Market Segmentations Covered: (By Product: Synthetic, Biotech; By Manufacturer Type: In-house, Outsourced; By Drug Type: Innovative, Generic; By Application: Oncology, Hormonal, Glaucoma, Others; By Region: North America, Europe, Asia Pacific, Latin America and Middle East and Africa) Global Analysis, Size, Trends, Leading Companies, Regional Outlook and Forecast 2026 to 2035.
Market Sizing and Forecast:
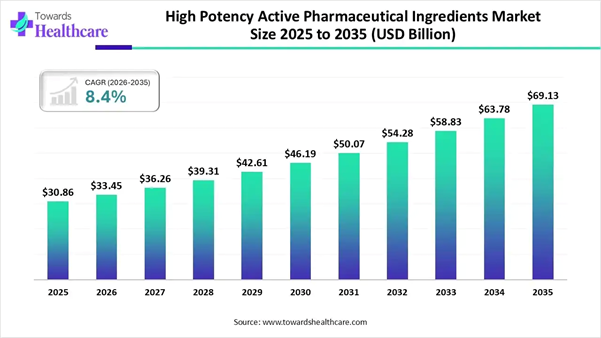
The global high-potency active pharmaceutical ingredients (HPAPI) market was valued at USD 30.86 billion in 2025 and is projected to grow from USD 33.45 billion in 2026 to around USD 69.13 billion by 2035, registering a CAGR of 8.4% during the forecast period from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6694
The highly potent APIs are pharmaceutical compounds with low occupational exposure limits, due to which the operator can be exposed to the ingredient only for a short time, which further avoids the risk of short or long-term effects on the operator’s health. Accordingly, high-potency APIs require specialized personnel, equipment, and facilities for safe handling. They are critical in the pharmaceutical manufacturing to provide innovative solutions for the treatment of cancer, central nervous system disorders, diabetes, and many other diseases.
Market Highlights
• High potency active pharmaceutical ingredients sector pushed the market to USD 33.45 billion by 2026.
• Long-term projections show USD 69.13 billion valuation by 2035.
• Growth is expected at a steady CAGR of 8.4% in between 2026 to 2035.
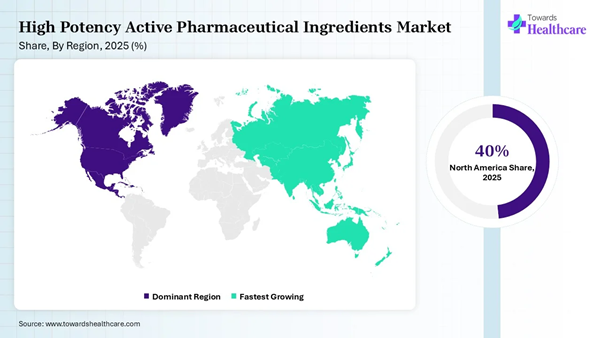
• North America dominated the global high potency active pharmaceutical ingredients market share by 40% in 2025.
• Asia Pacific is expected to grow at the fastest CAGR in the market during the forecast period.
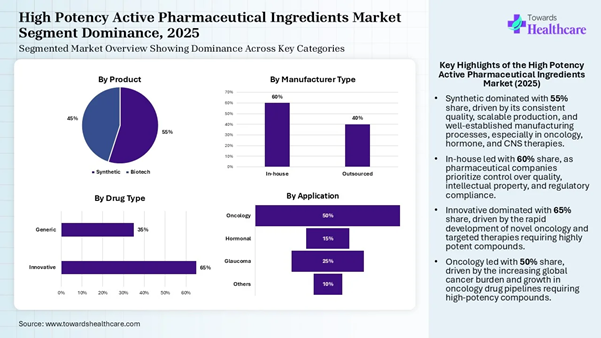
• By product, the synthetic segment holds a dominant position by 55% in the market in 2024.
• By product, the biotech segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By manufacturer type, the in-house segment accounted for a considerable revenue share of 60% in the market in 2025.
• By manufacturer type, the outsourced segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By drug type, the innovative segment led the global market share by 65%.
• By drug type, the generic segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By application, the oncology segment held the largest market by 50% in 2025.
