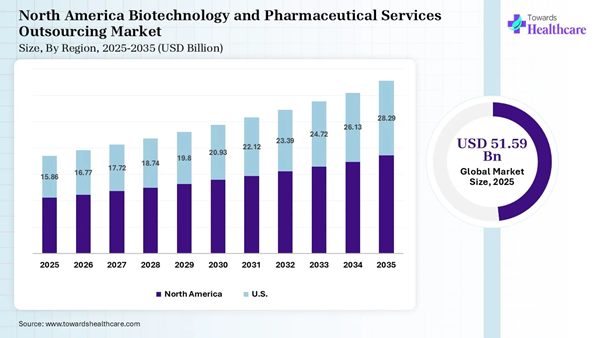
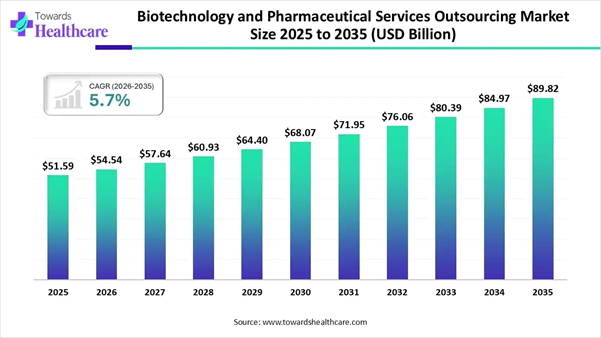
The global biotechnology and pharmaceutical services outsourcing market was valued at approximately USD 51.59 billion in 2025. It is expected to grow from USD 54.54 billion in 2026 to around USD 89.82 billion by 2035, registering a CAGR of 5.7% during the forecast period from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6631
Biotechnology and pharmaceutical companies prefer to contract certain activities related to pharmaceutical and biotechnology R&D, manufacturing, and commercialization to external third-party companies or service providers. Biotechnology prioritizes the development and modification of processes, products, and technologies for applications in agriculture, healthcare, and environmental sustainability.
Pharmaceutical services encompass R&D, manufacturing, quality control, quality assurance, regulatory compliance, and marketing of pharmaceutical products. Several laboratories and diagnostic centers outsourced testing services to handle the expanded volume of raw materials. The pandemic cases also raised the need for clinical trials, vaccine development, diagnostic testing, manufacturing, and laboratory services.
Market Highlights
• Biotechnology and pharmaceutical services outsourcing sector pushed the market to USD 54.54 billion by 2026.
• Long-term projections show USD 89.82 billion valuation by 2035.
• Growth is expected at a steady CAGR of 5.7% in between 2026 to 2035.
• North America dominated the biotechnology and pharmaceutical services outsourcing market share by 41% in 2025.
• Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
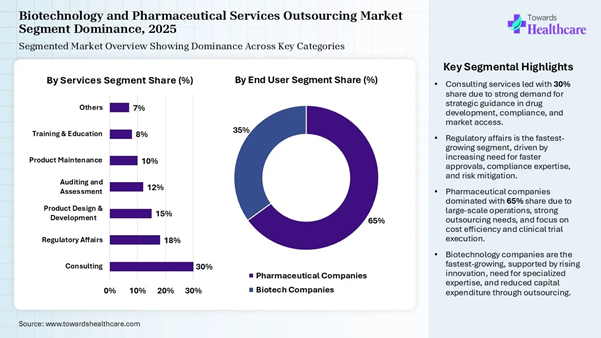
• By service, the consulting services segment dominated the market share by 30% in 2025.
• By service, the regulatory affairs segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By end user, the pharmaceutical companies segment led the market share by 65% in 2025.
• By end user, the biotechnology companies segment is expected to grow at the fastest CAGR in the market during the forecast period.
Momentous Growth of Biopharmaceutical Sectors
The biotechnology and pharmaceutical services outsourcing market is driven by outsourcing different services related to clinical development, biopharmaceutical development, diagnosis, etc. The expanding landscape of contract research organizations (CROs), contract development and manufacturing organizations (CDMOs), and contract manufacturing organizations (CMOs) is supporting the research and development of novel biologics, vaccines, medicines, biosimilars, and medicinal products.
