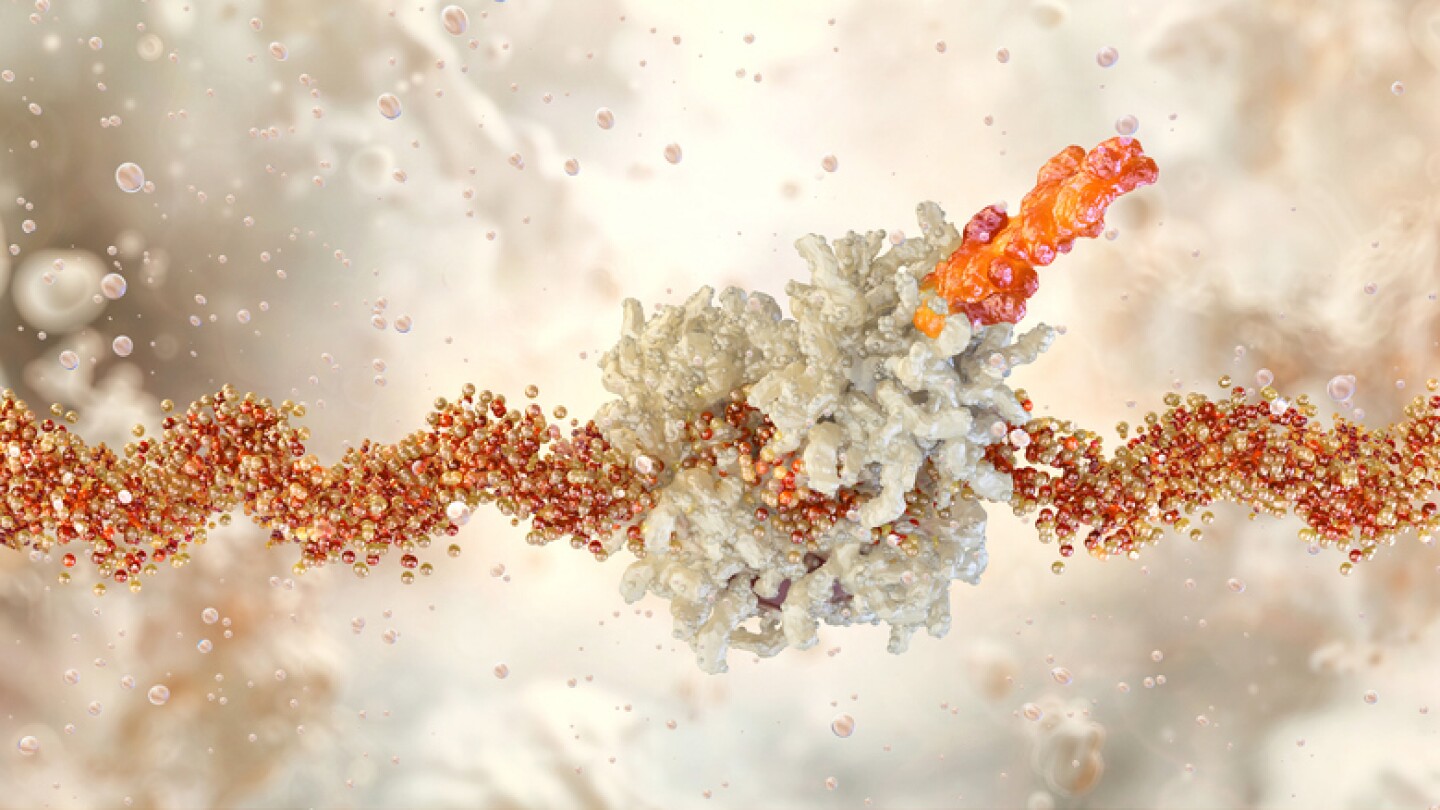
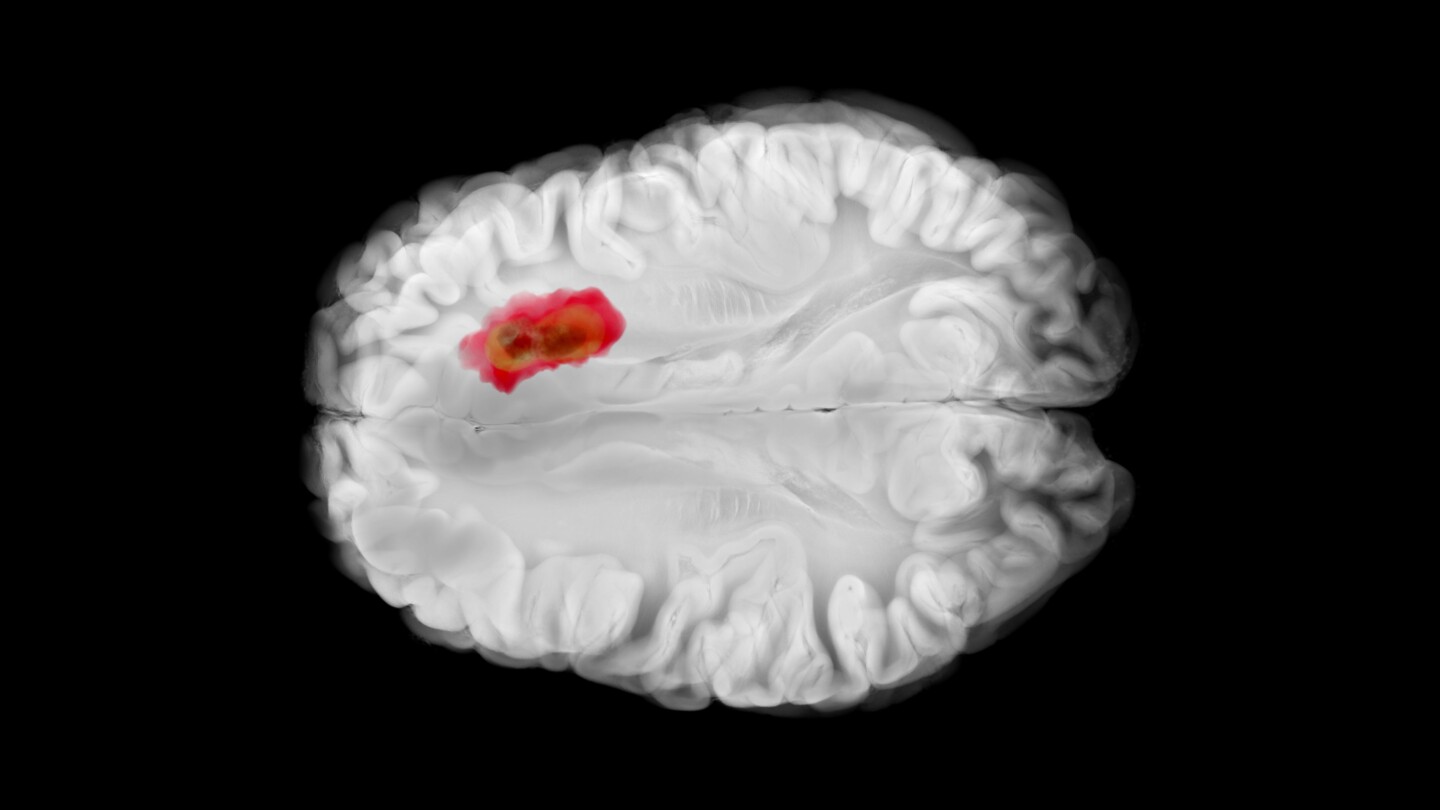
Cancer
VantAI will use its machine learning capabilities to identify novel target-effector pairs that Halda can use in designing its bifunctional small-molecule drugs.
The Department of Health and Human Services’ mRNA pullback only applies to their use in upper respiratory disease, according to Secretary Robert F. Kennedy Jr.
After two patients who received the investigational CDC7 blocker died, pushing forward with SGR-2921’s development would be “difficult,” according to Schrödinger, whose stock dropped 17.5% before the opening bell on Thursday.
For $1.3 billion in aggregate—including upfront and milestone payments—Bayer will get exclusive global access to Kumquat Biosciences’ small-molecule KRAS G12D blocker.
Phase Ib data show Hernexeos can elicit a confirmed objective response rate of 44% in patients with HER2-mutated NSCLC who had previously been treated with a directed antibody-drug conjugate.
The recent announcement of RFK Jr.’s termination of mRNA vaccine contracts is the latest effort to undermine this promising technology at the federal level. Pharmaceutical companies and private investors must fill the gap and ensure that research into this critical resource continues.
Arguably the FDA’s most anticipated decision this month is for a subcutaneous induction formulation of Biogen and Eisai’s Alzheimer’s drug Leqembi, which, according to Eisai, could “help reduce the burden on healthcare professionals and patients.”
Strand Therapeutics’ lead asset is STX-001, an intra-tumor self-replicating mRNA therapy that carries a payload expressing the immunomodulatory protein IL-12.
The small molecule drug, acquired by Jazz Pharmaceuticals in its $935 million Chimerix pick-up this spring, is intended for relapsed adult and pediatric patients with H3 K27M mutations.
Terns, once a rising star in obesity and the MASH space, will refocus on cancer and partner out a handful of obesity assets.
PRESS RELEASES