According to Nova One Advisor, the global cell and gene therapy CDMO market is estimated to be valued at USD 13.28 billion in 2026 and is expected to hit around USD 125.09 billion by 2035, exhibiting a compound annual growth rate (CAGR) of 28.3% from 2026 to 2035.
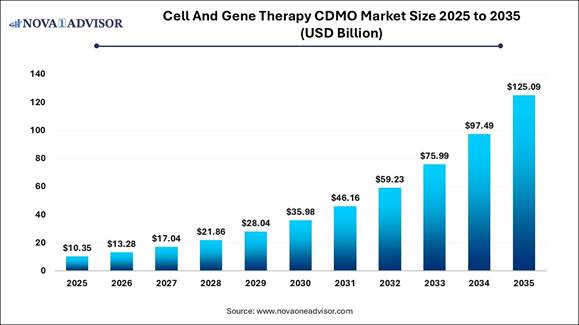
The cell and gene therapy CDMO market is rapidly evolving from manual processes to a digital-first ecosystem. This transition is accelerating development timelines from years to months by utilizing AI-driven analytics, modular automation, and advanced viral vector platforms. By moving toward standardized, GMP-compliant, and data-connected production methods, the sector is effectively addressing high costs and logistical challenges, making personalized cell and gene therapies scalable, reliable, and curative for patients worldwide.
Cell and Gene Therapy CDMO Market Key Takeaways
· North America dominated with the largest market share in 2025.
· Asia Pacific is anticipated to have the fastest growth with a notable CAGR during the forecast period.
· By phase, the pre-clinical segment contributed the highest market share in 2025.
· By phase, the clinical segment is growing at a strong CAGR between 2026 and 2035.
· By product type, the gene therapy segment accounted for the largest market share in 2025.
· By product type, the gene-modified cell therapy segment is projected to grow at a solid CAGR between 2026 and 2035.
· By indication, the oncology segment held a major market share in 2025.
· By indication, the rare diseases segment is expected to expand at a notable CAGR from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Sample Pages of this Report@ https://www.novaoneadvisor.com/report/sample/8435
Market Overview: The Accelerating Future of Advanced Therapies
The cell and gene therapy CDMO market consists of specialized organizations that provide outsourced services, including viral vector production, plasmid DNA, and cell processing for advanced therapies. It is experiencing rapid growth as biotech firms increasingly outsource complex, small-batch manufacturing to specialized partners, primarily due to rising cancer rates and an increase in clinical trials. This sector is transforming to meet high demand by employing advanced GMP-compliant services for viral vector and cell processing, facilitating the next generation of precision medicine.
Integration of Artificial Intelligence (AI) and Automation: A Major Potential
Incorporating AI and advanced automation into CDMO platforms is revolutionizing bioprocessing. This integration transforms manual, error-prone workflows into predictive, closed-loop GMP systems. This technological advancement optimizes critical steps, from cell culture to viral vector yields, enabling real-time monitoring, significantly reducing batch failures, and shortening time-to-market for complex therapies. By leveraging AI-driven insights to enhance efficiency, scalability, and quality, CDMOs are evolving from traditional service providers into strategic, data-driven partners in drug development.
AAV’s Scaling Bottleneck: A Major Limitation
The large-scale adeno-associated virus (AAV) production market is currently facing a significant bottleneck. Slow traditional capsid-ratio and genome integrity tests extend lot release times to up to four weeks. This analytical delay hampers overall campaign throughput, increases costs, and substantially delays clinical and commercial timelines. In response, CDMOs are compelled to invest in costly inline analytics and AI-driven platforms, which, while essential for future growth, could potentially squeeze profitability over the next 2 to 4 years.