The CDMOs provide integrated and end-to-end services to companies, including research expertise, technical consultation, technological integration, diagnostics, and therapeutics. The outsourcing contracts provide exclusive platforms for novel research, including cell and gene therapies, regenerative medicine, precision medicine, stem cell therapy, immunotherapy, etc.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Objectives of the CRO and CDMO
The contract research organization (CRO) aims to provide expertise in preclinical research and clinical trial management to prove the safety and efficacy of a drug. The CROs enable the research phase, which is important to obtain regulatory approval for entry into the market.
On the other hand, the goal of a contract development and manufacturing organization (CDMO) is to support pharma companies in drug manufacturing. The CDMOs also scale up the production process to ensure the safety, efficacy, effectiveness, and commercial viability of a drug.
Navigating the Complex Roles of CRO and CDMO
The CDMO companies provide outsourcing services to pharmaceutical companies and encompass both drug development and manufacturing phases. The core functions of CDMO lie in drug development, manufacturing, drug delivery systems, and regulatory support. A CDMO helps in the formulation of drug candidates, the optimization of active pharmaceutical ingredients, stability studies, and product scaling. Moreover, the CDMO contributes to large-scale production of clinical trial materials and commercial batches in compliance with good manufacturing practice (GMP) guidelines. A CDMO helps in the design of drug delivery systems for injectables, tablets, or biologics. It also guides pharma companies through regulatory submissions to ensure compliance with global standards.
However, the CRO is an organization that supports biotechnology and pharmaceutical companies in their research and clinical development lifecycle. The core functions played by CRO are preclinical studies, clinical trial management, etc. Moreover, many CROs offer services in regulatory strategy, drug discovery, data management, and post-marketing surveillance. The CROs help reduce development costs, streamline timelines, and ensure regulatory compliance across all phases of development. They primarily assist pharmaceutical companies in conducting research to test the safety and efficacy of drugs before they reach the market. The CROs contribute to preclinical studies, clinical trial management, and data analysis. They perform non-human studies and animal testing to collect data on a drug’s pharmacodynamics and pharmacokinetics. They manage logistics of clinical trials, analyze the data, prepare reports, and assist with regulatory submissions.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Regional Analysis
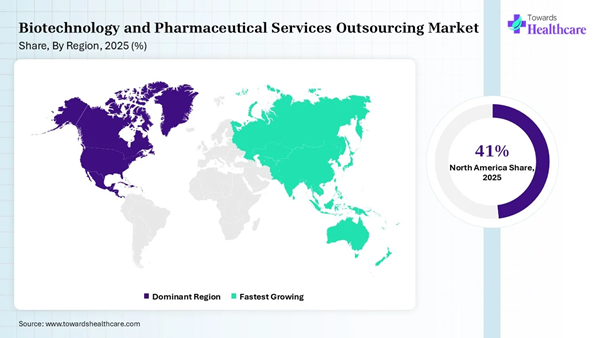
|
Region |
Market Share (%) |
|
North America |
41% |
|
Europe |
26% |
|
Asia Pacific |
21% |
|
South America |
7% |
|
MEA |
5% |
North America dominated the biotechnology
and pharmaceutical services outsourcing market share by 41% in 2025, owing to
increasing drug development complexity, a shift towards specialized R&D
expertise, and supply chain resilience. The National Institutes of Health
(NIH), the world’s largest public funder of biomedical research, invested
nearly $50 billion in its own research and laboratories, where research is
conducted at more than 2,500 hospitals, universities, and medical centers across
the U.S. The NIH also supports biopharmaceutical innovation and the development
of skilled scientific talent. U.S. Market Analysis The biotechnology and pharmaceutical
services outsourcing market in the U.S. is advancing due to decentralized
clinical trials, the rise of advanced therapies, and increased focus on
domestic manufacturing. The biotechnology companies focus on resolving
potential challenges related to clinical trial activity, new drug launches,
development productivity, commercialization models, and more. Asia Pacific is expected to grow at the
fastest rate in the biotechnology and pharmaceutical services outsourcing
market during the forecast period due to cost optimization, operational
