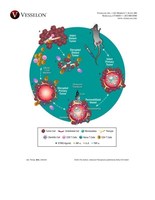
A preclinical study, authored by Merck & Co., Inc. in collaboration with Vesselon, Inc., and published in Advanced Therapeutics in September 2021, reported a new, safe method for STING agonist delivery other than the limiting intratumoral approaches that have been used.
|
NORWALK, Conn., Sept. 22, 2021 /PRNewswire/ -- A preclinical study, authored by Merck & Co., Inc. in collaboration with Vesselon, Inc., and published in Advanced Therapeutics in September 2021, reported a new, safe method for STING agonist delivery other than the limiting intratumoral approaches that have been used. Vesselon's acoustically active drug Imagent® augmented the pharmacokinetics of the STING agonist MSA-1 by increasing tumor uptake by 658%. When compared to a low MSA-1 intravenous dose that had no anti-tumor effect as a single agent, the addition of Imagent® under ultrasound exposure from conventional equipment generated complete tumor regressions. The data also surprisingly showed that the 5-minute procedure substantially elevated cytokine production and induced immunomodulatory benefits by sustaining STING-mediated immune activation while reversing its impact on immune suppression. These immune engaging aspects of the Vesselon drug resulted in complete regression of both primary and distant tumors in 44% of enrolled subjects. While the intratumoral dosed group had no effect on non-treated tumors, the procedure eradicated non-treated distant tumors. "We created a new field of therapeutics called 'acoustically active drugs,' that can raise the pharmacokinetics of drugs in virtually any therapeutic class," said Clay Larsen, Vesselon president and CEO. "This collaborative study with Merck underscores the benefits of our platform. We look forward to conducting clinical trials of our drugs in several important and underserved immuno-oncology indications." Also reported in the study, "the delivery strategy of MSA-1 in this study can be further combined with immune regulatory checkpoint blockades to fully realize the immunomodulatory effect of sonoporation. In the future, we aim to align and position this approach for clinical trials via an arm in an active treatment cohort or leveraging FDA's guidance document on the expanded access to investigational drugs for treatment to tackle some of the huge unmet needs in circumstances where lack of treatment efficacy can be attributed to lack of tumor exposure, ultimately helping patients in need." "Vesselon's approach does not require any modification of a therapeutic agent and uses an optimally designed FDA-approved lipid microsphere with diagnostic ultrasound equipment that is available in many cancer centers, making the clinical translation much easier than particulate-based approaches." This unique lipid microsphere can also encapsulate drugs for improved targeting, lower toxicity, and protection against degradation in circulation — key issues for the pharmaceutical industry. One proprietary drug is an encapsulated, unaltered IL-2 called SonoLeukin™. Vesselon plans to submit SonoLeukin™ INDs to improve the performance of Keytruda in recurrent, metastatic head and neck cancer as well as pancreatic cancer – two indications where patient outcomes remain poor. The full preclinical study titled "Sonoporation-Enhanced Delivery of STING Agonist Induced Robust Immune Modulation and Tumor Regression" is available online at https://bit.ly/3Ekbc2N. About Vesselon Inc.
SOURCE Vesselon, Inc. |