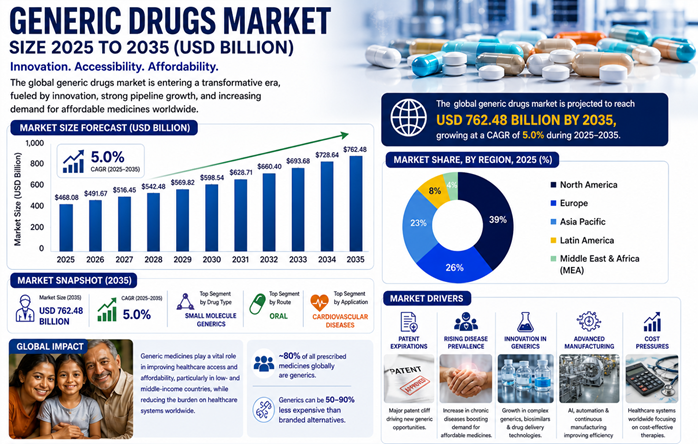
The generic drugs market is entering a transformative phase, driven by innovation and rising demand for affordable healthcare. Advances in biosimilars and AI-powered manufacturing are improving access and efficiency. With growing chronic diseases and patent expirations.
According to Precedence Research, the global generic drugs market size is expected to grow from USD 491.67 billion in 2026 to nearly USD 762.48 billion by 2035, with a CAGR of 5% over the next decade.
The industry is undergoing a structural transformation from high-volume, low-margin commoditized drugs to high-value, complex generics and biosimilars creating new opportunities for pharmaceutical companies, investors, and healthcare systems worldwide.
Across the globe, the generic drugs market is experiencing a significant transformation, shifting from commoditized small molecules to high-value, complex generics and biosimilars. This change is primarily driven by the expiration of patents for major biologics in oncology and chronic disease management. To mitigate pricing pressures and meet stringent FDA/EMA quality standards, manufacturers are accelerating the adoption of automated, continuous manufacturing and AI-driven bioequivalence modeling. These strategies aim to increase efficiency, shorten development cycles, and ensure long-term sustainability.
Where Data Meets Strategic Clarity 📥 View Sample Pages of the Complete
Report 👉https://www.precedenceresearch.com/sample/1205
Generic Drugs Market Highlights:
🔹North America dominated with the largest market share of 39% in 2025.
🔹Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period.
🔹By drug type, the simple generics segment contributed the highest market share in 2025.
🔹By drug type, the super generics segment is growing at a strong CAGR between 2026 and 2035.
🔹By brand, the pure generic drug segment held a major market share in 2025.
🔹By brand, the branded generic drugs segment is expected to expand at a notable CAGR from 2026 to 2035.
🔹By route of drug administration, the oral segment captured the highest market share in 2025.
🔹By route of drug administration, the injection segment is poised to grow at a healthy CAGR between 2026 and 2035.
🔹By therapeutic application, the cardiovascular segment generated the biggest market share in 2025.
🔹By therapeutic application, the oncology segment is expanding at the fastest CAGR between 2026 and 2035.
🔹By distribution channel, the retail pharmacy segment accounted for the largest market share in 2025.
🔹By distribution channel, the hospital pharmacy segment is projected to grow at a solid CAGR between 2026 and 2035.
Market Overview: The Expanding Power of Generic Drugs
The generic drugs market, which comprises non-branded, affordable alternatives to brand-name medications that are chemically identical in active ingredients, dosage, strength, and route of administration, is expanding rapidly. This growth is fueled by the rising prevalence of chronic diseases, cost-containment measures, and a significant patent cliff affecting high-revenue drugs. These products offer identical therapeutic effects at a fraction of the cost and are increasingly dominating the pharmaceutical sector, with the Asia-Pacific region serving as a major manufacturing hub.