Buy Now Full Report: https://www.novaoneadvisor.com/report/checkout/8435
Cell And Gene Therapy CDMO Market Report Scope
|
Report Attribute |
Details |
|
Market Size in 2026 |
USD 13.28 Billion |
|
Market Size by 2035 |
USD 125.09 Billion |
|
Growth Rate From 2026 to 2035 |
CAGR of 28.3% |
|
Base Year |
2025 |
|
Forecast Period |
2026 to 2035 |
|
Segments Covered |
Phase, Product Type, Indication, Region |
|
Market Analysis (Terms Used) |
Value (US$ Million/Billion) or (Volume/Units) |
|
Report Coverage |
Revenue forecast, company ranking, competitive landscape, growth factors, and trends |
|
Key Companies Profiled |
Lonza; Catalent, Inc; Cytiva; Samsung Biologics; Thermo Fisher Scientific Inc.; Novartis AG; WuXi AppTec; AGC Biologics; OmniaBio; Rentschler Biopharma SE; Charles River Laboratories |
For more information, visit the Nova One Advisor website or email the team at sales@novaoneadvisor.com | Call us: +1 804 420 9370
Cell and Gene Therapy CDMO Market: Segmental Analysis
By Phase Analysis
The pre-clinical segment led the market in 2025, primarily due to a substantial pipeline of early-stage therapeutic candidates, high outsourcing demand from biotech startups lacking in-house capabilities, and the pressing need for specialized vector design and small-scale manufacturing services. Small to mid-sized biotech companies, which are key players in the innovation pipeline, heavily rely on CDMOs for early-stage development to avoid significant capital investments in facilities and expertise. Early-stage development requires considerable optimization of manufacturing processes.
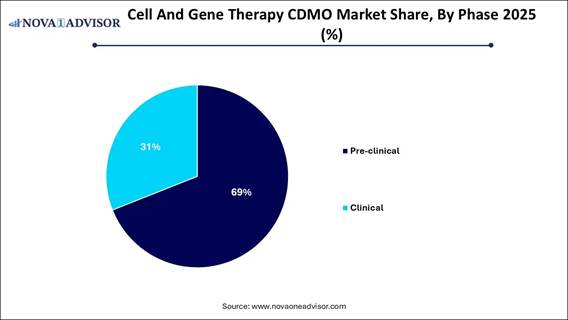
The clinical segment is anticipated to experience the fastest growth during the forecast period. This is largely driven by a surge in Phase II and III trials and the increasing requirement for specialized and compliant manufacturing. A growing number of CAR-T and gene editing studies necessitate specialized manufacturing capabilities. Public and private investments, including those from the NIH and NHGRI, are facilitating funding to advance compounds into clinical trials, which are essential for validating, scaling up, and managing complex analytical needs in clinical stages.
By Product Type Analysis
The gene therapy segment was the leading market segment in 2025. This growth is attributed to the rapid expansion of viral vector production for rare genetic diseases, neurological disorders, and cancer. The surge in gene therapy research and development, particularly targeting complex genetic disorders, requires outsourced expertise from CDMOs to manage complex viral vector production. An increase in FDA and EMA approvals, along with accelerated approval pathways, is driving demand for commercial-scale manufacturing of gene therapies.
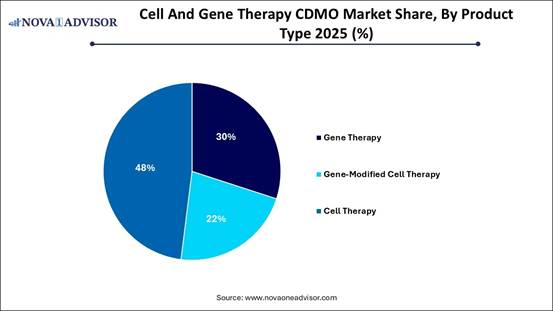
The gene-modified cell therapy segment is expected to experience the fastest growth during the forecast period. This is mainly fueled by rising demand for CAR-T, TCR-T, and NK cell therapies, along with increased investments in research and development and advances in gene-editing technologies. Many companies lack the capabilities for complex gene modifications, leading them to seek partnerships with CDMOs for viral vector supply, genetic engineering, and large-scale GMP manufacturing. Higher approval rates for novel cell-based therapies are encouraging additional investment in this sector.
