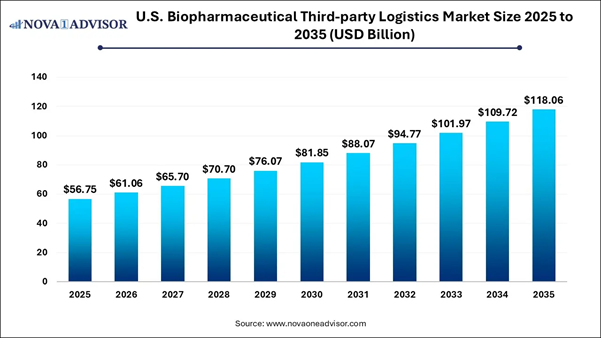
According to Nova One Advisor, the U.S. biopharmaceutical third-party logistics market size is calculated at 56.75 billion in 2025 and is projected to surpass USD 118.06 billion by 2035 with a remarkable CAGR of 7.6% from 2026 to 2035.
From clinical trials to final patient delivery, AI is transforming the U.S. biopharmaceutical third-party logistics market. This evolution has moved from experimental pilots to crucial supply chain orchestration within an AI-driven digital ecosystem. By integrating Internet of Things (IoT) technology, robotics, and generative AI, logistics providers are accelerating delivery timelines and ensuring real-time visibility for high-value therapies, turning complex supply chains into rapid, compliant, and patient-centric processes.
Key Takeaways:
· Non-cold chain logistics led the U.S. Biopharmaceutical Third-party Logistics (3PL) market with the largest revenue share of 80.2% in 2025.
· Cold chain logistics is projected to witness a lucrative growth rate of 10.1% over the forecast period.
· The warehousing & storage segment led in terms of both revenue and volume share in 2025 and accounted for 43.4% of the market.
· These are anticipated to witness lucrative growth over the forecast period.
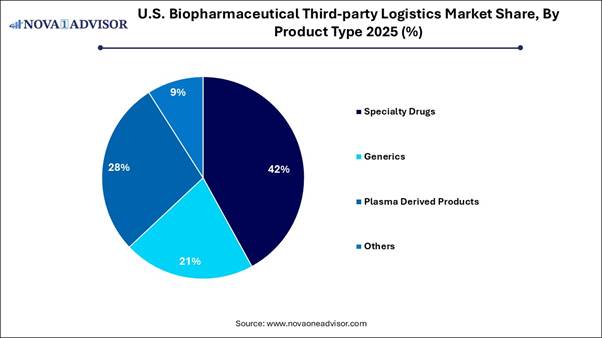
· Specialty drug products alone contributed to 42% of the industry in 2025.
· The segment is also anticipated to witness the fastest growth rate of 7.7% over the forecast period.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/6579
Market Overview: Powering the U.S. Biopharmaceutical Revolution
The U.S. biopharmaceutical third-party logistics market encompasses specialized outsourcing services, warehousing, cold-chain storage, transportation, and inventory management tailored for sensitive biological drugs, vaccines, and other specialized pharmaceutical products. This market is driven by the increasing demand for cold-chain-dependent biologics, biosimilars, and the need for strict FDA regulatory compliance. Manufacturers are shifting towards outsourcing to reduce costs and effectively manage complex, temperature-sensitive logistics focused on specialized drug distribution, AI-enabled warehousing, and end-to-end cold-chain visibility.
Explosive Demand for Cryogenic Logistics: Major Potential
The growing focus on personalized CAR-T and gene-editing therapies is causing a significant surge in demand for specialized cryogenic logistics, which require rigorous chain-of-identity and cold-chain oversight. This market is expected to more than double, presenting a high-value opportunity for third-party logistics providers capable of delivering end-to-end, real-time monitored cryogenic transport and storage. These providers are well-positioned to capitalize on the rapid expansion of complex, high-margin pharmaceutical logistics.
