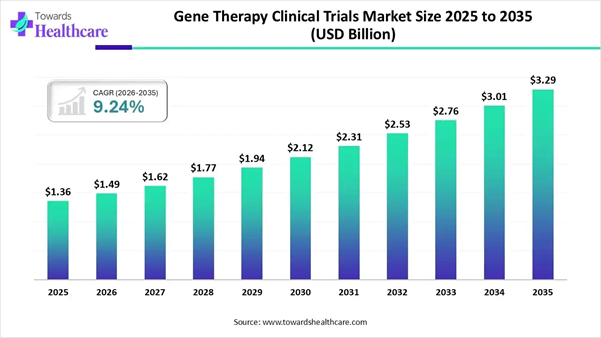
The gene therapy clinical trials market was valued at USD 1.36 billion in 2025 and is expected to reach USD 1.49 billion in 2026. It is further projected to grow to approximately USD 3.29 billion by 2035, expanding at a CAGR of 9.24% during the forecast period.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/6068
The clinical trials for gene therapy are important before it enters the global market. The different phases of clinical trials of gene therapy products are unique, which involve chemistry, preclinical challenges, manufacturing, clinical safety questions, and control processes. They are expanding due to the vast research studies on genetic diseases and cancer. Gene therapy clinical trials are performed for specific indications, including lymphoma, Non-Hodgkin's disease, unspecified solid tumor, leukemia, multiple myeloma, ovarian cancer, colorectal cancer, lupus, pancreatic cancer, and more.
The Gene Therapy Clinical Trials Market: Highlights
• Gene therapy clinical trials market to crossed USD 1.49 billion by 2026.
• Market projected at USD 3.29 billion by 2035.
• CAGR of 9.24% expected in between 2026 to 2035.
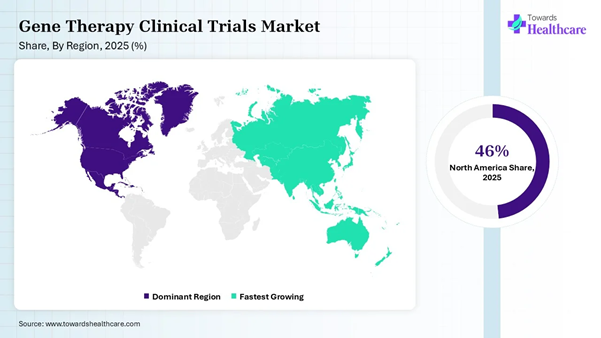
• North America held a major revenue share of approximately 46% in the market in 2025.
• Asia-Pacific is expected to host the fastest-growing market with the fastest CAGR in the coming years.
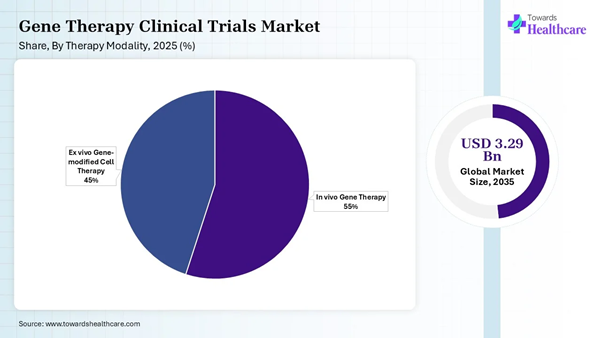
• By therapy modality, the in vivo gene therapy segment dominated the gene therapy clinical trials market with a share of approximately 55% in 2025.
• By therapy modality, the ex vivo gene therapy segment is expected to expand rapidly in the market in the coming years.
• By vector/delivery platform, the AAV (adeno-associated virus) segment led the market with a share of approximately 45% in 2025.
• By vector/delivery platform, the genome editing segment is expected to witness the fastest growth in the market over the forecast period.
• By therapeutic area, the oncology segment accounted for the highest revenue share of approximately 35% in the market in 2025.
• By therapeutic area, the neurology/CNS segment is expected to show the fastest growth in the gene therapy clinical trials market over the forecast period.
• By clinical phase, the phase I/ I/II segment held a dominant revenue share of approximately 60% in the market in 2025.
• By clinical phase, the Phase III & pivotal/confirmatory segment is expected to grow at the fastest CAGR in the market during the forecast period.
