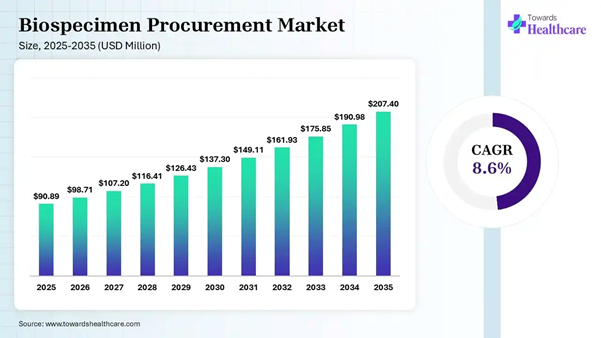
The global biospecimen procurement market size was estimated at USD 90.89 million in 2025 and is predicted to increase from USD 98.71 million in 2026 to approximately USD 207.40 million by 2035, expanding at a CAGR of 8.60% from 2026 to 2035.
The Complete Study is Now Available for Immediate Access | Download the Free Sample Pages of this Report @ https://www.towardshealthcare.com/download-sample/5573
The leading companies are making efforts to connect researchers to the patients and specimens to simplify biospecimen procurement. These specimens include banked research samples, remnant clinical or pathology samples, or custom research collections. The prominent ways to streamline bioprocurement are extensive access to banked biospecimens, custom specimen collections, collection consistency, confidence, a single point of compliance, and contracting. The expanding services in biospecimen procurement include biospecimen data and data management, study design, sample kits, logistics, and a variety of clinical and pathology laboratory services.
The Biospecimen Procurement Market: Highlights
• North America dominated the global biospecimen procurement market in 2025.
• Asia Pacific is expected to be the fastest-growing region during the forecast period.
• By type of biospecimen for oncological studies, the FFPE segment dominated the market in 2025.
• By type of biospecimen for non-oncological studies, the blood product segment dominated the global biospecimen procurement market in 2025.
Market Overview
Streamlined Procurement and Processing of Biospecimen Samples
The procurement of biospecimens includes biofluids, solid samples, and matched-pair samples, such as whole blood, non-blood-derived fluids, FFPE and frozen tissues, buffy coat samples, plasma samples, etc. The plasma samples are critical in the biospecimen procurement market, which offer ways to meet research needs in oncology, which are also used in plasma DNA isolation, bioassays, biomarker discovery, and proteomic analysis. The plasma samples are essential in liquid biopsy, genetic discovery, genetic analysis, genetic research, and applications. The biospecimen samples also include fresh frozen tissue samples in the form of arrays, blocks, and slides from human diseased, human normal, and tumor tissue types, which are critical in clinical and molecular oncology research. The specialized biorepository services include temperature-sensitive shipping, HLA typing, and peripheral blood mononuclear cell (PBMC) isolation that ensure the integrity of samples.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
The Emerging Need for Biospecimen Procurement Outsourcing
The major advantages of outsourcing biospecimen procurement to a well-established and experienced biospecimen procurement company are a single point of access to hundreds of biospecimen providers and flexibility and expertise to resolve any problems. Outsourcing these services to an expert saves time and effort and reduces costs across the biospecimen procurement market. Sourcing high-quality R&D is the emerging need in life sciences R&D. Human biospecimens are essential in all stages of the product development lifecycle, including basic research, pre-clinical studies, verification of research studies, and validation of studies.
Featured Biospecimen Categories by Precision for Medicine
|
Sr. No. |
Biospecimen Category |
Types of Biospecimen |
|
|
Blood, biofluids, and derivatives |
• Whole blood • Plasma • Serum • Other biofluids |
|
|
Tissues |
• FFPE tissue • Fresh and frozen tissue • Tissue microarrays • Dissociated tumor cells |
|
|
Viable cells |
• Peripheral blood mononuclear cells (PBMCs) • Bone marrow mononuclear cells (BMMCs) • Dissociated tumor cells • Purified cell subsets |
|
|
Remnant diagnostic specimens |
• Chemistry samples • Respiratory samples • Infectious disease samples • Cerebral spinal fluid (CSF) samples |
|
|
NGS characterized biospecimens |
• Tumor, normal, and adjacent tissues • DNA/RNA • H&E images • Detailed characterization data |
|
|
Custom biospecimen collections |
• Biomarker and drug discovery |
Become a valued research partner with us - https://www.towardshealthcare.com/schedule-meeting
Analysis of the Successful Biospecimen Collection with Precision
• Planning and Preparation: This stage is driven by deep scientific expertise, certified pathologists, and expert clinicians who design precise biospecimen collection protocols, which deliver successful results.