Severe Shortage of GDP-Trained Labor: Major Limitation
A critical shortage of Good Distribution Practice (GDP)-trained labor is severely impacting the specialized cold-chain logistics market, creating bottlenecks for high-stakes, temperature-sensitive biologics. This significant talent shortage is increasing operational costs and restricting 3PL capacity, hindering innovation, and limiting the ability to meet the rapid, compliance-driven demands of the expanding cell and gene therapy sector.
Immediate Delivery is Available | Get Full Report Access@ https://www.novaoneadvisor.com/report/checkout/6579
Report Scope of The U.S. Biopharmaceutical Third-party Logistics Market
|
Report Coverage |
Details |
|
Market Size in 2025 |
USD 61.06 Billion |
|
Market Size by 2036 |
USD 118.06 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 7.6% |
|
Base Year |
2025 |
|
Forecast Period |
2026-2035 |
|
Segments Covered |
Supply Chain, Service Type, Product Type |
|
Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
|
Key Companies Profiled |
FedEx; DHL International GmbH; SF Express; United Parcel Service of America, Inc.; AmerisourceBergen Corporation; DB Schenker; KUEHNE + NAGEL; Kerry Logistics Network Ltd.; Agility |
You can place an order or ask any
questions, please feel free to contact at sales@novaoneadvisor.com
| +1 804 441 9344 U.S. Biopharmaceutical Third-party
Logistics Market: Country Analysis New Jersey:
As a leading global pharmaceutical hub, New Jersey hosts a high concentration
of major pharma companies, including Johnson and Johnson, Merck, and Bristol
Myers Squibb. Thirteen of the top 20 biopharma companies have a significant
presence here, making it a critical international logistics center with
specialized warehousing, over 1,700 life sciences businesses, and large
research and development facilities designed for the swift and regulated
movement of temperature-sensitive biologics. California:
California leads the market with a massive, interconnected biotech ecosystem,
particularly in the Bay Area and Southern California (Conejo Valley), anchored
by industry leaders like Amgen and Genentech. It serves as a global focal point
for integrating AI-driven drug discovery with advancements in personalized
medicine and cell/gene therapy. Pennsylvania: Known for its high concentration of contract development and
manufacturing organizations (CDMOs), Pennsylvania is crucial for drug
packaging, testing, and distribution. The state is a prime location for
large-scale 3PL warehouse investments, including new cold-chain and Good
Manufacturing Practice (GMP) facilities, as well as the construction of
high-capacity manufacturing hubs for cell and gene therapies. Texas: Texas
is rapidly becoming a major distribution hub for the Southern U.S., supporting
large manufacturing centers and enhancing supply chain resilience. Its key role
involves managing high-volume cold-chain logistics for mRNA vaccines, specialty
drugs, and diagnostic distribution. North Carolina: Recognized for significant investments in the Research Triangle
Park, North Carolina is a national leader in biotechnology development and a
rapidly growing center for GMP manufacturing. The state offers advanced,
specialized cold-chain capabilities and is quickly expanding its footprint in
GMP storage and bioprocessing facilities. Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/6579
U.S. Biopharmaceutical Third-party Logistics
Market: Segmental Analysis By Supply Chain Analysis The cold chain segment dominated the market
in 2025, primarily driven by the increasing demand for temperature-sensitive
biologics, vaccines, and cell and gene therapies. Strict FDA regulations and
Good Distribution Practices require precise and documented temperature control
throughout the supply chain, leading to a reliance on specialized third-party
logistics providers. The integration of AI-driven logistics, real-time IoT
tracking, and advanced monitoring systems ensures product integrity and
minimizes spoilage, making premium third-party logistics services essential. The non-cold chain segment is expected to
experience the fastest growth during the forecast period. This is primarily due
to the high-volume distribution of ambient pharmaceuticals
and medical
devices. Non-cold chain logistics encompass room-temperature