➡️ Become a Valued Research Partner with Us https://www.precedenceresearch.com/schedule-meeting
Investing in Complex Generics and Biosimilars: Major
Potential
The market
is shifting toward complex generics and biosimilars, largely driven by the
upcoming patent expirations of blockbuster biologics. This high-value segment
promises higher profit margins due to increased technical and regulatory
barriers. With advancements in AI-driven modeling and drug delivery, the
biosimilar market is projected to expand significantly - especially in oncology
and autoimmune therapies. Pharmaceutical firms are focusing on these areas to
ensure long-term, sustainable growth. The High
Cost of Narrow Sourcing: Major Limitations The generic
drugs market faces substantial limitations due to intense competition, which
causes severe price erosion, often dropping below production costs upon market
entry. This scenario, combined with high capital expenditures for research and
development and quality compliance, heavily compresses profit margins and leads
to industry consolidation. Additionally, this economic pressure results in
strategic vulnerabilities, creating significant supply chain risks in the event
of regional disruptions. 🔗 Stay Ahead of the Market
Curve — Get the Full Report 👉 https://www.precedenceresearch.com/generic-drugs-market Generic
Drugs Market Leading Companies ➢ Mylan N.V. ➢ Abbott
Laboratories ➢ ALLERGAN ➢ Teva
Pharmaceutical Industries Ltd. ➢ Eli Lilly and
Company ➢ STADA
Arzneimittel AG ➢ GlaxoSmithKline
Plc. ➢ Baxter
International Inc. ➢ Pfizer Inc. ➢ Sandoz
International GmbH Value
Chain Analysis: Generic Drugs Market The generic
drugs market operates through a multi-stage value chain, encompassing research
and development, distribution, and patient-centric services. Each stage
involves specialized processes and key industry participants. Research
& Development (R&D) 🔸This initial stage focuses on
strategic planning and candidate selection, followed by pharmaceutical
development and bioequivalence studies. It also includes clinical evaluations,
scale-up processes, manufacturing readiness, and regulatory submission and
review. 🔸Key companies driving this segment
include Sandoz, Sun Pharma, Fosun Pharma, Viatris, Aurobindo Pharma, Teva
Pharmaceuticals, Cipla, and Lupin. Distribution
to Hospitals and Pharmacies 🔸This stage encompasses the core
supply chain operations, including the transfer of products from manufacturers
to distributors, warehousing, order management, and last-mile delivery to
hospitals and retail pharmacies. 🔸Major participants in this segment
include McKesson Corporation, Cencora, Cardinal Health, along with
manufacturers such as Sandoz, Teva Pharmaceuticals, Viatris, Sun Pharma, and
Fosun Pharma. Patient
Support & Services 🔸The final stage focuses on enhancing
patient outcomes through support programs. This includes patient enrollment,
awareness initiatives, access and affordability programs, therapy initiation,
education, adherence monitoring, and long-term treatment maintenance. 🔸Key organizations operating in this
space include Fortrea, AssistRx, CareMetx LLC, ConnectiveRx, Lash Group,
McKesson Corporation, Cardinal Health, EVERSANA, and United BioSource LLC. Generic
Drugs Market Report Coverage Report Coverage Details Market Size in 2025 USD 468.08 Billion Market Size in 2026 USD 491.67 Billion Market Size by 2035 USD 762.48 Billion Market CAGR (2026–2035) CAGR of 5% Largest Market North America Fastest Growing Region Asia-Pacific Base Year 2025 Forecast Period 2026 to 2035 Segments Covered Drug, Brand, Route of Drug
Administration, Therapeutic Application, Distribution Channel and Regions Regions Covered North America, Europe, Asia-Pacific,
Latin America, Middle East & Africa Key Growth Drivers Rising chronic diseases,
patent expirations, and strong demand for low-cost alternatives are driving
generic drug adoption globally. Key Technology Trends AI-based modeling, continuous
manufacturing, and advances in complex generics and biosimilars are improving
efficiency and innovation. Major Opportunity Biosimilars and complex
generics offer high-margin growth, supported by biologics patent expirations
and emerging market demand. Key Challenge Severe price erosion, API
dependency, and strict regulatory requirements continue to pressure margins
and delay approvals. Leading Market Participants Teva Pharmaceutical
Industries Ltd., Pfizer Inc., Sandoz International GmbH, Abbott Laboratories,
and Mylan N.V.