• Sample Collection: This stage ensures a rapid collection process of biospecimens from the internal donor network and an IRB-ready framework.
• Site Processing: This stage ensures consistency of specimen quality and adaptability with kits that feature tailored reagents and instructions for the collection and processing of specimens.
• Downstream Processing: This stage involves fully integrated specialty laboratory services and facilities that allow seamless processing, quality control, and storage of samples.
• Sample Analysis: This stage aims to develop and validate biomarker assays with integrated solutions across several analytical techniques.
• Transportation: This stage involves coordination between precision logistics teams that allow for sample shipping and delivery, and ensure compliance with the biospecimen lifecycle.
Potential Methods for Biospecimen Collection
• Planning: In this stage, certified pathologists and clinicians develop custom collection protocols and ensure high-quality sample acquisition.
• Sample Collection and Logistics: In this stage, a comprehensive operational support is provided to ensure high-quality and consistent collection of biospecimens.
• Custom Processing, Storage, and Analysis: This stage offers detailed laboratory support for advanced biospecimen analysis and data integrity.
Get the latest insights on life science industry segmentation with our Annual Membership: https://www.towardshealthcare.com/get-an-annual-membership
The Biospecimen Procurement Market: Regional Analysis
North America dominated the market in 2025, owing to government and private funding for biomedical research, increasing demand for personalized medicine, and the expansion of biobanking networks. The National Institutes of Health (NIH) published a policy that applies to all human clinical and research biospecimens obtained from U.S. persons. This policy also applies to those biospecimens that are funded or supported by any NIH grants, contracts, cooperative agreements, intramural support, and other transactions. On the other hand, the BIOSECURE Act launched new parameters for biotechnology companies that look for U.S. government funding and procurement. It is a national security measure that addresses the emergence and role of biotechnology supply chains, advanced manufacturing, and genomic data in strategic infrastructure.
For instance,
• In December 2025, iSpecimen secured $5.5 million in funding to support marketing and working capital, and enhance the company’s market competitiveness. This funding was secured through signing a securities purchase agreement with accredited investors.
U.S. Market Analysis
The biospecimen procurement market in the U.S. is experiencing a momentous growth, driven by a surge in clinical trial activities, supportive government initiatives that fuel genomic research, and the growing trend of outsourcing. The Centers for Disease Control and Prevention (CDC) launched the International Reagent Resource (IRR) to offer registered users test kits, reagents, and information for the study and detection of emerging viral and bacterial pathogens, and outbreak response.
For instance,
• In December 2025, Cedars-Sinai Biobank and Research Pathology Resource achieved multi-domain CAP accreditation. The biobank has set global standards in digital pathology, biospecimen research, and organoid innovation.
Asia Pacific is expected to grow at the fastest rate in the market during the forecast period due to the rising demand for targeted therapies, precision medicine, and genomics, modernized healthcare, digitalization, and adoption of digital biobank management systems. The governments are introducing large-scale programs to provide well-annotated and high-quality samples, including blood, tissues, and biofluids. These programs and related government initiatives aim to support the rapidly growing biopharma and precision medicine sectors across this region. The regional biospecimen procurement and biobanking industry is undergoing government-led expansion in countries like China, India, and South Korea.
India Market Trends
The biospecimen procurement market in India witnesses a global hub for clinical trials, a rising burden of chronic conditions like cancer, and the growing preference for personalized treatments. The CSIR-Institute of Genomics and Integrative Biology (IGIB) in New Delhi launched the national biobank, which is a state-of-the-art facility for genomic, clinical, and lifestyle data. Moreover, the Union Cabinet approved the Biotechnology for Economy, Environment, and Employment (BioE3) policy to boost high-performance biomanufacturing.
Access our comprehensive healthcare dashboard for detailed market insights, segment breakdowns, regional performance, and company profiles: https://www.towardshealthcare.com/access-dashboard
The Biospecimen Procurement Market: Segmentation Analysis
Type Insights
By type of biospecimen for oncological studies, the FFPE segment dominated the biospecimen procurement market in 2025, owing to the wide applications of formalin-fixed paraffin-embedded (FFPE) human tissue samples in modern research. They provide detailed information for the study of the development and progression of diseases and treatment efficacy. Patient biopsies in the form of FFPE samples are essential in oncology research to establish a cancer diagnosis, characterize cancers, select treatment options, and estimate treatment efficacy.