efficiency, and regulatory harmonization and reforms. Asia Pacific is evolving into
a hub for high-end and complex services, including cell and gene therapies,
biologics, and mRNA vaccines. The companies are investing in advanced
technologies like big data analytics and AI
for drug discovery and clinical trial optimization. India Market Trends The biotechnology and pharmaceutical
services outsourcing market in India is transforming through public-private
collaboration. India remains a hub for a robust scientific ecosystem, supported
by a wide network of private enterprises and public research institutions. The
research institutions aim to build stronger and more resilient
collaborations. Get the latest insights on life science
industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership Segmentation Analysis Product Insights Service Segment Market Share (%) Consulting 30% Regulatory Affairs 18% Product Design &
Development 15% Auditing and Assessment 12% Product Maintenance 10% Training & Education 8% Others 7%
The consulting services segment dominated
the biotechnology and pharmaceutical services outsourcing market share by 30%
in 2025, owing to the rising trend of infrastructure and cloud consulting, the
merging of AI and data analytics, the development of a cybersecurity
infrastructure, and the transformation of outdated platforms. The increased
focus on business model validation, data privacy, cyber laws, and FDI
compliance drives the growth of consulting services. They aid in wealth
management, financial planning, and building investor relations. The regulatory affairs segment is expected
to grow at the fastest CAGR in the biotechnology and pharmaceutical services
outsourcing market during the forecast period due to the integral role of
regulatory affairs professionals from the earliest stages of drug development.
They ensure adherence of the generated data with regulatory expectations and
provide strategic guidance on the design of preclinical studies. These experts
help to streamline the development process and reduce the time and costs needed
to bring a new therapy to the market. End User Insights End User Segment Market Share (%) Pharmaceutical Companies 65% Biotech Companies 35%
The pharmaceutical companies segment led
the biotechnology and pharmaceutical services outsourcing market share by 65% in
2025, owing to the trending innovations like digital
biomarkers, AI-driven drug R&D, and synthetic biology. The leading
pharma companies are enabled by intelligent systems that learn, optimize, and
adapt to emerging trends and technologies. They are involved in developing
direct-to-patient platforms that integrate adherence tools, education, and
data-sharing in one seamless interface. The biotechnology companies segment is
expected to grow at the fastest CAGR in the biotechnology and pharmaceutical
services outsourcing market during the forecast period due to the rise of top
biotechnology companies such as Thermo Fisher Scientific, Novo Nordisk, Amgen,
Gilead Sciences, Vertex Pharmaceuticals, Regeneron Pharmaceuticals, Samsung
Biologics, and many more. Biotechnology became promising due to high demand for
jobs in diagnostics, pharma, agriculture, R&D, and healthcare, and
government policies like the BioE3 policy. The BioE3 policy represents
biotechnology for economy, employment, and environment. Top Companies in the Market • Parexel International Corporation • The Quantic Group • Boston Scientific Corporation • IQVIA • Lachman Consultant Services, Inc. • GMP Pharmaceuticals Pty Ltd. • Charles River Laboratories International,
Inc. • Laboratory Corporation of America
Holdings (LabCorp) • ICON Plc • Lonza Group • Syneos Health • WuXi AppTec • Sartorius Stedim Biotech Access our comprehensive healthcare
dashboard for detailed market insights, segment breakdowns, regional
performance, and company profiles: https://www.towardshealthcare.com/access-dashboard Segments Covered in the Report By Service • Consulting o Regulatory Consulting o Clinical Development Consulting o Strategic Planning & Business
Development Consulting o Quality Management Systems consulting o Others • Regulatory Affairs o Legal Representation o Regulatory Writing & Publishing o Product Registration & Clinical Trial