Have questions or ready to move forward? Contact our experts
today @ sales@precedenceresearch.com | +1 804
441 9344 Generic Drugs
Market: Regional Analysis North America
dominated the market in 2025, primarily due to high healthcare expenditure, an aging
population, and a regulatory environment that prioritizes cost-effective,
high-quality alternatives to branded drugs. The high prevalence of chronic
conditions, such as diabetes, hypertension, and cardiovascular diseases,
necessitates long-term medication, driving demand for affordable, sustainable
treatment options. High drug costs put immense pressure on payers to promote
cheaper alternatives, while the patent cliff ensures a continuous pipeline of
new generic products. As the
world's largest generic drug market, the U.S. dominates prescriptions filled
by generics, driven by high demand from an aging population and cost-saving
initiatives. Regulated by the FDA's OGD to ensure quality, the market is
shifting toward complex generics, biosimilars, and automated manufacturing to
meet the growing need for affordable treatments. Get informed with deep-dive intelligence on AI’s market
impact 👉 https://www.precedenceresearch.com/ai-precedence U.S.
Generic Drugs Market Size and Growth 2025 to 2035 The U.S. generic drugs market size is expected to touch USD 243.70
billion by 2035 increasing from USD 153.40 billion in 2026, The market is
poised to grow at a CAGR of 5.25% between 2026 and 2035. The U.S.
Generic Drugs Market Report is Readily Available 📥 Download Sample Pages of the Report 👉 https://www.precedenceresearch.com/us-generic-drug-market Report Coverage Details Market Size in 2025 USD 146.04 Billion Market Size in 2026 USD 153.40 Billion Market Size by 2035 USD 243.70 Billion Growth Rate (2026–2035) CAGR of 5.25% Base Year 2025 Forecast Period 2026 to 2035 Segments Covered By Drug Type, By Brand,
By Route of Administration, By Therapeutic Application, By Distribution
Channel Market Position Largest global generic
drugs market with strong FDA regulatory framework Prescription Share Generics account for 90%+
of total prescriptions in the U.S. Cost Advantage Generics are 80–85%
lower in cost compared to branded drugs Key Growth Drivers Patent expirations,
rising chronic diseases, aging population, demand for affordable therapies Innovation Trends Growth in biosimilars,
complex generics, AI-driven drug development, and continuous manufacturing Regulatory Landscape Strong support through
FDA initiatives such as GDUFA III enhancing approvals and innovation Healthcare Impact Improves accessibility,
reduces healthcare costs, and supports sustainable healthcare systems
Leading
Players Shaping the U.S. Generic Drugs Market The U.S.
generic drugs market is driven by a combination of global pharmaceutical
leaders and cost-efficient manufacturers, focusing on affordability,
innovation, and supply reliability. 🔹 Key Market Participants ➢ Pfizer Inc.: Focuses on hospital-based
generics, particularly sterile injectables and anti-infectives. Its strong
domestic manufacturing network supports consistent supply and high-quality
standards. ➢ Teva Pharmaceuticals USA Inc.: A leading generics
provider with a broad portfolio across CNS, oncology, and cardiovascular
therapies. Increasingly focused on complex generics and specialty medicines. ➢ Aurobindo Pharma USA Inc.: Specializes in high-volume
oral solids and injectable generics. Its vertically integrated model enables
cost efficiency and competitive pricing. Other Major Key Players ➢ Sun Pharma Inc.: Expanding its U.S.