By type of biospecimen for non-oncological studies, the blood product segment dominated the global biospecimen procurement market in 2025 due to the importance of various types of blood biospecimens, such as plasma, serum, buffy coat, circulating tumor cells, and peripheral blood mononuclear cells in clinical research and development. They are widely used in the early detection of diseases, precision medicine, and therapeutic monitoring. Blood biospecimens enable molecular profiling to match patients with targeted therapies.
The Biospecimen Procurement Market Companies
• PrecisionMed
• Infinity BiologiX
• Medicove
• REPROCELL
• BioChain Institute
• Discovery Life Sciences
• BioIVT
• National BioService
• Creative Bioarray
• Precision for Medicine
Segments Covered in the Report
By Type
• Oncological Studies
○ Tissue-Based Biospecimens
■ Formalin-Fixed Paraffin-Embedded (FFPE)
■ Frozen Tissue
○ Blood-Based Biospecimens
■ Whole Blood
■ Plasma / Serum
○ Other Biospecimens
• Non-Oncological Studies
○ Tissue-Based Biospecimens
■ Formalin-Fixed Paraffin-Embedded (FFPE)
■ Frozen Tissue
○ Blood-Based Biospecimens
■ Whole Blood
■ Plasma / Serum
○ Other Biospecimens
By Region
• North America
• Asia Pacific
• Europe
• Latin America
• Middle East and Africa (MEA)
Immediate Delivery Available | Buy This Research Report Now @ https://www.towardshealthcare.com/checkout/5573
About Us
Towards Healthcare is a leading global provider of technological solutions, clinical research services, and advanced analytics, with a strong emphasis on life science research. Dedicated to advancing innovation in the life sciences sector, we build strategic partnerships that generate actionable insights and transformative breakthroughs. As a global strategy consulting firm, we empower life science leaders to gain a competitive edge, drive research excellence, and accelerate sustainable growth.
You can place an order or ask any questions, please feel free to contact us at sales@towardshealthcare.com
Europe Region: +44 778 256 0738
North America Region: +1 8044 4193 44
APAC Region: +91 9356 9282 04
Web: https://www.towardshealthcare.com
Our Trusted Data Partners
Precedence Research | Towards Packaging | Towards Food and Beverages | Towards Chemical and Materials | Towards Dental | Towards EV Solutions | Healthcare Webwire
Find us on social platforms: LinkedIn | Twitter | Instagram | Medium | Pinterest
Browse More Insights of Towards Healthcare:
The global gene vector market size is calculated at US$ 9.61 billion in 2025, grew to US$ 11.64 billion in 2026, and is projected to reach around US$ 64.38 billion by 2035. The market is expanding at a CAGR of 21% between 2026 and 2035.
The next-generation metabolic therapies market is on an upward trajectory, poised to generate substantial revenue growth, potentially climbing into the hundreds of millions over the forecast years from 2026 to 2035.
The global rotavirus vaccines market size was estimated at USD 5.25 billion in 2025 and is predicted to increase from USD 5.78 billion in 2026 to approximately USD 13.79 billion by 2035, expanding at a CAGR of 10.14% from 2026 to 2035.
The global recombinant albumin market size was estimated at USD 152.9 million in 2025 and is predicted to increase from USD 162.2 million in 2026 to approximately USD 275.89 million by 2035, expanding at a CAGR of 6.08% from 2026 to 2035.
The global endonucleases market size was estimated at USD 394.3 million in 2025 and is predicted to increase from USD 431.17 million in 2026 to approximately USD 963.86 million by 2035, expanding at a CAGR of 9.35% from 2026 to 2035.
The quantum flex cell expansion system market is rapidly advancing on a scale, with expectations of accumulating hundreds of millions in revenue between 2026 and 2035.
The cell therapy solvent suspension tubing market is experiencing significant expansion, with projections indicating a revenue increase reaching several hundred million dollars by the end of the forecast period, spanning 2026 to 2035.
The global single-use sterile connectors for cell therapy market size is expected to be worth around USD 4.34 Billion by 2035, from USD 1.30 billion in 2025, growing at a CAGR of 12.8% during the forecast period from 2026 to 2035.
The global transduction technologies market size is estimated at US$ 425.47 million in 2025, increased to US$ 466.74 million in 2026, and is expected to reach around US$ 1073.83 million by 2035. The market is growing at a CAGR of 9.7% between 2026 and 2035.
The global rare cell isolation market size is calculated at USD 2.25 billion in 2025, grew to USD 2.47 billion in 2026, and is projected to reach around USD 5.73 billion by 2035. The market is expanding at a CAGR of 9.79% between 2026 and 2035.