• By application, the glaucoma segment is expected to grow at the fastest CAGR in the market during the forecast period.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Market Overview
Collaborations Set the Future of High Potency APIs
The high potency active pharmaceutical ingredients market is accelerated by the increased demand for these highly potent APIs in cancer growth inhibitors and oncology indications. The future advancements revolve around drug delivery systems, biological high potency APIs, technological integration, and regulatory evolution. The novel drug delivery systems include nanoparticles, liposomes, and micelles, while antibody-drug conjugates are also gaining traction in therapeutics. The pharmaceutical companies are using AI and machine learning to optimize manufacturing processes and enhance quality control and efficiency. The regulatory frameworks promote innovation during production while ensuring safety.
Key Indicators and Highlights
|
Key Elements |
Scope |
|
Market Size in 2026 |
USD 33.45 Billion |
|
Projected Market Size in 2035 |
USD 69.13 Billion |
|
CAGR (2026 - 2035) |
8.4% |
|
Leading Region |
North America |
|
Historical Data |
2020 - 2023 |
|
Base Year |
2025 |
|
Forecast Period |
2026 - 2035 |
|
Measurable Values |
USD Millions/Units/Volume |
|
Market Segmentation |
By Product, By Manufacturer Type, By Drug Type, By Application, By Region |
|
Top Key Players |
BASF SE, CordenPharma, BristolMyers Squibb, CARBOGEN AMCIS AG, Pfizer, Inc., Boehringer Ingelheim, Dr. Reddy’s Laboratories |
✤ Mostly Asked Questions: ✤
➡️ How can pharmaceutical companies ensure the safety of their operators when dealing with HPAPIs?
Pharmaceutical companies can ensure the safety of operators handling High Potency Active Pharmaceutical Ingredients (HPAPIs) by implementing robust containment systems, such as isolators and closed-system equipment, to minimize direct exposure. Operators should be equipped with appropriate personal protective equipment (PPE), including respirators and full-body suits.
Engineering controls like local exhaust ventilation and automated systems help reduce human contact with HPAPIs. Regular monitoring of air quality, workplace cleaning protocols, and restricted access to high-risk areas further enhance safety. Continuous training, safety drills, and medical surveillance programs ensure operators are well-prepared and healthy. Finally, strict waste disposal protocols and preventive maintenance of equipment are essential for maintaining a safe working environment.
➡️ Why is standardization important in HPAPI classification and containment strategies?
Standardization in HPAPI (High Potency Active Pharmaceutical Ingredients) classification and containment strategies is crucial for ensuring consistency, safety, and regulatory compliance across the pharmaceutical industry. By establishing uniform guidelines for classification, companies can accurately assess the potency of HPAPIs, categorize them based on their risk levels, and implement appropriate safety measures.
Standardized containment strategies ensure that all handling, processing, and storage procedures meet the same high safety standards, reducing the risk of exposure and cross-contamination. This consistency not only helps in maintaining operator safety but also ensures that pharmaceutical products meet global regulatory requirements, protecting both public health and company reputation. Additionally, standardized practices streamline the training process, enhance efficiency, and foster better communication across different teams and facilities.
Strategies to Overcome HPAPI Production Challenges
➢ Enhanced Containment Systems: Since HPAPIs are potent and require stringent containment measures, investing in state-of-the-art containment systems ensures safety and regulatory compliance. Using advanced isolators and containment suites can minimize exposure and prevent cross-contamination.
➢ Process Optimization: Streamlining production processes through advanced manufacturing technologies, such as Continuous Manufacturing (CM), can improve efficiency and reduce operational costs. Automating key processes also helps maintain consistency and minimize human error.
➢ Employee Training and Safety Protocols: Due to the toxic nature of HPAPIs, regular and specialized training for personnel on handling, exposure risks, and safety protocols is crucial. Ensuring staff is well-versed in working with potent compounds prevents accidental exposure and boosts productivity.
➢ Robust Quality Control: Rigorous quality control measures, including frequent testing for purity, stability, and consistency, are vital. Implementing Real-Time Release Testing (RTRT) and enhanced monitoring systems can ensure high-quality standards and prevent product defects.
➢ Supply Chain Management: HPAPI production often involves complex raw material sourcing and stringent storage conditions. Collaborating with reliable suppliers who can maintain high standards of quality and delivery is essential for the uninterrupted flow of production.
➢ Facility Design and Adaptability: Building production facilities that are flexible and capable of handling different types of HPAPIs while ensuring scalability is essential. This includes designing adaptable facilities that can accommodate future expansions or changes in production demands.