presence with a diversified generics and specialty portfolio. ➢ Abbott Laboratories Inc.: Maintains a strong
position through global reach and trusted healthcare solutions. ➢ Lupin Pharmaceuticals, Inc.: Focused on respiratory and
cardiovascular segments, with growing investments in complex generics. ➢ Mylan (Viatris): A major supplier of
affordable medicines, offering a wide and diversified generics portfolio. ➢ Dr. Reddy’s Laboratories: Strengthening its
footprint through biosimilars and advanced generic formulations. ➢ Novartis (Sandoz Division): A global leader in biosimilars
and complex generics, driving innovation in high-value segments. ➢ Eli Lilly and Company: Expanding into generics
and biosimilars through strategic partnerships and product development
initiatives. 🔓 Instant Access. Zero Waiting. 📥 Buy the Premium Market
Research Report Now 👉 https://www.precedenceresearch.com/checkout/2486 U.S. Generic Drugs Market Segments Covered in the Report By Drug Type • Simple Generics • Super Generics By Brand • Pure Generic Drugs • Branded Generic Drugs By Route of Administration • Oral • Injection • Cutaneous • Others By Therapeutic Application • Central Nervous System (CNS) • Cardiovascular • Infectious Diseases • Musculoskeletal Diseases • Respiratory • Oncology • Others By Distribution Channels • Retail Pharmacy • Hospital Pharmacy • Online and Others Browse Our Subscription Plans@ https://www.precedenceresearch.com/get-a-subscription Canada
holds a significant position in the generic drug market, characterized by high utilization
rates and a robust regulatory framework under Health Canada that ensures safety
and cost management. The sector leverages a strong manufacturing footprint and
an aggressive shift toward biosimilars to secure savings for complex
treatments. As the
world's largest provider of generic drugs, India holds a dominant global
position by supplying export volume and boasting the highest number of U.S.
FDA-approved manufacturing sites. Renowned for affordable, high-quality
medications, particularly in vaccines, cardiovascular, and antibiotic segments, the industry is critical
to global health security, as India remains heavily dependent on China for
approximately two-thirds of its APIs and key starting materials. 🔸 In March 206, the Production Linked Incentive
(PLI) Scheme is a strategic initiative aimed at reducing import reliance on
China by offering financial incentives to boost domestic manufacturing of APIs,
Key Starting Materials, and drug intermediates. (Source: https://www.pib.gov) As the
world’s leading manufacturer and exporter of APIs, China dominates the global
generic drug supply chain by providing the vast majority of critical ingredients
for essential medicines. While maintaining unmatched cost efficiency and scale,
China is aggressively shifting its market strategy to move up the value chain
by rapidly upgrading quality standards, strengthening regulatory compliance in
advanced manufacturing and biotech technologies.
Generic Drugs Market: Segmentation Analysis
By Drug Type Analysis
The simple generics segment dominated the market in
2025, largely due to high demand for
affordable treatments, widespread patent expirations on blockbuster drugs, and
established manufacturing pipelines. These generics provide the same therapeutic
effects as branded drugs but at a significantly lower cost, driving their
adoption among patients and healthcare systems aiming to control spending. The
rising prevalence of chronic conditions ensures a consistent, high-volume
demand for these often oral medications.
The super generics segment is expected to witness the
fastest growth during the forecast period. This growth is primarily driven by the intense demand for improved
patient compliance and enhanced bioavailability. Also known as value-added generics,
super generics often enhance the original drug's formulation, such as improving
its absorption rate, making them superior to standard generics. The growing
number of brand-name drug patents expiring, combined with the rising prevalence
of chronic conditions, is driving demand for these superior alternatives.
By Brand
Analysis The pure
generic drug segment led the market in 2025, primarily driven by affordability and bioequivalence
to branded counterparts. Pure generics, which do not have a brand name and
typically use the generic name, are preferred for controlling healthcare costs.