By Indication Analysis
The oncology segment dominated the market in 2025, primarily due to the high prevalence of cancer, increasing demand for personalized CAR T-cell therapies, and growing investment in research and development for solid tumors. A significant rise in investigational new drug applications for oncology, with a shift from early to late-phase trials, is creating substantial demand for CDMOs to manage viral vector production, cell expansion, and large-scale GMP manufacturing. Startups, in particular, rely on CDMOs for expertise in gene editing, viral vector supply, and cell engineering.
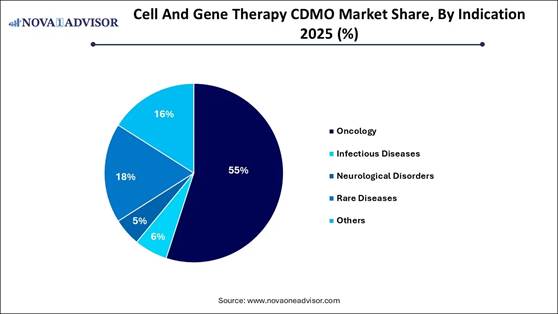
The rare diseases segment is projected to experience the fastest growth during the forecast period. This growth is largely driven by high demand for specialized manufacturing of therapies for genetic, metabolic, and neuromuscular disorders. Treatments for rare diseases often require specialized, high-expertise manufacturing, such as viral vector manufacturing and complex bioprocessing, which biotechnology firms predominantly outsource. The market is focusing on genetic disorders and enzyme restoration, leading to valuable partnerships for clinical and commercial production.
Cell and Gene Therapy CDMO Market: Regional Analysis
North America is projected to dominate the market by 2025, driven by substantial investment in biotechnology, a high concentration of clinical trials, and a supportive regulatory environment that accelerates approval processes. The region hosts the largest number of cell and gene therapy clinical trials globally, especially in the pre-clinical and early clinical stages, necessitating extensive outsourcing to CDMOs. The FDA's accelerated approval pathways, including the Regenerative Medicine Advanced Therapy designation, Orphan Drug Status, and breakthrough designations, facilitate quicker commercialization for specialized manufacturing.
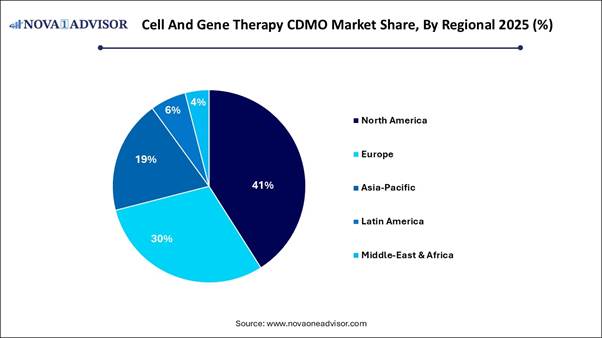
The U.S. stands at the forefront of the global market, equipped with the largest concentration of clinical trials and a mature biotech ecosystem backed by significant investments and top-tier talent. Fueled by the FDA’s expedited regulatory pathways and advancements in automation, key players such as Catalent and Thermo Fisher are setting new standards in scaling high-complexity therapies, ensuring the U.S. remains a central hub for cell and gene therapy commercialization.
· In March 2026, Lonza expanded its strategic partnership with Genetix Biotherapeutics, increasing capacity at its Houston site to meet the growing demand for ZYNTEGLO™, the FDA-approved gene therapy for transfusion-dependent beta-thalassemia.
Canada is emerging as a strategic hub in North America, specializing in automated manufacturing, autologous therapies, and bridging the gap between research and GMP-compliant production. Supported by key institutions like CCRM and new specialized facilities, Canada is solidifying its position as a high-tech partner for scaling advanced therapies.