➢ Regulatory Compliance: Staying ahead of regulatory changes and ensuring that all facilities and processes meet global standards is vital for smooth HPAPI production. Working with experts in regulatory affairs can help anticipate and meet evolving compliance requirements efficiently.
Access our comprehensive healthcare dashboard for detailed market insights, segment breakdowns, regional performance, and company profiles: https://www.towardshealthcare.com/access-dashboard
Regional Analysis
|
Regions |
Shares 2025 (%) |
|
North America |
40% |
|
Europe |
30% |
|
Asia-Pacific |
21% |
|
Latin America |
6% |
|
Middle East & Africa |
3% |
Explanation:
• North America dominates the market with a 40% share, driven by advanced pharmaceutical manufacturing capabilities, high demand for high-potency drugs, and strong regulatory support in the U.S. and Canada.
• Europe holds 30% of the market, supported by a well-established pharmaceutical industry, high research and development investment, and stringent regulatory standards across countries like Germany and the UK.
• Asia-Pacific accounts for 21%, growing steadily due to the increasing pharmaceutical production capacity and rising demand for high-potency drugs in emerging markets like China and India.
• Latin America represents 6% of the market, experiencing gradual growth as pharmaceutical companies expand in the region to meet rising healthcare needs.
• Middle East & Africa holds 3% of the market, with limited growth compared to other regions, but increasing healthcare investments are driving demand for high-potency active pharmaceutical ingredients.
Brief Reasonings:
North America dominated the high potency active pharmaceutical ingredients market share by 40% in 2025, owing to the expanding oncology sector, rising outsourcing to contract development and manufacturing organizations (CDMOs), and robust R&D infrastructure. The rising incidence of obesity, diabetes, cancer, neurological disorders, respiratory syndromes, and other chronic conditions has raised the demand for novel biologics, biosimilars, vaccines, drug formulations, APIs, and other medicinal products to save lives. The U.S. FDA aims to simplify the process for pharmaceutical companies to set up manufacturing facilities in the U.S. through its newly launched PreCheck program.
For instance,
• In August 2025, AbbVie Inc. announced the investment of $195 million to expand active pharmaceutical ingredient manufacturing in the U.S. This initiative was a part of the company’s commitment to invest more than $10 billion of capital in the U.S. to expand critical manufacturing capabilities and facilities and support innovation.
U.S. Market Analysis
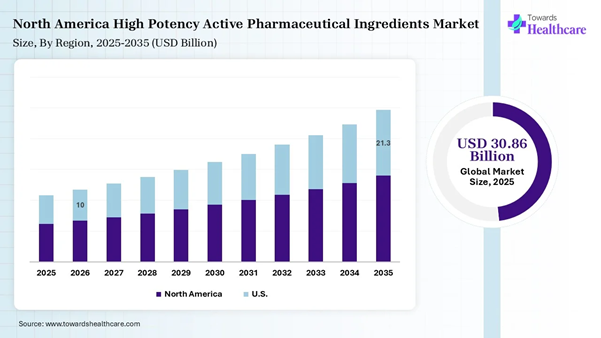
The high potency active pharmaceutical ingredients market in the U.S. is advancing by 25% share due to the expansion of contract manufacturing organizations (CMOs) and contract development and manufacturing organizations (CDMOs), and their specialized expertise and resources in R&D. The U.S. FDA launched a PreCheck scheme to boost the pharmaceutical manufacturing in the U.S. and streamline the production of new pharmaceutical production facilities across the country.
For instance,
• In March 2026, the API Innovation Center and the U.S. Department of Health and Human Services (HHS) under the U.S. government collaborated to advance the domestic production of critical medicines through expanded public-private partnerships and strengthen the resilience of the pharmaceutical supply chain in the U.S. This program aims to expand API and finished drug manufacturing, and develop propofol active pharmaceutical ingredient (API) and metoprolol tablets.
Asia Pacific is expected to grow at the fastest rate in the high potency active pharmaceutical ingredients market during the forecast period due to a strong government support for advancing pharmaceutical manufacturing, healthcare innovation, and the resilient supply chain of pharmaceutical companies. The Asia Pacific is gaining momentum in the pharmaceutical sector due to diverse healthcare systems, well-established early-stage economies, and differences in technology adoption, culture, politics, and language. Multiple nations work together to meet the manufacturing needs for medical products, which creates favorable opportunities for the pharmaceutical industry.