The expiration of brand-name patents creates significant market opportunities,
allowing for the rapid adoption of affordable alternatives. There is also high demand
for low-cost, effective medication in emerging economies, further enhancing the
market share of pure generic products. The
branded generic drugs segment is expected to experience the fastest growth
during the forecast period. This growth is fueled by the increasing demand for cost-effective
alternatives to branded drugs, the rising burden of chronic diseases, and
improved regulatory approval rates. Patients and healthcare providers prefer
lower-cost branded alternatives, which boosts demand. Branded generics offer
familiar names, fostering greater trust and loyalty among consumers, while the
patent cliff enables many high-revenue branded drugs to transition to generic
manufacturing. By Route
of Drug Administration Analysis The oral
segment accounted for the largest market share in 2025. This is largely due to the
unparalleled convenience, cost-effectiveness, and high patient adherence
associated with oral tablets, capsules, and liquids. Oral medications are
non-invasive and easy to self-administer, making them preferable for patients,
especially those managing chronic diseases that require long-term, daily
medication. Additionally, oral solid dosage forms such as tablets and capsules
are generally easier and cheaper to manufacture, store, and transport, contributing
to lower pricing. The
injection segment is projected to experience the fastest growth during the
forecast period.
This growth is mainly driven by the increasing demand for biosimilars, complex
generics, and therapies for chronic diseases that require high bioavailability.
There is substantial demand for injectable medications in the treatment of
cancer, diabetes, and autoimmune disorders, driving adoption. Injections
provide an immediate onset of action and are ideal for drugs that are poorly absorbed
when taken orally, ensuring 100% bioavailability in many cases, which allows
for the rapid rise of cheaper injectable alternatives. By
Therapeutic Application Analysis The
cardiovascular segment led the market in 2025, primarily due to the high prevalence rates of
chronic conditions such as hypertension and high cholesterol, which ensure
sustained long-term demand. Cardiovascular diseases are a leading cause of
mortality worldwide, necessitating ongoing medication use and ensuring a steady
revenue stream. The generic cardiovascular segment benefits from the loss of
patent protection for numerous blockbuster drugs, facilitating the rapid
introduction of cheaper, bioequivalent alternatives. The oncology
segment is experiencing the fastest growth during the forecast period, primarily due to the high
prevalence of cancer and the expiration of patents on blockbuster drugs.
Cost-effective, high-quality generic alternatives are replacing expensive
brand-name drugs, driving demand. Additionally, the increasing adoption of
targeted therapies and biosimilars further accelerates market growth. The
introduction of biosimilars, generic alternatives for complex biologic drugs, is
transforming the market and encouraging the adoption of generic drugs. By Distribution
Channel Analysis The
retail pharmacy segment dominated the market in 2025, primarily due to its ability to
provide convenient access to affordable, daily-use medications for chronic
diseases. The widespread availability of retail pharmacies, along with trusted
counseling services and high consumer foot traffic, facilitates quick access to
and fulfillment of prescriptions. Retailers often offer competitive pricing and
discounts on generic drugs, making them the primary channel for lower-cost trade
generics, which helps consumers manage their healthcare costs. The
hospital pharmacy segment is expected to witness the fastest growth during the
forecast period.
This growth is driven by increasing hospital admissions, rising demand for
inpatient care, and the consolidation of purchasing power through institutional
group purchasing organizations. Hospital pharmacies play a crucial role in the
growing use of specialty generics, including oncology and parenteral drugs for
complex therapies in acute care settings. The rising number of hospital
admissions and the need for immediate access to treatments necessitate a strong
reliance on hospital pharmacies. ✚ Related
Topics You May Find Useful: ➡️ Inhalation and Nasal
Spray Generic Drugs Market: Explore how respiratory therapies and convenient drug delivery
formats are shaping generic drug adoption ➡️ Antibody Drug
Conjugates Market:
Discover advancements in targeted cancer therapies combining precision
biologics with potent cytotoxic agents ➡️ Chemical Drug CDMO
Market: Understand the
growing role of outsourcing in drug development and manufacturing efficiency ➡️ Transdermal Drug
Delivery Systems Market:
Analyze the shift toward non-invasive delivery methods improving patient
compliance ➡️ Nasal Drug Delivery
Technology Market:
Examine innovations enabling rapid absorption and enhanced therapeutic
effectiveness ➡️ Drug Discovery
Informatics Market:
See how data-driven technologies and AI are transforming pharmaceutical
research processes ➡️ Antipsychotic Drugs
Market: Gain insight
into treatment advancements addressing mental health disorders worldwide ➡️ Genitourinary Drugs
Market: Track evolving
therapies targeting urinary and reproductive health conditions Emerging Innovation Description and Focus Area Application Area 3D Printing for Personalised Dosages On-demand manufacturing and patient compliance.