Applications o Regulatory Submissions o Regulatory Operations o Others • Product Design & Development o Research, Strategy, & Concept
Generation o Concept & Requirements Development o Detailed Design & Process Development o Design Verification & Validation o Process Validation & Manufacturing
Transfer o Production & Commercial Support • Auditing and Assessment • Product Maintenance • Training & Education • Others By End Use • Pharmaceutical Companies • Biotech Companies By Region • North America • Asia Pacific • Europe • Latin America • Middle East and Africa (MEA) Immediate Delivery Available | Buy This
Research Report Now @ https://www.towardshealthcare.com/checkout/6631 About Us Towards Healthcare is a leading global provider of technological solutions, clinical
research services, and advanced analytics, with a strong
emphasis on life science research. Dedicated to
advancing innovation in the life sciences sector, we build strategic
partnerships that generate actionable insights and transformative
breakthroughs. As a global strategy consulting firm, we empower life science
leaders to gain a competitive edge, drive research excellence, and accelerate
sustainable growth. You can place an order or ask any
questions, please feel free to contact us at sales@towardshealthcare.com Europe Region: +44 778 256 0738 North America Region: +1 8044 4193 44 APAC Region:
+91 9356 9282 04 Web: https://www.towardshealthcare.com Our Trusted Data Partners Precedence
Research | Towards Packaging | Towards
Food and Beverages | Towards
Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest Browse More Insights of Towards
Healthcare: ➡️ The global biotechnology
& pharmaceutical services market size is calculated at USD 76.51 in
2024, grew to USD 80.7 billion in 2025, and is projected to reach around USD
130.56 billion by 2034. The market is expanding at a CAGR of 5.48% between 2025
and 2034. ➡️ The global biotechnology
CMO and CDMO market size is estimated at US$ 67.25 billion in 2024, is
projected to grow to US$ 74.01 billion in 2025, and is expected to reach around
US$ 199.67 billion by 2034. The market is projected to expand at a CAGR of
11.54% between 2025 and 2034. ➡️ The agricultural
biotechnology market size in 2025 was US$ 137.46 billion, expected to
grow to US$ 149.62 billion in 2026 and further to US$ 320.68 billion by 2035,
backed by a robust CAGR of 8.84% between 2026 and 2035. ➡️ The global biotechnology
based chemicals market size was estimated at USD 109.53 billion in 2025
and is predicted to increase from USD 120.09 billion in 2026 to approximately
USD 274.93 billion by 2035, expanding at a CAGR of 9.64% from 2026 to 2035. ➡️ The global biotechnology
reagents & kits market size is calculated at USD 813.38 billion in
2025, grew to USD 902.36 billion in 2026, and is projected to reach around USD
2297.07 billion by 2035. The market is expanding at a CAGR of 10.94% between
2026 and 2035. ➡️ The global life
science tools market size is forecast to grow at a CAGR of 13%, from
USD 156.53 billion in 2025 to USD 531.34 billion by 2035, over the forecast
period from 2026 to 2035 ➡️ The global biotechnology
instruments market size is projected to reach USD 138.06 billion by
2034, growing from USD 97.33 billion in 2025, at a CAGR of 3.96% during the
forecast period from 2025 to 2034. ➡️ The global clinical
stage biotechnology market size was estimated at USD 47.19 billion in
2025 and is predicted to increase from USD 54.66 billion in 2026 to
approximately USD 205.32 billion by 2035, expanding at a CAGR of 15.84% from 2026
to 2035. ➡️ The global membranes
for pharmaceuticals and biotechnology market size was estimated at USD
5.13 billion in 2025 and is predicted to increase from USD 5.49 billion in 2026
to approximately USD 10.15 billion by 2035, expanding at a CAGR of 7.06% from
2026 to 2035. ➡️ The global pharmaceutical
& biotechnology environmental monitoring market size was estimated
at USD 1.78 billion in 2025 and is predicted to increase from USD 1.88 billion
in 2026 to approximately USD 3.11 billion by 2035, expanding at a CAGR of 5.74%
from 2026 to 2035.