
Frederick National Laboratory for Cancer Research
A rewarding career with global impact.
Whether you’re an expert in your field or just starting out, we have a career opportunity for you. We’re always looking for people to join us in fulfilling the mission of the Frederick National Laboratory: discovery, innovation, and success in the biomedical sciences.
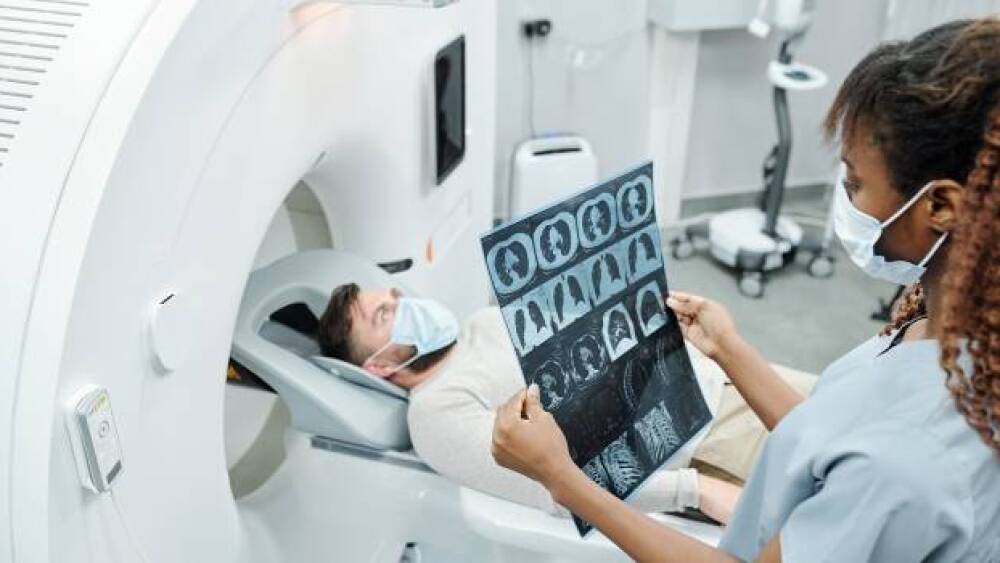
Our team of 2,400+ scientists, technicians, administrators, and support staff work at the forefront of basic, translational, and preclinical science, with a focus on cancer, AIDS, and other infectious diseases.
We collaborate with colleagues across the National Cancer Institute, National Institutes of Allergy and Infectious Diseases, and others throughout the National Institutes of Health. We also engage with extramural investigators in academia, government and industry.
Your path to joining our team begins with the desire to work for the only national laboratory dedicated to biomedical research. Our employees share a common desire to help make a difference in cancer research and public health concerns. As you search for a career that fits your education, skills, and abilities, explore the core values that guide us and emphasize work-life balance.
Discover why joining the Frederick National Laboratory team could be the most important career step you take.
Our core values of accountability, compassion, collaboration, dedication, integrity, and versatility serve as a guidepost for how we do our work every day in serving the public’s interest. These values unite us and set us up for continued success.
-
“It is incredibly satisfying to know that our efforts have a real impact. The data we generate is used by treating physicians to make informed treatment decisions, putting patients on therapies that are most likely to benefit them based on our sequencing data. I often remind my team that we are genuinely making a difference.”
Chris Karlovich, Ph. D.
Director, Molecular Characterization Laboratory