India Market Trends
The high potency active pharmaceutical ingredients market in India witnesses the presence of big pharma companies, biotechnology firms, startups, clinical research institutes, and academic research expertise that are dedicated to boosting biopharmaceutical innovations across the country. The government announced Biopharma SHAKTI, which is a ₹10,000 Crore initiative over five years under the Union budget 2026-27. There will be a shift from a high volume of generics to a high value of biologics across the manufacturing, supply chain, microcap businesses, and SMEs.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
Segmentation Analysis
Product Insights
|
By Product Segment |
Shares 2025 (%) |
|
Synthetic |
55% |
|
Biotech |
45% |
Explanation:
• Synthetic eads the market with a 55% share, driven by the cost-effectiveness and scalability of synthetic methods in producing high-potency active pharmaceutical ingredients.
• Biotech accounts for 45%, gaining momentum due to the growing demand for biologic drugs, particularly in areas like oncology and autoimmune diseases.
Brief Reasonings:
The synthetic segment dominated the high potency active pharmaceutical ingredients market by 55% in 2025, owing to the essential role of synthetic products for hormonal therapies and chemotherapy agents. The synthetic routes possess standardized production protocols, predictable performance, and cost efficiency, which are advantageous to manufacturing. They are critically used in standard oncology and hormonal treatments, offer high scalability, and play a major role in established manufacturing processes.
The biotech segment is expected to grow at the fastest rate in the high potency active pharmaceutical ingredients market during the forecast period due to the robust research and development in diverse areas like regenerative medicine, cell and gene therapy, oncology, precision medicine, nanotechnology, genetic engineering, agriculture, food, and cosmetics. Biotechnology is revolutionizing healthcare by providing life-saving solutions and services to prevent and treat severe health conditions. It is impacting globally by positioning itself as the global leader in clinical research, pharmaceuticals, diagnostics, and therapeutics.
Manufacturer Type Insights
|
By Manufacturer Type |
Shares 2025 (%) |
|
In-house |
60% |
|
Outsourced |
40% |
Explanation:
• In-house dominates with a 60% share, as pharmaceutical companies prefer to maintain control over production processes, ensuring quality and regulatory compliance.
• Outsourced holds 40% of the market, growing as companies seek to reduce costs and leverage specialized manufacturing expertise offered by contract manufacturers.
Brief Reasonings:
The in-house segment led the high potency active pharmaceutical ingredients market share by 60% in 2025, owing to the presence of large pharmaceutical companies and the increased production of high-value and sensitive compounds, internally to protect assets. In-house manufacturing is the preferred choice for innovative and proprietary drugs. It is essential to maintain strict control over intellectual property.
The outsourced segment is expected to grow at the fastest rate in the high potency active pharmaceutical ingredients market during the forecast period due to the surging importance of high potency APIs in the pharmaceutical industry to develop more targeted treatments for chronic diseases like cancer. The expanding R&D in oncology and targeted therapies, and global supply chains, are driving the growth of outsourcing. The smaller pharmaceutical firms that lack high potency API capabilities are increasingly outsourcing manufacturing.
Drug Type Insights
|
By Drug Type |
Shares 2025 (%) |
|
Innovative |
65% |
|
Generic |
35% |
Explanation:
• Innovative leads with 65%, driven by the ongoing demand for new, patented drugs that offer unique therapeutic benefits, particularly in oncology and rare diseases.
• Generic represents 35%, with steady demand as the market for cost-effective alternatives to branded drugs continues to expand.
Brief Reasonings:
The innovative segment dominated the high potency active pharmaceutical ingredients market by 65% in 2025, owing to the significant efforts of large pharmaceutical companies for the development of antibody drug conjugates, which drive innovation by utilizing high potency APIs. A rapid shift towards precision medicine and targeted therapies, including oncology, is driving the production of innovative and life-saving drugs. There is a surge in the production of synthetic small molecules and biotechnology-derived high potency APIs, such as proteins, peptides, and oligonucleotides.
The generic segment is expected to grow rapidly in the high potency active pharmaceutical ingredients market during the studied period due to the rising demand for cost-effective treatments for chronic diseases like cancer. The high-value biologics and small molecules are enabling the production of generics and biosimilars. Manufacturers are prioritizing the production of complex generics like, inhalers and long-acting injectables over simple generics to maintain higher margins.
