government in positioning America as a global
leader in pharmaceutical innovation. It enables patients to pay lower
prices for their prescription medicines. For instance, ➔ In
April 2025, Novartis announced its initiatives to expand its U.S.-based
manufacturing and advance its R&D footprint with a total investment of $23
billion over the next five years. These plans enabled Novartis to expand its
R&D and manufacturing presence across the U.S. with 10 facilities. Asia Pacific is expected to grow at the
fastest rate in the market due to the expansion of biomanufacturing
infrastructure, supportive government policies for biotechnology
innovations, and the rising clinical trial activities. This region
experiences the boom of gene and cell therapy manufacturing, and rapid growth
through biotechnology investments in Japan, China, India, and South Korea. The
investments in cell and gene therapies are made to advance R&D in oncology
and regenerative
medicine. For instance, ➔ In
March 2026, AstraZeneca announced its plans to establish cell therapy
production and supply base in Shanghai, which positions this company as the
first global leading biopharmaceutical company with end-to-end cell therapy
capabilities in China. It has also signed a MoU with the Shanghai Municipal
Science and Technology Commission and leading institutions in scientific
research and financial sectors in the UK. India Market Trends The gene therapy cell culture media market
in India is growing and expanding due to supportive government policies like
the BioE3 policy and Biopharma SHAKTI, cost-effective manufacturing, and a
shift to specialized media to meet international regulatory standards. India
faces the growing challenges of cancer, which makes it rank second globally in
cancer mortality. Research suggests that about 40% of patients relapse or
resist chemotherapy
and other standard treatments for aggressive B-cell malignancies like large
B-cell lymphoma and acute lymphoblastic leukemia. For instance, In January 2026, Cipla partnered with
ImmunoACT to expand access to the World’s first humanised CAR-T cell therapy
named talicabtagene autoleucel (hashtag#NexCAR19) for patients in Africa. This
collaboration aims to address critical unmet needs
in oncology through affordability, innovation, and scientific
excellence. Under this partnership, Cipla will commercialize India’s first CAR-T
cell therapy in Morocco, the Republic of South Africa, and Algeria.
Furthermore, NexCAR19 was honored among the top three innovations in India, in
the presence of India’s Hon’ble Prime Minister, Shri Narendra Modi. Get the latest insights on life science
industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership Gene Therapy Cell Culture Media Market:
Segmentation Analysis Product/Media Type Insights Market Segment 2025 (%) Serum-Free Media 46% Custom/Optimized Media (CDMO
offerings) 10% Basal Media 15% Xeno-Free Media 8% Feed & Supplement
Solutions 6% Cryopreservation &
Holding Media 6% FreeStyle / Chemically
Defined Suspension Media 9%
The serum-free media segment dominated the
market by 46% in 2025, owing to the immense potential of this media to
streamline and optimize CAR-T cell manufacturing workflows. It offers ways to
overcome challenges associated with cost, complexity, and availability of CAR-T
therapies to cancer patients. Researchers test and qualify all the components
used in serum-based expansion media to ensure high levels of sterility and
absence of impurities. The custom/optimized media is expected to
grow at the fastest rate in the gene therapy cell culture media market due to the
use of custom media early in process development to achieve significant savings
in time and resources. The customizable cell culture media are beneficial for
therapeutic development and streamline early-stage discovery. Custom media
services accelerate product timeline to market and provide flexibility and
regulatory support. Application/Process Stage Insights The upstream viral vector production
segment led the market in 2025, owing to the pivotal stages, like cell line
selection, cell line expansion, and the production of viral vectors. The viral
yield is impacted by the productivity and decision-making in upstream
processing. The choice of media and transfection methods determines the
productivity in upstream processing. The process development & optimization
segment is expected to grow at the fastest rate in the gene therapy cell
culture media market due to the importance of process development in
manufacturing a clinically and commercially viable therapeutic product.