pharmaceuticals, over-the-counter drugs, and medical devices, which hold a
significant market share thanks to extensive distribution networks. Lower
operational costs compared to cold chain logistics, along with the capability
to handle large-scale shipments, make this segment a preferred choice for
traditional pharmaceutical distribution. By Service Type Analysis The transportation
segment led the market in 2025, driven by the need for secure, timely, and
compliant delivery of sensitive, high-value products through advanced
technologies. The rise of temperature-sensitive biologics, vaccines, and cell
and gene therapies necessitate strict and specialized transportation, thereby
increasing demand for 3PL expertise. There is a high demand for specialized
refrigerated transportation, such as air freight, to ensure rapid delivery of
biologics. Additionally, the shift toward direct-to-patient delivery models creates
a strong demand for specialized, flexible logistics solutions and last-mile
delivery. The warehousing and storage segment is
anticipated to experience the fastest growth during the forecast period. This
growth is mainly driven by the increasing demand for specialized,
temperature-controlled facilities and regional distribution hubs, fueled by the
expansion of biologics, cell therapies, and personalized medicine. Beyond
storage, warehouses are evolving into key service providers, offering
value-added services such as specialized packaging, labeling compliance, and
real-time inventory tracking. These enhancements improve efficiency, reduce
human error, and address the complexities of logistics. By Product Type Analysis The specialty drugs segment dominated the
market in 2025, largely due to high-value, temperature-sensitive therapies that
require specialized handling. Specialty drugs, including those used for cancer
and arthritis, are often high-cost and necessitate specific
temperature-controlled storage and transportation. The introduction of
innovative and sensitive medicines requires advanced 3PL services for secure
distribution. Specialty medications, such as CAR-T therapies, demand unique
logistics solutions, including specialized handling, patient-specific delivery,
and end-to-end traceability. The plasma-derived products segment is
expected to see the fastest growth during the forecast period. This growth is
primarily driven by the increasing demand for temperature-sensitive
immunoglobulins and therapeutics used to treat chronic and rare diseases. The
growing patient population in need of immunoglobulins, albumin, and other
plasma-derived therapies is resulting in increased logistical volumes. As a
subset of the rapidly expanding biopharmaceutical segment, these products are
significantly fueling the demand for specialized and compliant storage and
distribution services. Key Innovations for the U.S.
Biopharmaceutical Third-party Logistics Market Innovation Description Impact on Market IoT-Enabled Real-time Monitoring Deployment of smart sensors to track temperature, humidity, light,
and location in real time during transit. Enables proactive intervention to prevent spoilage of biologics
and vaccines, reducing costly losses. AI-Driven Predictive Analytics Using AI/ML for demand forecasting, inventory optimization, and
route planning to avoid delays. Increases supply chain efficiency, optimizes warehouse inventory,
and reduces logistical bottlenecks. Blockchain for Chain-of-Custody Creating decentralized, immutable ledgers to track the movement
and handling of drugs, particularly CGTs. Ensures high-level transparency, data integrity, and compliance
with DSCSA serialization requirements. Autonomous Warehouse Robotics Use of autonomous mobile robots (AMRs) for automated picking, packing,
and sorting in warehouses. Addresses labor shortages, increases warehouse throughput, and
allows 3PLs to scale without expanding headcount. Advanced Cold Chain Packaging Adoption of passive, reusable, and VIP (Vacuum Insulated Panel)
packaging for cryo and ultra-frozen shipments. Protects specialized therapies (cell/gene) while reducing waste
and lowering carbon footprints.
U.S. Biopharmaceutical Third-party
Logistics Market Companies ·
FedEx ·
United Parcel Service ·
AmerisourceBergen Corporation ·
KUEHNE+NAGEL ·
Kerry Logistics Network Ltd. ·
Agility Major Shifts in the U.S.