Asia Pacific region is expected to experience the fastest growth during the forecast period. This growth is driven by high patient demand, favorable regulatory reforms, and cost-effective manufacturing capabilities. Governments in China, Japan, and South Korea are significantly investing in biotech infrastructure and establishing welcoming regulatory frameworks. A surge in local startups and the growing need to address domestic patients' chronic illnesses have created a vibrant biotech ecosystem that relies on CDMOs for scale-up, making this region a viable alternative to Western hubs.
India is rapidly establishing itself as the leading global hub for cell and gene therapy CDMOs. It offers a high-quality, lower-cost alternative to China, supported by a pool of English-speaking technical talent and advanced single-use automation. Amid geopolitical shifts, this sector, especially the lucrative pre-clinical development phase, is attracting significant investments in artificial intelligence and analytics to accelerate innovation.
· In January 2026, Eyestem Research received approval from the Central Drugs Standard Control Organization (CDSCO) to begin Phase 2 clinical trials for Eyecyte-RPE™ in India. This pioneering cell therapy aims to treat dry age-related macular degeneration (Dry AMD).
China, with major players like WuXi Biologics and Pharmaron, currently holds a substantial share of the global market. This dominance is reinforced by strategic government policies and extensive infrastructure investments. By leveraging state-backed capital to build large, compliant facilities, China has transitioned from being a local manufacturer to a dominant global player.
Cell and Gene Therapy CDMO Market Companies
· Lonza
· Cytiva
· Samsung Biologics
· Thermo Fisher Scientific Inc.
· Novartis AG
· WuXi AppTec
· OmniaBio
· Rentschler Biopharma SE
· Charles River Laboratories
Emerging Government Initiatives in the Cell and Gene Therapy CDMO Market
|
Emerging Initiative |
Focus Area |
Programs |
|
National Bio-manufacturing Policies |
Scaling up production, reducing import dependency, and fostering indigenous manufacturing. |
India: BioE3 (Biotechnology for Economy, Environment, and Employment) Policy. |
|
Specialized Manufacturing Infrastructure |
Building specialized CGT centers, GMP facilities, and cell manipulation capacity. |
UK: Advanced Therapy Treatment Centres (ATTCs) funded by Innovate UK. India: BIRAC support for local CAR-T (NexCAR19) production. |
|
Accelerated Regulatory Pathways |
Fast-tracking approvals for advanced therapies to speed up time-to-market. |
Japan: Sakigake designation system for regenerative medicines. USA: FDA CMC flexibility initiatives. |
|
Public-Private Partnerships (PPP) |
Co-developing therapies and manufacturing technologies, combining academic innovation with industrial scalability. |
EU: Innovative Medicines Initiative (IMI) projects (e.g., STEMBANCC, EBiSC). India: CSIR-IGIB and Serum Institute partnership. |
Major Shifts in the Cell and Gene Therapy CDMO Market
· In June 2025, uBriGene Biosciences launched fully characterized induced pluripotent stem cell (iPSC) banks using its proprietary RNA-LNP reprogramming technology. These GMP-compliant banks, derived from healthy donor fibroblasts, are available for research and clinical applications, significantly cutting down the time and uncertainty around donor cell sourcing. Each bank undergoes rigorous quality testing to ensure identity, stability, and purity.
· In April 2025, AGC Biologics will introduce a new Cell and Gene Technologies Division under Luca Alberici, focusing on innovation, scalable manufacturing, and customer experience. The Milan facility will serve as the central hub, boasting extensive experience and numerous commercial approvals, establishing a strong manufacturing pathway from IND to commercialization.
· In May 2025, Astraveus SAS announced plans for a novel CAR-T manufacturing platform while a new global CDMO was launched to tackle industry challenges. Aspen Neuroscience will present promising data for its Parkinson’s disease trial at the ISCT 2025 meeting. Astraveus is collaborating with NecstGen to assess its Lakhesys Benchtop Cell Factory™ for efficient CAR-T manufacturing, aiming to lower costs and improve efficiency.