Allows precise dosing, customized shapes, and rapid-dissolving forms for
special populations. Spritam (levetiracetam): 3D printed for rapid
dissolution; FabRx oral thin films for personalized doses. Continuous Manufacturing (CM) Quality and efficiency. Replaces batch processes
with seamless, automated flow, enhancing quality assurance (QbD) and lowering
costs. Small molecule tablets: CM is used to cut production
times by up to 50% for high-volume generic drugs. Nanotechnology in Drug Delivery Bioavailability and targeting. Encapsulating APIs in
nano-carriers improves solubility and targeted delivery, specifically for
poorly soluble drugs. Abraxane (paclitaxel): Albumin-bound nanoparticle
formulation; SUBACAP (itraconazole) for enhanced solubility. AI-Driven Bioequivalence (BE) Modeling Regulatory approval speed. AI/ML algorithms analyze
datasets to simulate pharmacokinetic studies, reducing the need for costly
human trials. FDA BEAM Tool: Bioequivalence Assessment Mate to
automate and accelerate ANDA approvals. Biosimilars and Complex Generic Development Access to high-value biologics. Developing generic
alternatives to complex biologics facing patent cliffs in oncology and
immunology. Stelara (Ustekinumab) biosimilars: Targeted for
launch following patent expiration.
Major
Shifts in the Generic Drugs Market 🔸In March 2026, the first wave of generic versions
of Novo Nordisk’s GLP-1 weight-loss drugs launched in India, with domestic
drugmakers cutting prices by up to 80% after the patent expired. This is
crucial, with around 100 million people living with diabetes and a significant
obesity rate and aims to be among the top players in this space. (Source: https://www.cnbc.com) 🔸In March 2026, Hyderabad-based pharma major Dr
Reddy’s Laboratories (DRL) has launched its semaglutide generic, Obeda, for
Type 2 diabetes management in India. The launch comes a day after semaglutide
lost its patent protection. The cost to the patient for Obeda is ₹4,200 per
month for its 2 milligram (mg) and 4 mg versions- a 62 per cent drop from the
highest dosage form of the innovator drug Ozempic. (Source: https://www.business-standard.com)
🔸 In January 2026, Dr. Reddy’s also announced the U.S.
launch of olopatadine hydrochloride (HCl) ophthalmic solution, a generic
version of Alcon’s Pataday, marking its first-to-market launch. It is an
over-the-counter eye drop for allergy relief approved for use in adults and
children aged 2 and above. (Source: https://glance.eyesoneyecare.com) Generic
Drugs Market Segmentation By Drug
Type 🔹Simple Generics 🔹Super Generics By Brand 🔹Pure generic drugs 🔹Branded generic drugs By Route
of Drug Administration 🔹Oral 🔹Injection 🔹Cutaneous 🔹Others By
Therapeutic Application 🔹Central Nervous System (CNS) 🔹Cardiovascular 🔹Infectious Diseases 🔹Musculoskeletal Diseases 🔹Respiratory 🔹Oncology 🔹Others By
Distribution Channel 🔹Retail Pharmacy 🔹Hospital Pharmacy 🔹Online and Others By Region 🔹North America 🔹Asia Pacific 🔹Europe 🔹Latin America 🔹Middle East and Africa (MEA) Thanks for reading you can also get individual
chapter-wise sections or region-wise report versions such as North America,
Europe, or Asia Pacific. Don’t Miss Out! | Instant Access to This
Exclusive Report 👉 https://www.precedenceresearch.com/checkout/1205 You can place an order or ask any questions, please feel free
to contact at sales@precedenceresearch.com | +1 804 441 9344 Stay Ahead with Precedence Research Subscriptions Unlock
exclusive access to powerful market intelligence, real-time data, and
forward-looking insights, tailored to your business. From trend tracking to competitive
analysis, our subscription plans keep you informed, agile, and ahead of the
curve. Browse
Our Subscription Plans@ https://www.precedenceresearch.com/get-a-subscription About Us Precedence
Research is a global market intelligence and consulting powerhouse, dedicated
to unlocking deep strategic insights that drive innovation and transformation.