Biopharmaceutical Third-party Logistics Market ·
In February 2025, FedEx launched FedEx
Surround®, an intelligent solution for enhanced monitoring and intervention in
logistics, specifically for customers in Korea. This service combines three
levels: Select, Preferred, and Premium, and supports various industries like
healthcare and aerospace. Key benefits include near real-time visibility,
operational enhancements for sensitive shipments, and 24/7 expert support,
empowering businesses to optimize operations and prevent disruptions. ·
In April 2025, DHL Supply Chain opened a €10
million Pharma Hub in Singapore as part of a €500 million investment in Asia
Pacific to strengthen its Life Sciences and Healthcare infrastructure. This
GMP-compliant facility features advanced cold chain infrastructure and strategic
connectivity to major transport hubs, enhancing the delivery of healthcare
products in a region with increasing demand. ·
In January 2026, UPS announced an expansion of
healthcare capabilities in Louisville, adding a 42,000 square foot cold storage
cooler for temperature-sensitive medicines for $6.1 million. This is part of
its existing healthcare facilities, including the $100 million UPS Labport,
which integrates diagnostics and logistics to accelerate patient care. Related Report ➡️ Advanced Therapy Medicinal Products CDMO Market:
Explore how demand for outsourced manufacturing is accelerating innovation in
advanced cell and gene therapies ➡️ U.S. Integrated Delivery Network Market: Discover
how coordinated healthcare systems are transforming patient care and
operational efficiency across the U.S. ➡️ U.S. Digital Health Market: Analyze the rapid
expansion of telehealth, AI diagnostics, and connected healthcare technologies ➡️ Healthcare Contract Research Organization Market:
See how clinical outsourcing and drug development complexity are driving CRO
market growth ➡️ Cell And Gene Therapy CDMO Market: Track the
growing need for scalable manufacturing solutions in next-generation therapeutics ➡️ Viral Vector Production (Research-use) Market:
Understand how research-driven demand is boosting viral vector production
capabilities worldwide ➡️ Allogeneic Cell Therapy Market: Discover how
off-the-shelf cell therapies are reshaping regenerative medicine and cancer
treatment ➡️ Viral Capsid Development Market: Explore
innovations in viral capsid engineering supporting safer and more targeted gene
delivery ➡️ mRNA Therapeutics Market: Gain insight into the
expanding applications of mRNA technology beyond vaccines ➡️ Life Sciences BPO Market: Learn how outsourcing
strategies are helping life sciences companies improve efficiency and reduce
costs ➡️ Stable Isotope Labeled Compounds Market: Examine
the increasing role of isotope-labeled compounds in pharmaceutical research and
diagnostics ➡️ Risk-based Monitoring Software Market: See how
advanced analytics and compliance requirements are driving adoption of clinical
trial monitoring solutions ➡️ Biopharmaceutical CMO and CRO Market: Discover how
biopharma outsourcing partnerships are accelerating drug development timelines ➡️ U.S. Biopharmaceuticals Contract Manufacturing Market:
Analyze the growing demand for flexible biologics manufacturing infrastructure
in the U.S. ➡️ Biopharmaceutical Excipients Market: Explore how
formulation advancements are increasing the importance of high-performance
excipients in biologics ➡️ U.S. Cell Culture Market: Track the rising adoption
of cell culture technologies in vaccine production, drug discovery, and
regenerative medicine ➡️ U.S. Biopharmaceutical CMO and CRO Market:
Understand how expanding biologics pipelines are strengthening demand for outsourced
research and manufacturing services Segments Covered in the Report This report forecasts revenue growth at
country levels and provides an analysis of the latest industry trends in each
of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc.
has segmented the U.S. biopharmaceutical third-party logistics market Supply Chain Service Type Product Type Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/6579 About-Us Nova One Advisor is a global leader
in market intelligence and strategic consulting, committed to delivering deep,
data-driven insights that power innovation and transformation across
industries. With a sharp focus on the evolving landscape of life sciences, we
specialize in navigating the complexities of cell and gene therapy, drug
development, and the oncology market, enabling our clients to lead in some of
the most revolutionary and high-impact areas of healthcare. Our expertise spans the entire
biotech and pharmaceutical value chain, empowering startups, global
enterprises, investors, and research institutions that are pioneering the next
generation of therapies in regenerative medicine, oncology, and precision
medicine. Web: https://www.novaoneadvisor.com/ Our Trusted Data Partners: Nova One Advisor - Market
Research Reports & Consulting Firm Nova One Advisor offers
comprehensive market research reports with in-depth industry analysis and
market data. Call us: +1 804 420 9370 Email: sales@novaoneadvisor.com Web: https://www.novaoneadvisor.com/ You can place an order or ask any questions,
please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344