• By route of administration, the intravenous/systemic segment contributed the biggest revenue share of approximately 40% in the market in 2025.
• By route of administration, the intrathecal/intracerebroventricular segment is expected to grow with the highest CAGR in the market during the studied years.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Market Overview
Gene Therapy Clinical Trials are Enhancing Precision in Therapeutics
The gene therapy clinical trials market is experiencing a momentous growth due to robust research in major therapeutic areas, including oncology, autoimmune diseases, inflammation, central nervous system disorders, cardiovascular diseases, metabolic diseases, and endocrinology. The development of gene therapy requires close coordination between manufacturing, preclinical, and clinical teams. They are empowered to advance research and development, clinical research, manufacturing, and commercialization solutions to help researchers transform patient lives. The industries adopt comprehensive and integrated approaches to bring an advanced therapy to market.
The Gene Therapy Clinical Trials Market: Regional Analysis
|
Region |
Share (%) |
|
North America (Dominant) |
46% |
|
Asia Pacific (Fastest Growing) |
25% |
|
Europe |
15% |
|
South America |
7% |
|
Middle East & Africa |
7% |
North America dominated the market share by 46% in 2025, owing to a strong regulatory environment for chemistry, manufacturing, and controls, and government programs for regenerative medicine, which are boosting innovations and reducing development timelines. The Centers for Medicare & Medicaid Services (CMS) launched a program to manage the high costs of newly approved cell and gene therapies. The Center for Biologics Evaluation and Research (CBER) and the Center for Drug Evaluation and Research (CDER) supported the rare disease innovation hub of the U.S. FDA to streamline the review of products for small patient populations.
U.S. Market Analysis
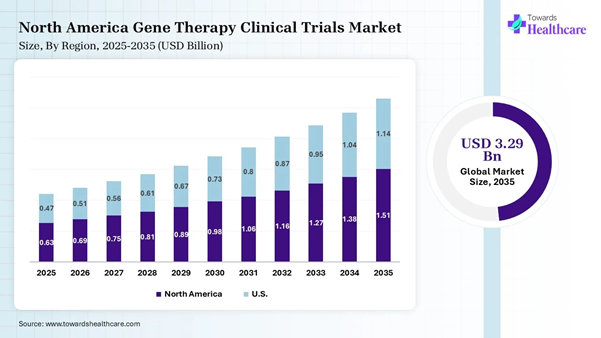
The gene therapy clinical trials market in the U.S. to reach USD 1.14 billion in 2035, gaining traction due to a robust research pipeline in cell and gene therapies, supportive regulatory frameworks, and the rise of CRISPR Cas9 gene editing. The U.S. Food and Drug Administration (FDA) published guidance on innovative cell and gene therapy trial designs for cell and gene therapy studies.
Asia Pacific is expected to grow at the fastest rate in the market during the forecast period due to major investments in GMP-compliant manufacturing hubs, advances in cold chain logistics, and rising public-private partnerships. The global credibility, robust infrastructure, and experienced site management organizations in Japan are critical to manage the operational demands for CAR-T cell therapy and other advanced therapies. China is also a rapidly growing market for cell and gene therapies due to growing investments in innovation and rapid regulatory reforms.
India Market Trends
The gene therapy clinical trials market in India is advancing due to cost competitiveness, decentralized clinical trials, AI-driven research, and the expansion of clinical sites. The Indian pharmaceutical industry is investing in research, developing complex and high-value products such as biosimilars and biopharmaceuticals.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
The Gene Therapy Clinical Trials Market: Segmentation Analysis
Therapy Modality Insights
|
Segments |
Share (%) |
|
In vivo Gene Therapy |
55% |
|
Ex vivo Gene-modified Cell Therapy |
45% |
The in vivo gene therapy segment dominated the gene therapy clinical trials market share by 55% in 2025, owing to the increased focus of researchers on developing therapies to treat muscular deficiencies, metabolic disorders, lysosomal storage diseases, and others. These therapies are applicable in gene replacement, gene silencing, and gene modification. They are also used to treat eye disorders, neuromuscular diseases, liver conditions, and central nervous system disorders.