With a laser focus on the dynamic world of
life sciences,
we specialize in decoding the complexities of cell and
gene therapy,
drug development, and oncology markets, helping our clients stay ahead in
some of the most cutting-edge and high-stakes domains in healthcare. Our
expertise spans across the biotech and pharmaceutical ecosystem, serving
innovators, investors, and institutions that are redefining what’s possible in regenerative medicine, cancer care, precision
therapeutics, and beyond. Web: https://www.precedenceresearch.com Our
Trusted Data Partners: Towards Healthcare | Nova One Advisor | Statifacts Get
Recent News 👉 https://www.precedenceresearch.com/news For
Latest Update Follow Us:

🔸 In
January 2026, GDUFA
Regulatory Science Initiatives under GDUFA III are prioritizing the integration
of AI, machine learning, and advanced manufacturing
technologies to modernize generic drug development and assessment, aiming to
enhance efficiency and facilitate the development of complex generic medicines.
(Source: https://www.fda.gov)
U.S. Generic Drugs Market Scope and Key Insights:
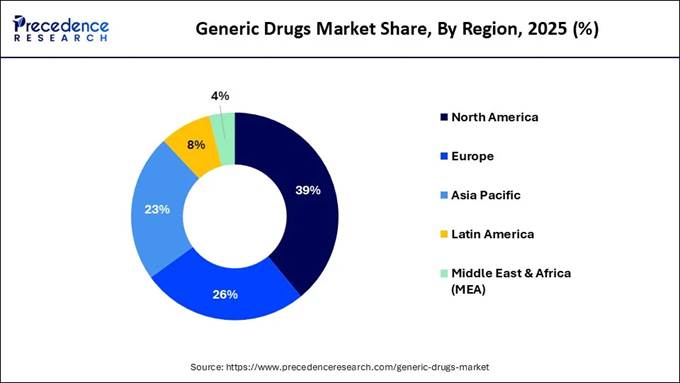
Asia-Pacific region is anticipated to experience the fastest growth during the
forecast period. This growth is driven by a massive population, an increasing
burden of chronic diseases, significant government initiatives to reduce
healthcare costs, and a strong local manufacturing base. Governments are advocating
for affordable treatments to minimize overall health spending, especially given
the high prevalence of chronic illnesses requiring long-term therapies. India
and China are emerging as global hubs for generic drug manufacturing, offering
competitive pricing due to skilled labor and established supply chains.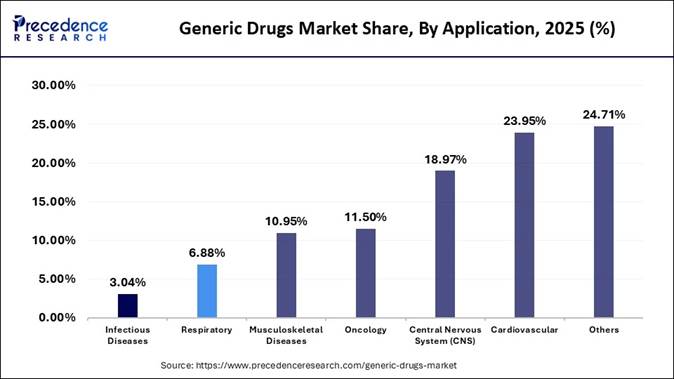
Emerging innovations in the Generic Drugs