Related Report –
➡️ Topical Drugs CDMO Market – Industry Size, Growth Trends, and Forecast Analysis
➡️ Active Pharmaceutical Ingredients CDMO Market – Market Insights, Demand Outlook, and Competitive Landscape
➡️ U.S. Pharmaceutical CDMO Market – Regional Growth, Opportunities, and Industry Analysis
➡️ U.S. Small Molecule Innovator API CDMO Market – Market Trends, Innovation Pipeline, and Forecast
➡️ Advanced Therapy Medicinal Products CDMO Market – Emerging Therapies, Market Growth, and Future Outlook
➡️ Biologics CDMO Market – Industry Expansion, Key Drivers, and Forecast Analysis
➡️ Pharmaceutical CDMO Market – Global Market Size, Trends, and Strategic Insights
➡️ U.S. Advanced Therapy Medicinal Products CDMO Market – Regional Analysis, Growth Drivers, and Opportunities
➡️ Investigational New Drug CDMO Market – Pipeline Development, Market Trends, and Forecast
➡️ Active Pharmaceutical Ingredients CDMO Market – Production Trends, Market Dynamics, and Growth Outlook
➡️ Veterinary CRO And CDMO Market – Animal Healthcare Trends, Market Growth, and Industry Insights
➡️ U.S. Active Pharmaceutical Ingredients CDMO Market – Regional Market Analysis, Trends, and Forecast
➡️ Oligonucleotide CDMO Market – Innovation Trends, Market Expansion, and Future Opportunities
➡️ Biopharmaceutical CDMO Market – Industry Overview, Growth Drivers, and Competitive Analysis
➡️ North America Topical Drugs CDMO Market – Regional Insights, Market Trends, and Forecast Analysis
Segments Covered in the Report
This report forecasts revenue growth at country levels and provides an analysis of the latest industry trends in each of the sub-segments from 2021 to 2035. For this study, Nova one advisor, Inc. has segmented the cell and gene therapy CDMO market.
By Phase
· Pre-clinical
· Clinical
By Product Type
· Gene Therapy
o Ex-vivo
o In-vivo
· Gene-Modified Cell Therapy
o CAR T-cell Therapies
o CAR-NK Cell Therapy
o TCR-T Cell Therapy
o Other
· Cell Therapy
By Indication
· Oncology
· Infectious Diseases
· Neurological Disorders
· Rare Diseases
· Others
By Region
· North America
o U.S.
o Canada
· Asia Pacific
o China
o Japan
o India
o South Korea
o Thailand
· Europe
o Germany
o UK
o France
o Italy
o Spain
o Sweden
o Denmark
o Norway
· Latin America
o Brazil
o Mexico
o Argentina
· Middle East and Africa (MEA)
o South Africa
o UAE
o Saudi Arabia
o Kuwait
Immediate Delivery Available | Buy This Premium Research https://www.novaoneadvisor.com/report/checkout/8435
About-Us
Nova One Advisor is a global leader in market intelligence and strategic consulting, committed to delivering deep, data-driven insights that power innovation and transformation across industries. With a sharp focus on the evolving landscape of life sciences, we specialize in navigating the complexities of cell and gene therapy, drug development, and the oncology market, enabling our clients to lead in some of the most revolutionary and high-impact areas of healthcare.
Our expertise spans the entire biotech and pharmaceutical value chain, empowering startups, global enterprises, investors, and research institutions that are pioneering the next generation of therapies in regenerative medicine, oncology, and precision medicine.
Web: https://www.novaoneadvisor.com/
Our Trusted Data Partners:
Nova One Advisor - Market Research Reports & Consulting Firm
Nova One Advisor offers comprehensive market research reports with in-depth industry analysis and market data.
Call us: +1 804 420 9370
Email: sales@novaoneadvisor.com
Web: https://www.novaoneadvisor.com/
You can place an order or ask any questions, please feel free to contact at sales@novaoneadvisor.com | +1 804 441 9344