Researchers need to consider the best practices and identify manufacturing gaps
during process development. Appropriate bioprocessing and development studies
are conducted to produce a promising therapeutic product. Cell Type/Target Cell Line Insights Market Segment 2025 (%) HEK293 / HEK293T 38% Primary T Cells 10% Stem Cells (iPSC / MSC) 15% CHO (Biologics / Some Vector
Workflows) 12% Other Producer or Specialist
Cell Lines (e.g., insect cells) 25%
The HEK293/HEK293T segment dominated
the market share by 38% in 2025, owing to its role as a novel platform to
produce viral vectors and recombinant proteins. It is the second most employed
cell line used in the production
of biopharmaceuticals and the conduct of academic research. The HEK293
cells have high transfection efficiency, which makes them a unique and ideal
platform for expressing recombinant proteins. The primary T cells segment is expected to
grow at the fastest rate in the gene therapy cell culture media market due
to the high preference of researchers for primary T cells for genetic
engineering through viral vectors or non-viral methods. They present a more
relevant physiological environment during R&D and quality control.
Industries prefer them to test nutrient and metabolic profiling that impacts
cell function. Formulation/Format Insights Market Segment 2025 (%) Liquid Ready-to-Use Media 62% Powder / Concentrate Media 10% Kits (media + supplements +
feeds) 10% Custom Blends 18%
The liquid ready-to-use media segment
dominated the market share by 62% in 2025, owing to the high preference for
liquid media for bench-scale processes. It requires minimal preparation steps
and is ready-to-use, which makes it an ideal solution for small-scale gene
therapy development. It supports streamlined operations and consistent
manufacturing processes. The custom blends segment is expected to
grow at the fastest rate in the gene therapy cell culture media market due
to its core roles in maximizing viral vector yields and enabling seamless
scale-up in industrial processes. They allow manufacturers to maintain
regulatory compliance and protect therapeutic efficacy. They reduce the risk of
viral contamination and ensure batch-to-batch consistency. End User Insights Market Segment 2025 (%) Pharmaceutical &
Biotechnology Companies 50% CDMOs / Viral Vector CMOs 20% Academic & Research
Institutes 15% Hospitals / Cell Therapy
Treatment Centers 5% Diagnostics / Lab Service
Providers 10%
The pharmaceutical & biotechnology
companies segment dominated the market share by 50% in 2025, owing to the
globally leading innovations, scientific expertise, and transparency in
customer relationships. The global biopharmaceutical companies have set their
goals and are committed to delivering promising healthcare solutions. They
focus on reducing industrial process timelines and producing cost-efficient
products and services. The CDMOs/viral vector CMOs segment is
expected to grow at the fastest rate in the gene therapy cell culture
media market due to the expansion of the leading contract manufacturing
organizations and contract development manufacturing organizations, which
provide services like viral vector production. They collaborate with the
leading partners from pharmaceutical and biotechnology companies to offer deep
expertise, technical consultation, bioinstrumentation, products, services, and
technological innovations. Access our comprehensive healthcare
dashboard for detailed market insights, segment breakdowns, regional
performance, and company profiles: https://www.towardshealthcare.com/access-dashboard Gene Therapy Cell Culture Media Market
Companies • PromoCell • CellGenix • Thermo Fisher/Invitrogen • Merck Millipore • Corning Life Sciences • HiMedia/Biological supply regional
players • WuXi AppTec • VectorBuilder • Irvine Scientific • Sartorius • Miltenyi Biotec • Biological Industries • Cytiva • Takara Bio • BioLife Solutions • Charles River Laboratories • Thermo Fisher Scientific • Lonza • Merck/MilliporeSigma • FUJIFILM Irvine Scientific Segments Covered in the Report By Product/Media Type • Basal Media • DMEM/F12 variants (adherent HEK
workflows) • FreeStyle/chemically defined suspension
media (HEK293 suspension) • Serum-Free Media • Formulated serum-free for adherent cells • Serum-free for suspension cells • Xeno-Free Media • Human platelet lysate alternatives • Fully defined xeno-free formulations • Feed & Supplement Solutions • Feed media (fed-batch for HEK/CHO) • Transfection enhancers & boosters • Cryopreservation & Holding Media • CryoStor®-type freezing media • HypoThermosol® and shipping media • Custom/Optimized Media (CDMO offerings) By Application/Process Stage • Upstream Viral Vector Production • AAV production (transient transfection or
producer cell lines) • Lentiviral/Retroviral production