The ex vivo gene therapy segment is expected to grow rapidly in the gene therapy clinical trials market during the forecast period due to its key therapeutic roles in treating blood and immune disorders, cancer immunotherapy, and correcting genetic skin diseases. The more efficient ex vivo techniques are CRISPR Cas 9 and electroporation than in vivo techniques due to fewer physical barriers. The ex vivo gene therapies are ideal to treat or cure cancers and genetic diseases.
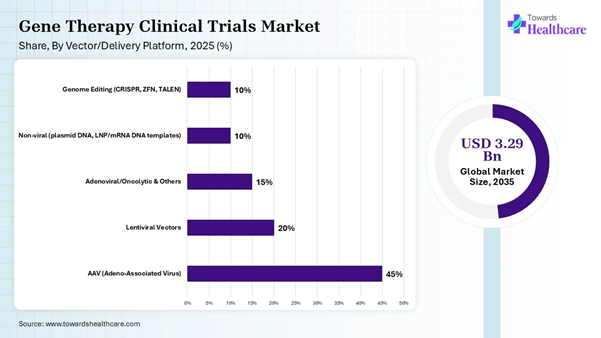
Vector/Delivery Platform Insights
|
Segments |
Share (%) |
|
AAV (Adeno-Associated Virus) |
45% |
|
Lentiviral Vectors |
20% |
|
Adenoviral/Oncolytic & Others |
15% |
|
Non-viral (plasmid DNA, LNP/mRNA DNA templates) |
10% |
|
Genome Editing (CRISPR, ZFN, TALEN) |
10% |
The AAV (adeno-associated virus) segment led the gene therapy clinical trials market share by 45% in 2025, owing to the wide use of AAV vectors in trials for gene replacement, and is ideal for targeting long-lived tissues. The clinical trials are expanding beyond gene replacement into silencing of toxic genes and gene editing. The AAV vectors are used for producing high-quality viral vectors in large quantities for common diseases.
The genome editing segment is expected to grow at the fastest rate in the gene therapy clinical trials market during the studied period due to its great impact in drug discovery, therapeutics, diagnostics, and disease models. The use of CRISPR Cas9 genome editing helps to combat disease transmission by insects. The modifications in the genetic code enable scientists to protect the environment by advancing biofuel research.
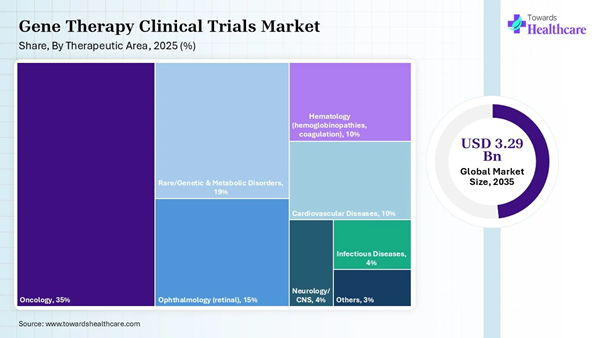
Therapeutic Area Insights
|
Segments |
Share (%) |
|
Oncology |
35% |
|
Rare/Genetic & Metabolic Disorders |
19% |
|
Ophthalmology (retinal) |
15% |
|
Hematology (hemoglobinopathies, coagulation) |
10% |
|
Neurology/CNS |
4% |
|
Cardiovascular Diseases |
10% |
|
Infectious Diseases |
4% |
|
Others |
3% |
The oncology segment dominated the gene therapy clinical trials market share by 35% in 2025, owing to the role of gene therapy as one of the potential strategies for the treatment of cancer. Gene therapy has great potential to employ different genes, such as any suicidal gene, anti-angiogenesis, siRNA gene, immunotherapeutic gene, oncolytic gene, pro-apoptotic gene, and gene-directed enzyme prodrug. Advancements in gene transfer technologies introduced many innovative strategies for the treatment of cancer.
The neurology/CNS segment is expected to grow rapidly in the gene therapy clinical trials market in the coming years due to the emergence of gene therapy as a promising therapeutic method, which enables scientists to directly target genetic mechanisms. Advances in viral and non-viral vector structures are used in gene therapy for treating neurodegenerative conditions. These conditions include Parkinson’s disease, Alzheimer’s disease, Huntington’s disease, amyotrophic lateral sclerosis, and multiple sclerosis.