Application Insights
|
By Application |
Shares 2025 (%) |
|
Oncology |
50% |
|
Hormonal |
15% |
|
Glaucoma |
25% |
|
Others |
10% |
Explanation:
• Oncology dominates the market with a 50% share, fueled by the increasing prevalence of cancer and the growing pipeline of high-potency oncology drugs.
• Hormonal holds 15%, driven by the demand for hormonal treatments for conditions like menopause, fertility, and hormone-related cancers.
• Glaucoma accounts for 25%, with increasing demand for treatments due to the rising incidence of glaucoma and other ocular diseases.
• Others represents 10%, covering a range of niche applications, though it holds a smaller share compared to oncology and glaucoma.
Brief Reasonings:
The oncology segment led the high potency active pharmaceutical ingredients market share by 50% in 2025, owing to the essential role of high potency APIs in cancer treatments, like chemotherapy and immunotherapy. The high potency APIs reduce dosage frequency, minimize toxicity, and enhance efficacy. The APIs target specific pathways in cancer cell proliferation, which leads to more effective treatments with fewer side effects.
The glaucoma segment is expected to grow at a rapid rate in the high potency active pharmaceutical ingredients market during the forecast period due to the impact of several emerging technologies like AI, home monitoring, and sustained-release drug delivery on the detection, monitoring, and treatment of glaucoma. Research makes efforts for an automated detection of glaucoma progression from optical coherence tomography. A sustained release or controlled release drug delivery in glaucoma conditions is achieved through intracameral implants and next-generation depots.
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
Top Companies in the Market

• BASF SE
• CordenPharma
• BristolMyers Squibb
• CARBOGEN AMCIS AG
• Pfizer, Inc.
• Boehringer Ingelheim
• Dr. Reddy’s Laboratories
• Lonza Group AG
• Novartis AG
• Sanofi
• AbbVie Inc.
• Merck KGaA
• Cambrex & Piramal Pharma Solutions
Segments Covered in the Report
By Product
• Synthetic
• Biotech
By Manufacturer Type
• In-house
• Outsourced
By Drug Type
• Innovative
• Generic
By Application
• Oncology
• Hormonal
• Glaucoma
• Others
By Region
• North America
• Europe
• Asia Pacific
• Latin America
• Middle East & Africa
Immediate Delivery Available | Buy This Research Report Now @ https://www.towardshealthcare.com/checkout/6694
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region: +44 778 256 0738
North America Region: +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Our Trusted Data Partners
Precedence Research | Towards Packaging | Towards Food and Beverages | Towards Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Browse More Insights from Towards Healthcare:
✅ The API CDMO market reached USD 136.92 billion in 2025 (from USD 127.45 billion in 2024) and is expected to hit USD 260.98 billion by 2034, growing at a CAGR of 7.43%.
✅ The pharmaceutical ERP market was valued at USD 2.96 billion in 2025 and is projected to grow to USD 9.66 billion by 2035 at a CAGR of 12.56%.
✅ The pharmaceutical-grade melatonin market grew from USD 479.9 million in 2024 to USD 521 million in 2025 and is forecast to reach USD 1,044.3 million by 2034, at a CAGR of 8.56%.
✅ The U.S. pharmaceutical market is expected to expand from USD 553.55 billion in 2026 to USD 965 billion by 2035, registering a CAGR of 6.37%.
✅ The U.S. pharmaceutical CDMO market is projected to grow from USD 41.65 billion in 2026 to USD 72.98 billion by 2035 at a CAGR of 6.43%.
✅ The pharmaceutical excipients market is set to rise from USD 11.27 billion in 2026 to USD 16.12 billion by 2035, growing at a CAGR of 4.06%.
✅ The pharmaceutical filtration market is anticipated to grow from USD 14.95 billion in 2025 to USD 32.51 billion by 2035 at a CAGR of 8.08%.
✅ The pharmaceutical spray drying market increased from USD 2.37 billion in 2024 to USD 2.55 billion in 2025 and is expected to reach USD 4.93 billion by 2034, at a CAGR of 7.67%.
✅ The contract pharmaceutical manufacturing market is projected to grow from USD 255.41 billion in 2026 to USD 599.3 billion by 2035, at a CAGR of 9.94%.
✅ The biotechnology & pharmaceutical services market grew from USD 76.51 billion in 2024 to USD 80.7 billion in 2025 and is forecast to reach USD 130.56 billion by 2034, at a CAGR of 5.48%.