(transfection or stable producer lines) • Cell Engineering & Expansion • Primary T-cell expansion for CAR-T/NK
cell therapies • Stem cell expansion (iPSC, MSC) • Transfection & Transduction • Transient transfection media + enhancers • Stable cell line maintenance media • Process Development & Optimization • Media optimization services for scale-up
and regulatory compliance. By Cell Type/Target Cell Line • HEK293/HEK293T • Adherent HEK media subvariants • Suspension HEK optimized media • CHO (Biologics/some vector workflows) • Primary T Cells • T-cell activation & expansion media • Cytokine-supplemented feeds (IL-2,
IL-7/15 supports) • Stem Cells (iPSC/MSC) • Other producer or specialist cell lines
(e.g., producer lines, insect cells for certain vectors) By Formulation/Format • Liquid Ready-to-Use Media • Powder/Concentrate Media (cost/storage
advantages) • Kits (media + supplements + feeds) • Custom Blends By End User • Pharmaceutical & Biotechnology
Companies • In-house vector production & cell
therapy pipelines. • CDMOs/Viral Vector CMOs (Fastest Growing) • Outsourced large-scale AAV/lenti
production requiring tailored GMP media. • Academic & Research Institutes • Hospitals/Cell Therapy Treatment Centers • Diagnostics/Lab Service Providers By Region • North America • Asia Pacific • Europe • Latin America • Middle East and Africa (MEA) Immediate Delivery Available | Buy This
Research Report Now @ https://www.towardshealthcare.com/checkout/6089 About Us Towards Healthcare is a leading global provider of technological solutions, clinical
research services, and advanced analytics, with a strong
emphasis on life science research. Dedicated to
advancing innovation in the life sciences sector, we build strategic
partnerships that generate actionable insights and transformative
breakthroughs. As a global strategy consulting firm, we empower life science
leaders to gain a competitive edge, drive research excellence, and accelerate
sustainable growth. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Europe Region: +44 778 256 0738 North America Region: +1 8044 4193 44 APAC Region:
+91 9356 9282 04 Web: https://www.towardshealthcare.com Our Trusted Data Partners Precedence
Research | Towards Packaging | Towards
Food and Beverages | Towards
Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest Browse More Insights of Towards
Healthcare: The gene
therapy platform market size was valued at
US$ 2.25 billion in 2025 and is projected to grow to 2.59 billion in 2026.
Forecasts suggest it will reach approximately US$ 9.29 billion by 2035,
registering a CAGR of 15.23% during the period. The worldwide gene
therapy for sickle cell disease market is
experiencing significant expansion, with projections indicating a revenue
increase reaching several hundred million dollars by the end of the forecast
period, spanning 2025 to 2034. The global gene
therapy market size is calculated at USD 9.5 in
2024, grew to USD 11.4 billion in 2025, and is projected to reach around USD
58.87 billion by 2034. The market is expanding at a CAGR of 20% between 2025
and 2034. The gene
therapy clinical trials market size was valued
at US$ 1.36 billion in 2025 and is projected to grow to 1.49 billion in 2026.
Forecasts suggest it will reach approximately US$ 3.29 billion by 2035,
registering a CAGR of 9.24% during the period. The viral
vector gene therapy market size reached US$
13.14 billion in 2024 and is anticipate to increase to US$ 14.62 billion in
2025. By 2034, the market is forecasted to achieve a value of around US$ 38.39
billion, growing at a CAGR of 11.23%. The AAV
gene therapy market size was estimated at US$ 3.85 billion in 2025, projected to
increase to US$ 5.4 billion in 2026 and reach US$ 112.24 billion by 2035,
showing a healthy CAGR of 40.1% across the forecast years. The U.S.
cell and gene therapy CRO market size was
estimated at USD 2.33 billion in 2025 and is predicted to increase from USD
2.56 billion in 2026 to approximately USD 6.02 billion by 2035, expanding at a
CAGR of 9.95% from 2026 to 2035. The AAV
manufacturing service market size touched US$
1.44 billion in 2025, with expectations of climbing to US$ 1.66 billion in 2026
and hitting US$ 6.25 billion by 2035, driven by a CAGR of 15.84% over the
forecast period. The global cell
& gene therapy logistics market size is
estimated at US$ 6.41 billion in 2025, is projected to grow to US$ 7.76 billion
in 2026, and is expected to reach around US$ 43.14 billion by 2035. The market
is projected to expand at a CAGR of 21% between 2026 and 2035. The global cell
and gene therapy CRO market size is estimated
at US$ 4.90 billion in 2024, is projected to grow to US$ 5.39 billion in 2025,
and is expected to reach around US$ 12.59 billion by 2034. The market is
projected to expand at a CAGR of 9.9% between 2025 and 2034.