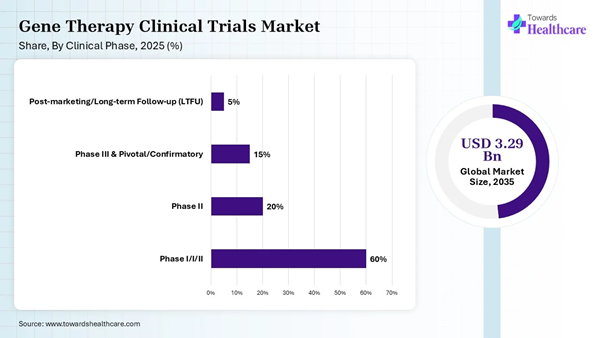
Clinical Phase Insights
|
Segments |
Share (%) |
|
Phase I/I/II |
60% |
|
Phase II |
20% |
|
Phase III & Pivotal/Confirmatory |
15% |
|
Post-marketing/Long-term Follow-up (LTFU) |
5% |
The phase I/ I/II segment dominated the gene therapy clinical trials market share by 60% in 2025, owing to the integral functions of phase I clinical trials in safety monitoring, pharmacokinetics, dose finding, and feasibility. The phase I/II combined trials increase efficiency and reduce development time and costs for severe health conditions. Phase I/II combined trials are used to test a new genetic treatment in humans.
The phase III & pivotal/confirmatory segment is expected to be the fastest-growing in the gene therapy clinical trials market during the predicted timeframe, due to the importance of these trials in ensuring the product’s safety and effectiveness for regulatory approval. These trials primarily provide therapeutic benefits and avoid risks in the patient population. They also measure complex biomarkers such as cell engraftment, gene expression, and specific protein levels, and provide functional clinical outcomes.
Elevate your healthcare strategy with Towards Healthcare. Enhance efficiency and drive better outcomes schedule a call today: https://www.towardshealthcare.com/schedule-meeting
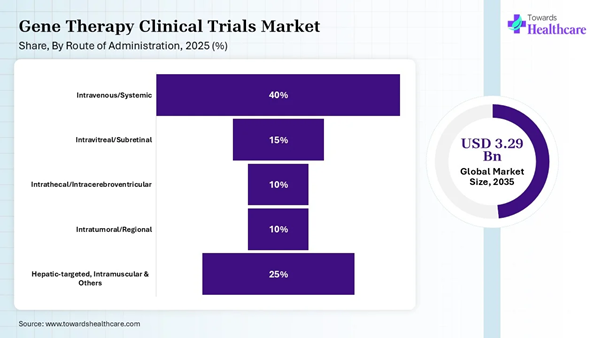
Route of Administration Insights
|
Segments |
Share (%) |
|
Intravenous/Systemic |
40% |
|
Intravitreal/Subretinal |
15% |
|
Intrathecal/Intracerebroventricular |
10% |
|
Intratumoral/Regional |
10% |
|
Hepatic-targeted, Intramuscular & Others |
25% |
The intravenous/systemic segment led the gene therapy clinical trials market share by 40% in 2025, owing to the key role in targeting systemic diseases like Hemophilia A and B. The intravenous (IV) or systemic route of administration is mostly preferred for its minimally invasive nature over surgical local injections. It is also used to treat neuromuscular disorders like Duchenne Muscular Dystrophy.
The intrathecal/intracerebroventricular segment is expected to be the fastest-growing in the gene therapy clinical trials market during the upcoming period due to long-term use of these delivery methods to treat several central nervous system disorders. Research suggests that the intracerebroventricular route is a powerful method for prolonged central nervous system drug delivery in adult and pediatric populations. Intrathecal delivery methods enable the dispensing of soluble therapeutics directly into the central nervous system.
The Gene Therapy Clinical Trials Market Companies
• Regeneron
• Sarepta Therapeutics
• Bluebird bio
• CRISPR Therapeutics
• Editas Medicine
• Novartis
• Roche/Genentech
• Pfizer
• Biogen
• Intellia Therapeutics
• Verve Therapeutics
• Sana Biotechnology
• Beam Therapeutics
Access our comprehensive healthcare dashboard for detailed market insights, segment breakdowns, regional performance, and company profiles: https://www.towardshealthcare.com/access-dashboard
Segments Covered in the Report
By Therapy Modality
• In vivo Gene Therapy
• Ex vivo Gene-modified Cell Therapy
By Vector/Delivery Platform
• AAV (Adeno-Associated Virus)
• Lentiviral Vectors
• Adenoviral/Oncolytic & Others
• Non-viral (plasmid DNA, LNP/mRNA DNA templates)
• Genome Editing (CRISPR, ZFN, TALEN; incl. AAV/LNP delivered)
By Therapeutic Area
• Oncology
• Rare/Genetic & Metabolic Disorders
• Ophthalmology (retinal)
• Hematology (hemoglobinopathies, coagulation)
• Neurology/CNS
• Cardiovascular Diseases
• Infectious Diseases
• Others
By Clinical Phase
• Phase I/ I/II
• Phase II
• Phase III & Pivotal/Confirmatory
• Post-marketing/Long-term Follow-up (LTFU) cohorts
By Route of Administration
• Intravenous/Systemic
• Intravitreal/Subretinal
• Intrathecal/Intracerebroventricular
• Intratumoral/Regional
• Hepatic-targeted, Intramuscular & Others
By Region
• North America
• Asia Pacific
• Europe
• Latin America
• Middle East and Africa (MEA)
Immediate Delivery Available | Buy This Research Report Now @ https://www.towardshealthcare.com/checkout/6068
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region: +44 778 256 0738
North America Region: +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Our Trusted Data Partners
Precedence Research | Towards Packaging | Towards Food and Beverages | Towards Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Browse More Insights of Towards Healthcare:
The gene therapy platform market size was valued at US$ 2.25 billion in 2025 and is projected to grow to 2.59 billion in 2026. Forecasts suggest it will reach approximately US$ 9.29 billion by 2035, registering a CAGR of 15.23% during the period.
The global cell and gene therapy cold chain logistics market size is calculated at US$ 1.89 billion in 2024, grew to US$ 2.19 billion in 2025, and is projected to reach around US$ 8.06 billion by 2034. The market is expanding at a CAGR of 15.64% between 2025 and 2034.
The global cell and gene therapy isolator market size is calculated at USD 1.35 billion in 2024, grew to USD 1.52 billion in 2025, and is projected to reach around USD 4.47 billion by 2034. The market is expanding at a CAGR of 12.54% between 2025 and 2034.
The global gene therapy cell culture media market size began at US$ 214.42 billion in 2025 and is forecast to rise to US$ 237.66 billion by 2026. By the end of 2035, it is expected to surpass US$ 600.11 billion, growing steadily at a CAGR of 10.84%.
The viral vector gene therapy market size reached US$ 13.14 billion in 2024 and is anticipate to increase to US$ 14.62 billion in 2025. By 2034, the market is forecasted to achieve a value of around US$ 38.39 billion, growing at a CAGR of 11.23%.
The AAV gene therapy market size was estimated at US$ 3.85 billion in 2025, projected to increase to US$ 5.4 billion in 2026 and reach US$ 112.24 billion by 2035, showing a healthy CAGR of 40.1% across the forecast years.
The U.S. cell and gene therapy CRO market size was estimated at USD 2.33 billion in 2025 and is predicted to increase from USD 2.56 billion in 2026 to approximately USD 6.02 billion by 2035, expanding at a CAGR of 9.95% from 2026 to 2035.
The AAV manufacturing service market size touched US$ 1.44 billion in 2025, with expectations of climbing to US$ 1.66 billion in 2026 and hitting US$ 6.25 billion by 2035, driven by a CAGR of 15.84% over the forecast period.
The global cell & gene therapy logistics market size is estimated at US$ 6.41 billion in 2025, is projected to grow to US$ 7.76 billion in 2026, and is expected to reach around US$ 43.14 billion by 2035. The market is projected to expand at a CAGR of 21% between 2026 and 2035.
The global cell and gene therapy thawing equipment market size recorded US$ 0.96 billion in 2024, set to grow to US$ 1.1 billion in 2025 and projected to hit nearly US$ 3.56 billion by 2034, with a CAGR of 14.24% throughout the forecast timeline.


